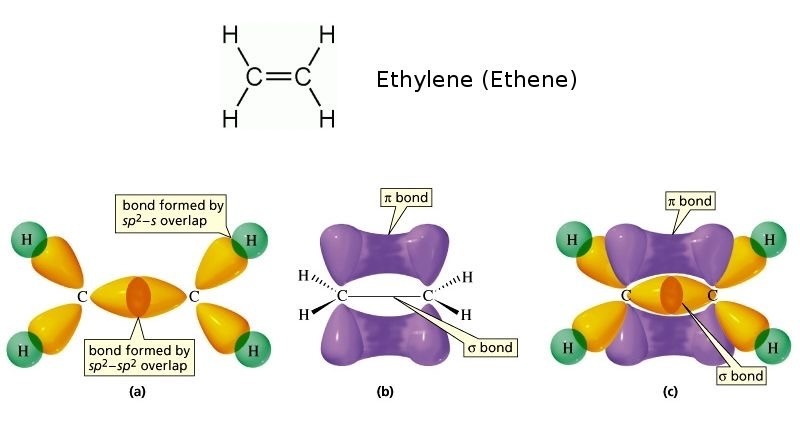
Bonding in ethene
Sigma (σ) Bond Formation
Between Carbon Atoms:
Each carbon atom in ethene forms a sigma bond with the other carbon atom.
This sigma bond is made by the direct overlap of the sp² orbitals from each carbon atom.
Between Carbon and Hydrogen Atoms:
Each carbon atom also forms sigma bonds with the hydrogen atoms.
These sigma bonds are formed by the overlap of sp² orbitals on the carbon atoms with the 1s orbitals of the hydrogen atoms.
Pi (π) Bond Formation
Between Carbon Atoms:
The pi bond in ethene is the second part of the double bond between the two carbon atoms.
It is formed by the sideways overlap of the p orbitals from each carbon atom.
Key Points:
The sigma bond is formed by the direct overlap of orbitals (sp² with sp², or sp² with 1s).
The pi bond is formed by sideways overlap of p orbitals, and it makes the double bond weaker than the sigma bond.