CHEM211 - MODULE 7
CHAPTER 7: QUANTUM THEORY AND ATOMIC STRUCTURE
7.1 The Nature of Light
Light: A form of electromagnetic radiation exhibiting wave properties.
Electromagnetic Radiation Properties:
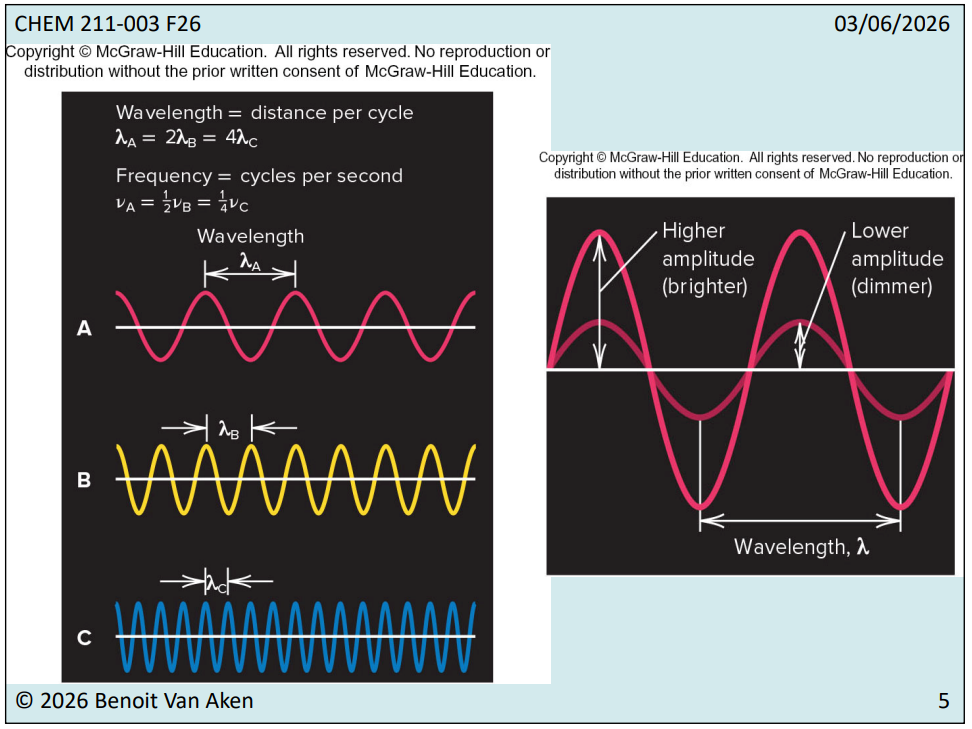
Frequency : Number of waves or cycles per second (unit = 1/second-s^-1 or hertz-Hz)
Wavelength: Distance a wave travels in one cycle (unit = meters-m, nanometers-nm, etc.)
Speed: Distance the radiation travels per unit time (unit = meters/second-m/s)
Amplitude: Height of the wave crest or depth of the trough, representing intensity.
7.2 Relationship Between Frequency and Wavelength
The speed of light (c) is defined by the equation: c = \nu \times \lambda
Where:
lambda = wavelength (m)
nu = frequency (s^-1)
c = speed of light in vacuum (approximately 3.00 x 10^8 m/s).
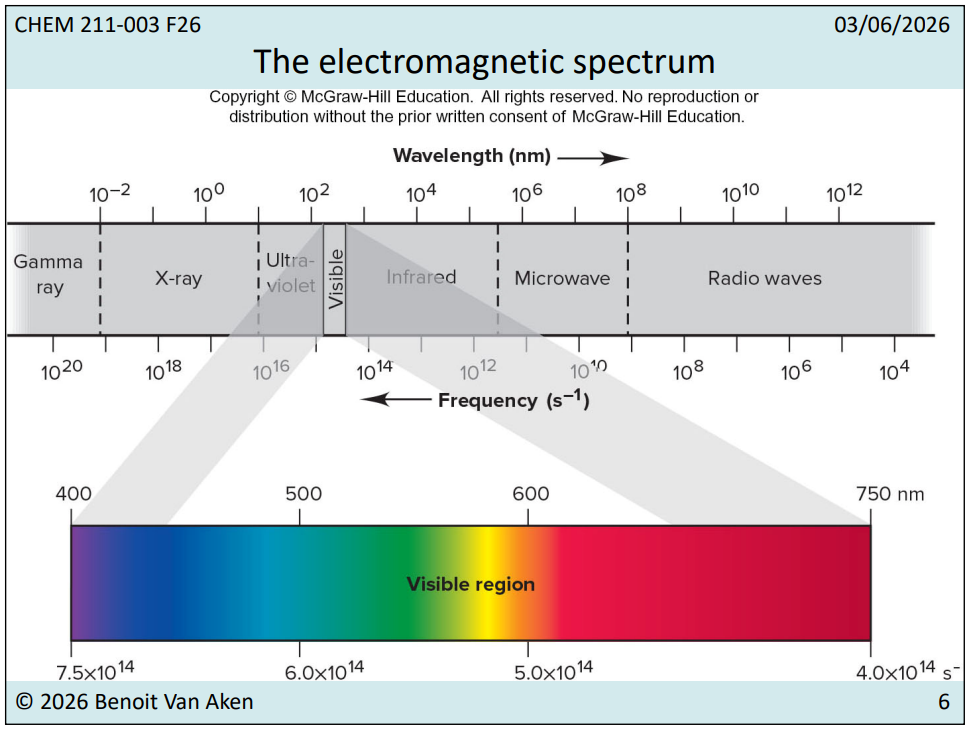
7.3 The Electromagnetic Spectrum
Wavelengths & Frequencies: Explanation of the spectrum ranges, from gamma rays to radio waves, accompanied by corresponding wavelengths in nanometers (nm).
