BIO 120 - Carbohydrates & Proteins
4 classes of large macromolecules: Carbs, proteins, nucleic acids. Lipids are NOT macros.
Macromolecules are made up of monomers, Polymers of Monomers
Monomers are small molecules of identical atom structures with 2 or more binding sites
Polymers are covalently bonded monomers into a train-like link
Class of macros. is determined by type of monomers present.
Dehydration: the act of removing H2O from a monomers to create a bond of polymers (synthesis polymers)
Hydrolysis: the act of intercepting H2O in a polymer to break it apart (break down polymers)

Carbohydrates
Monomer: monosaccharides
Bond: Glycosidic
POLAR due to -OH function groups
Composed of C,H & O CH2O
Act as fuel & building materials for cells
Monosaccharides: Simplest unit of sugar
EX: Glucose, galactose, fructose
Disaccharides: Two monosaccharides joined by dehydration synthesis
Ex: Sucrose
Polysaccharides: Polymer of many monosaccharides (monomer) joined by many dehydration reactions
Ex: Starch, Cellulose
Proteins
Mono: amino acids
Bond: peptide
NONPOLAR
Proteins are involved in almost everything a protein does
THE most important building block of life
Enzymes: speed up chemical reactions, without, death
Storage: egg whites hold amino acids necessary for embryo
Transport: hemoglobin/membrane proteins
Cellular Communications: insulin to blood sugar
Movement: Muscles, actin and myosin
Immune system: antibodies vs. viruses
Response from cell to chem stimuli
Structure: hair
Amino acid - amino group bonded to carboxyl group
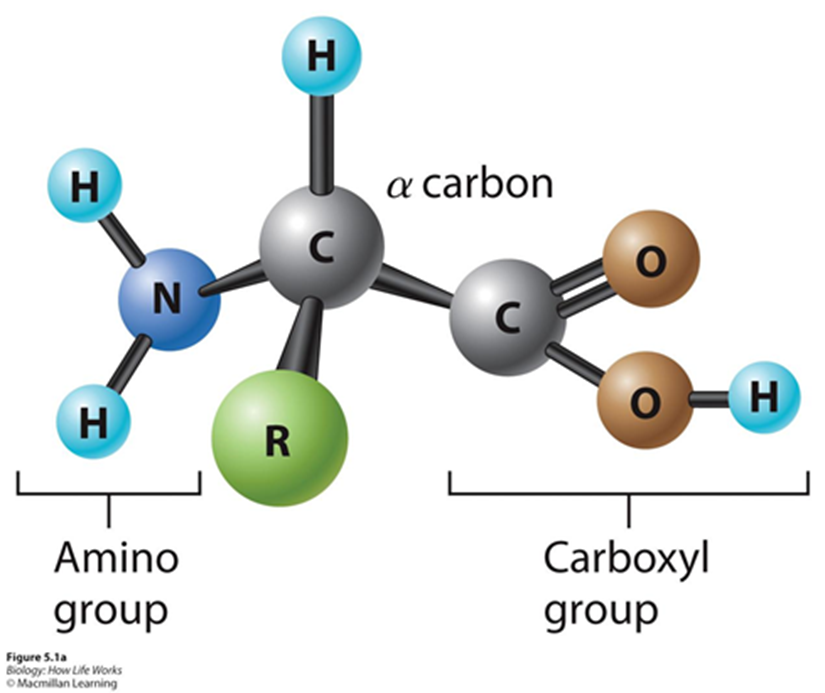
R group make one amino group different from another
20 diff amino acids
R groups can be polar, nonpolar, acidic, or basic
Peptides: a single chain of two or more amino acids linked by peptide bonds
Polypeptides: a chain of many amino acids (less than 50)
Forming Polypeptides: Dehydration link the carboxyl group on one amino acid yo the other amino acid via peptide (covalent) bonds
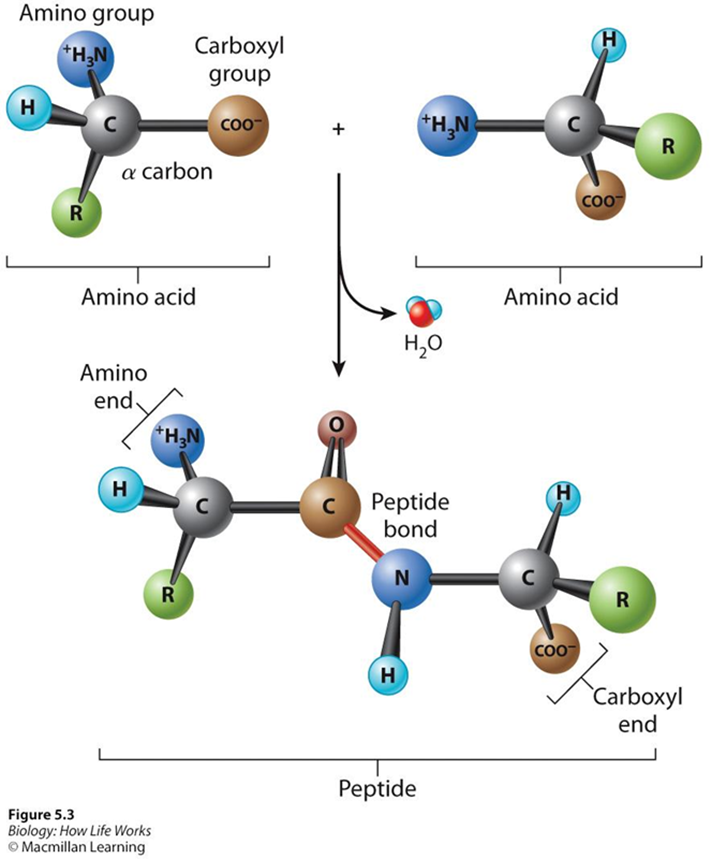
Proteins are made up of long chains of amino acids !!!
