Regulatory T cells
Objectives:
know what a Treg is and their various phenotypes
recall key cytokines and molecules
know how they’re induced and how their effects are mediated
know the consequences of overactivation or lack of Tregs
Definitions:
Regulatory T cells = immunosuppressive T cells with cytokine profiles distinct from Th1 and Th2 cells, specialised in suppression of immune response to both foreign antigens and self
Notes:
4: Evidence of active T-cell mediated regulatory mechanisms was found in the 70s - ‘suppressor T cells’.
Scurfy mice have FoxP3 mutation and suffer severe autoimmune condition.
Irradiation and/or neonatal thymectomy (surgical removal of thymus from newborn mice) → impaired immune response AND autoimmunity. Transfer of normal lymphocytes could prevent disease, whereas transfer of CD25+ deficient T cells into SCID (germ-free) mice → multi-organ autoimmunity
5: There are 4 primary basic mechanisms used by Tregs:
modulate APC maturation and function - APCs often proinflammatory
kill target cells
disrupt metabolic pathways
produce anti-inflammatory cytokines
These mechanisms downregulate immune response but don’t prevent initial T cell activation. They prevent chronic immunopathology
6: Tregs prevent cytokine storms from occurring in response to infection. Although other Th cells have beneficial functions when needed, they also can have harmful effects which need suppressing eg:
Th1 induces autoimmunity eg IBD, MS, T1 diabetes etc
Th2 → allergy
Th17 induces inflammation - induced by IL-17 and TGF-beta, development inhibited by IFN-gamma and IL-4
7: Tregs prevent aggressive autoimmune responses, prevents the immune system responding to harmless antigens and microbes, and controls immune response magnitude to prevent tissue damage
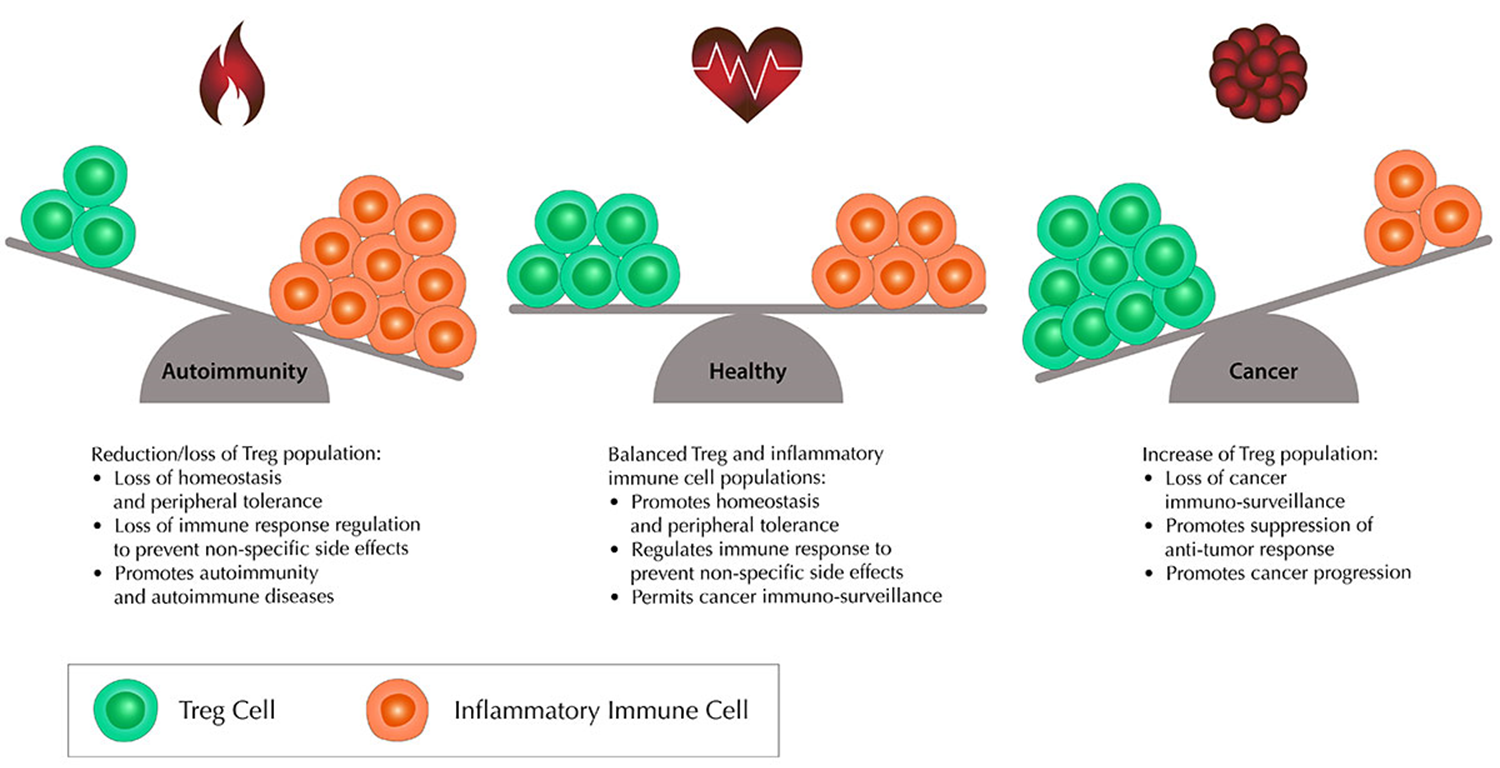
However balance between inflammatory immune cells and Tregs if important. Increased Treg population causes loss of cancer immunosurveillance, promotes suppression of the anti-tumour response, and promotes cancer progression.
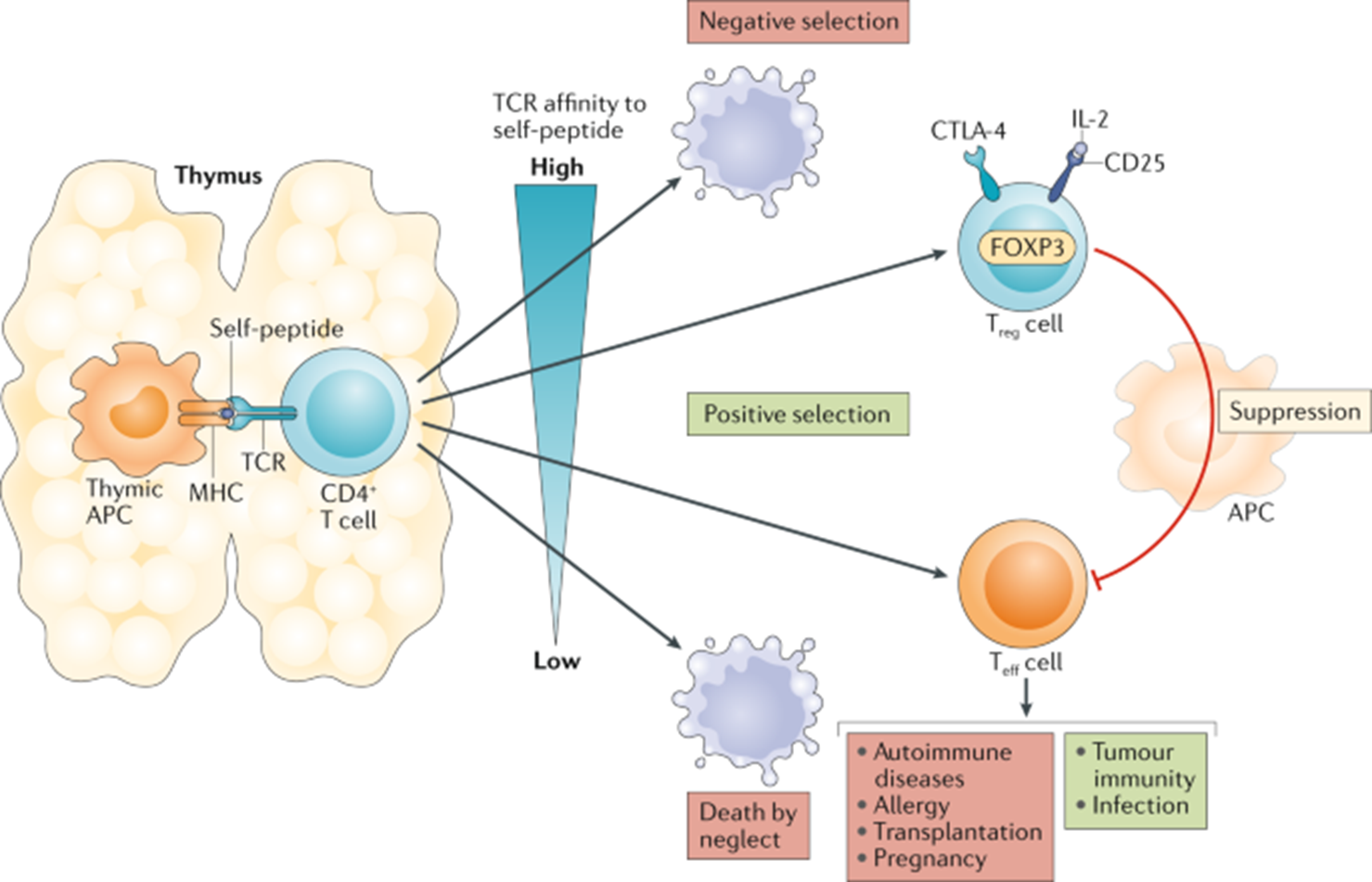