Higher Amplitude means brighter light.
7.4 Distinction Between Energy and Matter
Refraction: Light changes direction at the interface of two media.
Dispersion: Separation of light into its component colors.
Diffraction and Interference: Waves interact and cause patterns based on the wave properties of light.
7.1 The Particle Nature of Light
**Observations Confounding Physicists: **
Blackbody Radiation
Photoelectric Effect
Atomic (Line) Spectra
The need for quantum theory emerged to explain these phenomena.
7.2 Blackbody Radiation
Emission spectrum of heated objects does not conform to classical wave models; energy is quantized into packets (quanta).
7.3 The Photoelectric Effect
Description: When light of sufficient energy interacts with a metal plate, it ejects electrons, creating an electrical current.
Wave Theory Failures: Inability to explain threshold frequency and time lag.
Discovery: Light is made of particles (photons).
7.4 The Line Spectrum
Gases excited by electric currents emit light, resulting in a distinct line spectrum.
7.2 Quantum Theory and the Bohr Model
Max Planck (1858–1947): Introduced the concept of quantized energy levels.
Energy Relation: E = h\nu
Where:
$h$ = Planck's constant ($6.626 \times 10^{-34} \ J \cdot s$)
$\nu$ = frequency
The energy change in an atom is described as:
\Delta E{atom} = E{final} - E{initial} = E{photon} = \Delta n h \nu
7.3 Bohr's Model of the Hydrogen Atom
Three Postulates
The hydrogen atom has only certain stationary energy levels, each correlating to a circular orbit.
Within a stationary state, the atom does not radiate energy.
Transition between stationary states occurs only via absorption or emission of a photon.
Energy Transition: The radius of the orbital relates to $n^2$, indicating stability increases as electrons move closer to the nucleus.
Emission of Photons: Electrons drop from higher energy levels, releasing energy as light, which corresponds with spectral lines.
7.4 Quantum Mechanics and Energy Levels
Rydberg's Equation: \frac{1}{\lambda} = R\left(\frac{1}{n1^2} - \frac{1}{n2^2}\right)
Where: $R = 1.096776 \times 10^7 \ m^{-1}$.
Energy Levels of Hydrogen Atom
Energy of levels:
E = -2.18 \times 10^{-18} J \left(\frac{Z^2}{n^2}\right)
For hydrogen ($Z = 1$):
E_{ground \ state} = -2.18 \times 10^{-18} J \cdot (1/1) = -2.18 \times 10^{-18} JIonization Energy Calculation:
\Delta E = (2.18 \times 10^{-18} J/atom) \cdot (6.022 \times 10^{23} atoms/mol) \cdot (1 kJ/10^3 J) = 1.31 \times 10^3 kJ/mol
7.3 Wave-Particle Duality of Matter and Energy
Wave Properties:
E = mc^2De Broglie's Hypothesis: Matter can exhibit wave properties, leading to the de Broglie wavelength equation:
\lambda = \frac{h}{mu}Particle Nature of Photons:
Momentum ($p$) can be described as:
p = \frac{h}{\lambda}
indicating higher momentum in shorter wavelength photons.
7.4 Heisenberg's Uncertainty Principle
Postulate: It is impossible to simultaneously determine the position and momentum of a particle:
\Delta x \cdot m\Delta u \geq \frac{h}{4\pi}
7.4 Quantum-Mechanical Model of the Atom
Schrödinger Equation: Describes electron position in terms of probability, leading to the concept of the electron cloud.
Quantum Numbers
Four Quantum Numbers:
Principal Quantum Number ($n$): Size and energy of the orbital (positive integer).
Angular Momentum Quantum Number ($l$): Describes shape of the orbital (integer from $0$ to $n-1$).
Magnetic Quantum Number ($m_l$): Positions or orientations of an orbital in space relevant to $l$.
Spin Quantum Number ($m_s$): Specifies electron spin direction (+1/2 or -1/2).
Orbital Shapes Overview:
s (l=0): Spherical.
p (l=1): Double-lobed.
d (l=2): Complex shapes with four lobes.
f (l=3): Even more complex, with multiple lobes and orientations.
Hierarchy of Quantum Numbers: Describing the energy states in atomic orbitals and potential configurations.
Summary of Energy Levels and Sublevels
Each electron is characterized by a unique set of quantum numbers, allowing detailed predictions about the atom's energy states and configurations.