8: T reg selection is seen as a ‘Goldilocks’ process. Very strong signals for self antigen result in apoptotic death. Weak signals results in selection to become either an effector cell, or cell death. Intermediate signal → regulatory cell.
All T cell populations with a particular TCR contain a mixture of effector and regulatory T cells. The relative proportions of these populations is defined by the affinities of the T cell for self-peptide MHC
9:
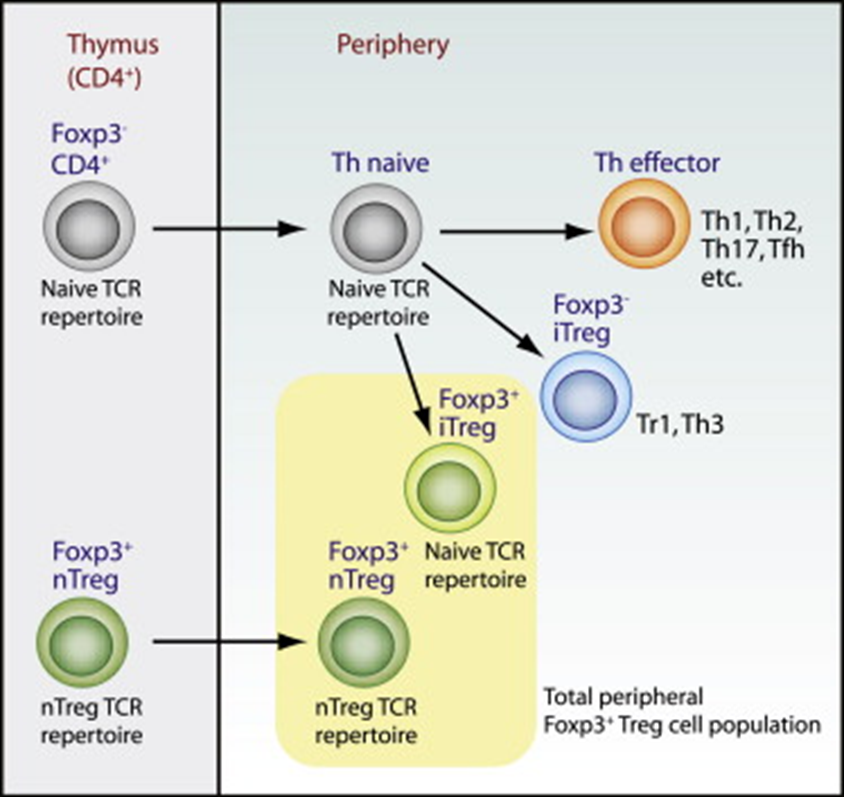
10: Some T cells generated in the thymus but others generated in the periphery. Polarising cytokines required for differentiation into Tregs in the periphery are IL10 and TGF beta
11: FoxP3 is a expressed by many Tregs but isn’t a definitive marker of Tregs. It encodes Forkhead box P3 transcription factor and controls development of Tregs and their function. FoxP3 is likely to control expression of key molecules which mediate suppression. T cells can express FoxP3 transiently after activation. FoxP3 deletion/inactivation causes T cell hyperactivation eg immune dysregulation, polyendocrinopathy or enteropathy X-linked (IPEX) syndrome. IPEX patients exhibit high mortality rate at young age, 70% develop thyroiditis, 90% develop diabetes.
12: Naturally occurring Tregs, aka thymus derived, are educated in the thymus during negative selection and exit as a regulatory population. They express FoxP3 and are CD4+CD25+. Make up 5 to 10% of CD4+ cells. Dynamic response team, adapt to suppress adaptive immune response depending on location found in.
13: Inducible Tregs (iTregs) originate as CD4+ cells from the thymus. They’re induced by TGF beta in the thymus and express FoxP3 and upregulate CD25.
iTreg Tr1 is induced by IL-10, and does not express CD25 or FoxP3. They mediate suppression by IL-10 (primary anti inflammatory cytokine) secretion and are identified by cell-surface expression of LAG-3 and integrin alpha 2 subunit CD49b -identified eg using flow cytometry.
iTreg Th3 cells are induced from naive CD4 T cells by TGF beta and can be FoxP3+
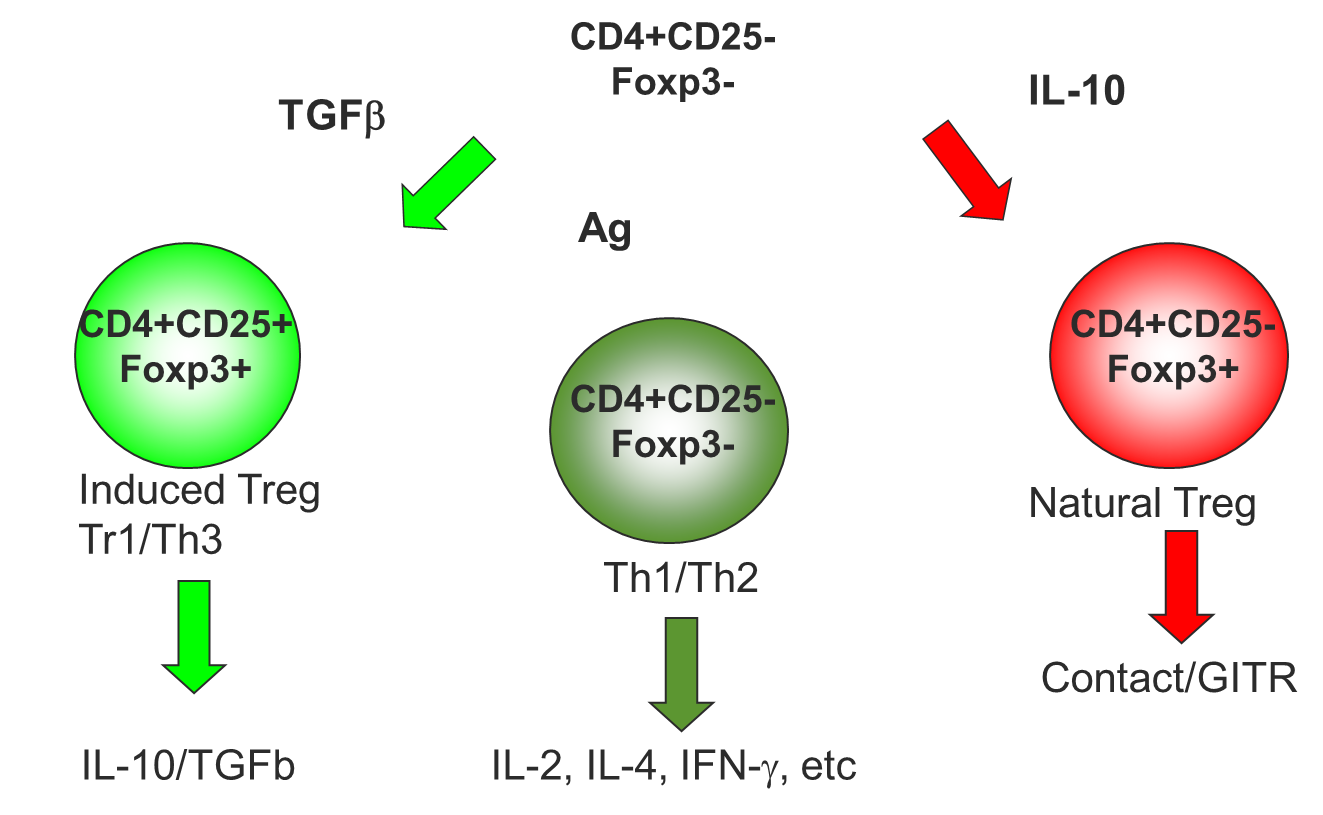
14: Stimulation of CD4+CD25-FoxP3- by different cytokines produces different cells (can ignore this slide)
15: Inhibitory cytokines secreted by Tregs include IL-10, TGF beta and IL-35.
IL-10:
inhibits IL-12 production (pro-inflammatory)
inhibits dendritic cell maturation
prevents CD28 phosphorylation (costimulatory signal involved in T cell activation)
TGF beta
promotes indoleamine 2,3-dioxygenase (iDO) - prevents T cell proliferation eg by producing TF Foxo1
inhibits IL-2 production
Finally, IL-35 promotes conversion of naive T cells into Treg cells
16: Tregs also work by inducing metabolic disruption via IL-2, cAMP and adenosine.
IL-2:
high IL-2R (CD25) expression
IL-2 = growth factor
Treg competes with proliferating cells via IL2
cAMP:
regulates functional activity of effector cells and APCs
cAMP transferred to target cell → PKA activation → cell proliferation and differentiation inhibited
Adenosine (via CD39/CD73 interaction):
Treg converts ATP to AMP
effector signalling pathways induced which impacts TFs controlling effector response
17: Tregs induce cytolysis by secreting granzymes and perforin which form pores in overactive effector cell membranes → apoptosis
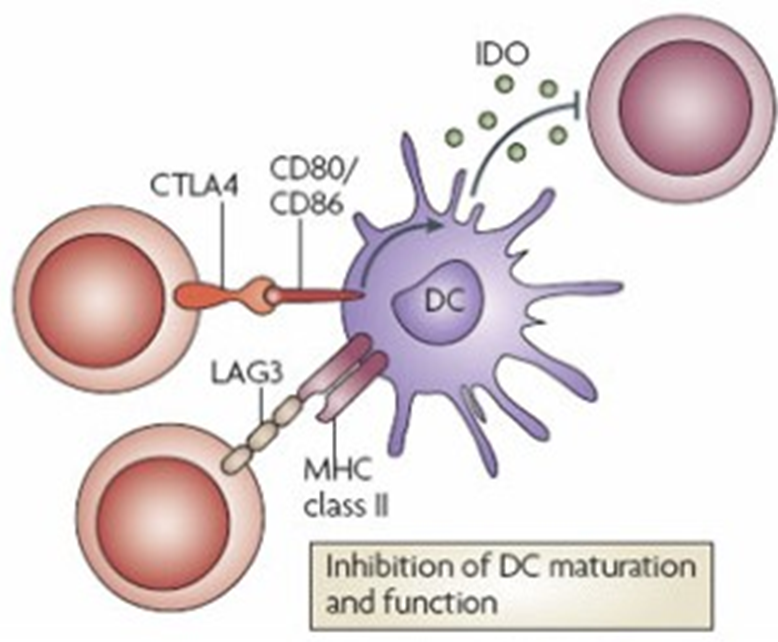
18: Tregs target dendritic cells via CTLA-4 and LAG3.
CTLA-4: (cytotoxic T lymphocyte associated protein 4)
protein receptor but also acts as immune checkpoint
has high affinity for B7 (CD80/CD86) - Treg competes with effector cells to bind costimulatory molecules
induces IDO (catalyses tryptophan → kynurenine) - tryptophan required for T cell activation
IDO activates caspase 8 directly to induce apoptosis
LAG3, or lymphocyte activation gene 3, is a homologue of the CD4 receptor and binds MHC II with a higher affinity
19: overview of all different aspects of Tregs
nTreg | iTreg Tr1 | iTreg Th3 | |
development | thymus | periphery (MALT?) | periphery (MALT) |
phenotype | CD4+CD25+CD127(low) | CD4+CD25- | CD4+CD25+ from CD25- precursors |
other associated markers | CTLA4+GITR+FoxP3+ | CD45$B(low)FoxP3- | CD25(low-variable)CD45RB(low)FoxP3+ |
suppression | contact-, granzyme-B dependent, produces TGF beta | IL-10 mediated | TGF beta mediated |
target cells | APCs and effector T cells | effector T cells | unknown |
CD28 involvement | thymic development and maintenance in periphery | unnecessary | unnecessary |
in vivo role | suppression of autoreactive T cells | mucosal immunity and inflammatory response | mucosal immunity and inflammatory response |
20: Too many Tregs → cancer, too few → autoimmune conditions. Need to have Treg balance
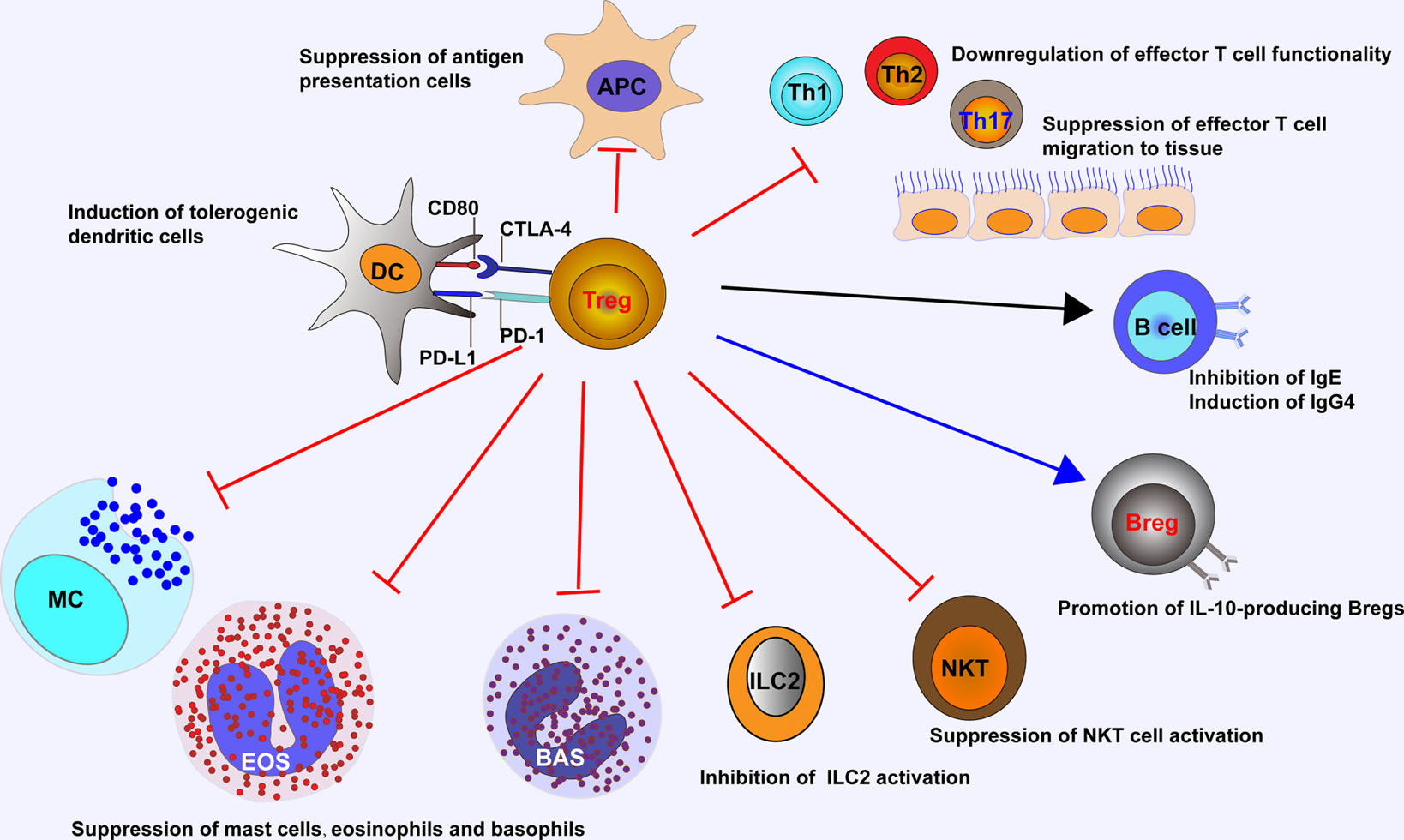
21: Tregs inhibit migration and function of Th cells, inhibit allergen specific IgE and induce IgG4, promote Bregs to produce IL10, suppress eosinophil, basophil and mast cell activation preventing degranulation which otherwise causes immune response
22: Embryos express paternal antigens, which are foreign. This makes babies semi-allografts yet they aren’t rejected due to Tregs.
The depletion of CD25+ Treg cells in mice’s adaptive transfer system causes gestational pregnancy. In humans there is an increase in circulating Tregs during early pregnancy, peaking in the second trimester and tapering off towards birth. Weak suppression of Th1 during pregnancy causes pregnancy to fail eg infertility/preeclampsia/miscarriage.
23: Tregs regulate immune response to both commensal and pathogenic bacteria
Colonisation with Clostridia (commensal) → increased Treg abundance. This is because short chain fatty acids produced in bacterial fermentation of dietary fibre promote Treg expansion. Clostridia also stimulate conventional T cells to produce IL2 which promotes colonic Treg proliferation. Commensal B. fragilis produces polysaccharide A which induces Treg induction.
The ability to module adaptive immune reactions is crucial for microbial persistence. During Mtb infection, expansion of Tregs parallel to effector T cell expansion is observed to balance out pro vs anti-inflammatory response. Foxp3+ Tregs have been found inside lung granulomas suggesting them may act locally at the infection site - recruited by Tb to downregulate anti-inflammatory response.
Extra reading REQUIRED:
why do we need Tregs? eg what responses do they suppress, what would happen if we didn’t have them/were deficient but also look into overpopulation of Tregs
thymic selection of Tregs
Treg differentiation at the periphery
Treg mechanisms
CD28 phosphorylation purpose
Tregs in health and disease