Functional protein is madeup of one or more polypeptides that have been twisted, folded, and coiled into a molecule with a unique shape
Function depends on its ability to recognize and bind to some other molecule
LARGE
Primary: refers to the amino acid single chain polypeptide that acts as a base for the other structures
Ex: DNA, the blueprint of a cell
Secondary: Alpha helix or Beta plated single polypeptides, these have different shapes due to differing primary structure, odd shape is from Hydrogen bonds, not peptide bonds
Polar R groups - H bonds
Charged R groups - Ionic bonds
Non-Polar R groups - hydrophobic
R groups - Disulfide bridge
Tertiary: also a polypeptide chain, but folds the secondary structure in globule-like molecules due to its different bond abilities like hydrophobic reactions, still held by Hydrogen bonds
Proteins vs Polypeptides: proteins are twisted into a unique shape, polypeptides are not (by themselves)
Quaternary: proteins with multiple polypeptide chains (hemoglobin)
Nucleic Acids
NONPOLAR
Mono: Nucleotide
Bond: Phosphodiester
DNA - RNA - Protein
Nucleic Acids: provide the blueprints/info used to produce/maintain the structure & function of an organism
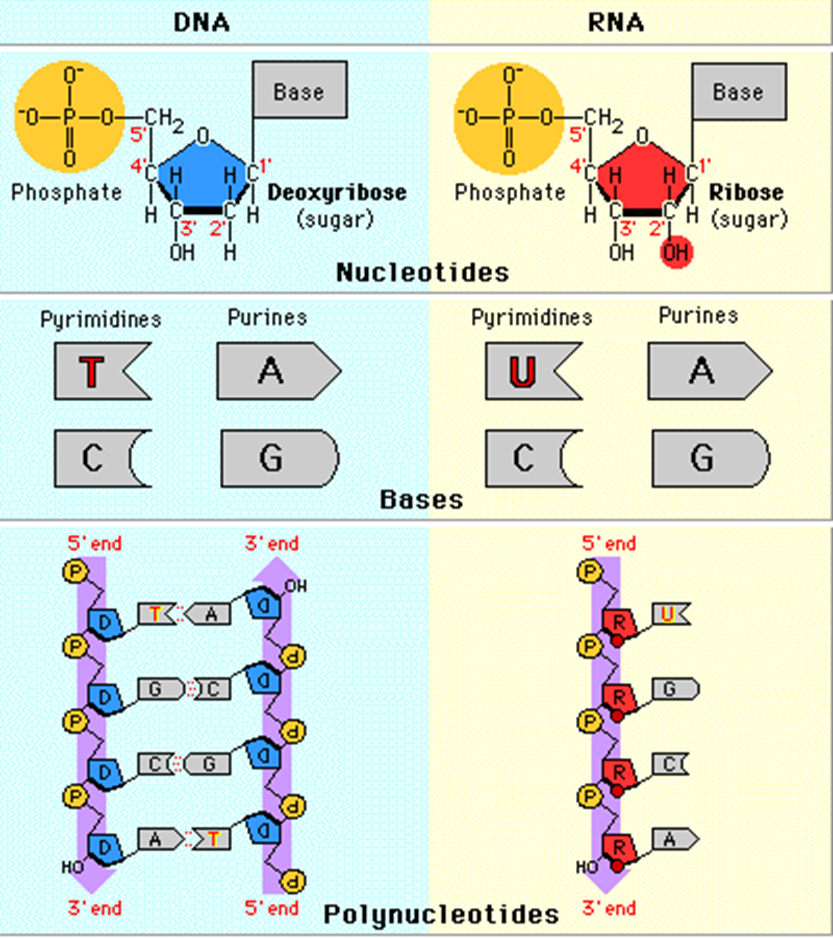
DNA: not involved in everything a cell does, just stores info that builds
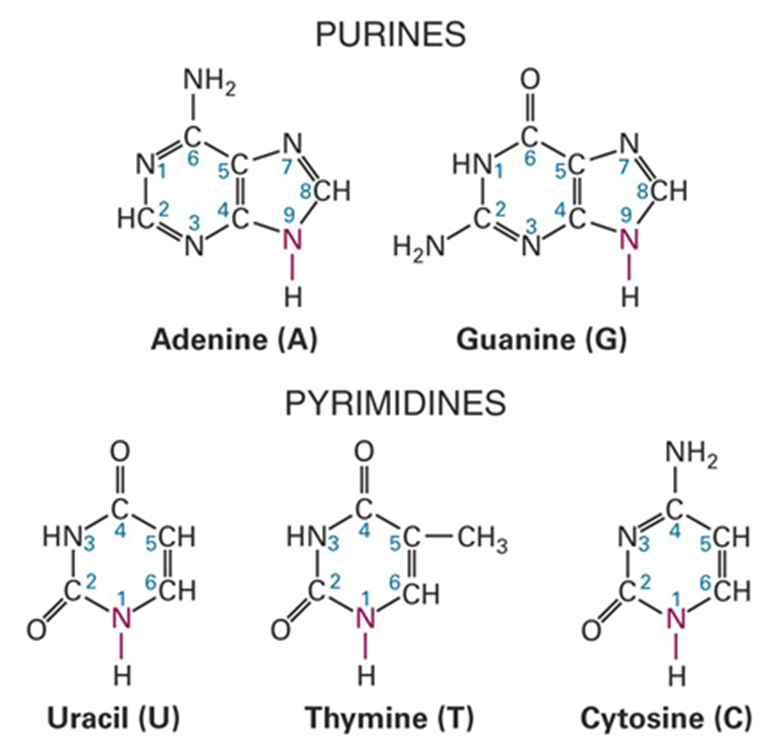
PURINES: Adenine, Guanine (either)
PYRAMIDINES: Uracil - RNA base, Thymine - DNA base, Cytosine - BOTH
Phosphodiester: Sugar, nucleic base, and Phosphate
Polynucleotides: many nucleotides that have been bonded by Phosphodiester
Lipid
Nonpolar, not a macromolecule. Not made of monomers.
Fat (Unsaturated & Saturated)
Phospholipids
Steroids: Cholesterol/Hormones
FATS
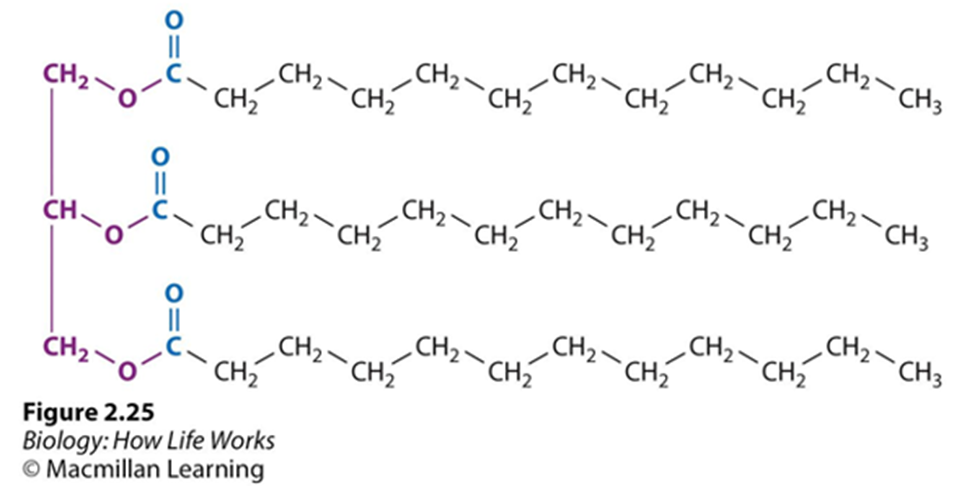
NO CHARGE
Glycerol head & 3 fatty acid tails
Bonded by ESTER LINKAGE
Dehydration reaction needed to bond
1 grams stores 2x a polysaccharide energy gram
Saturated: no double bonds, may stack, bad, animal fat
Unsaturated: one or more double bonds, can’t stack, good, plant/fish fat
PHOSPHOLIPIDS
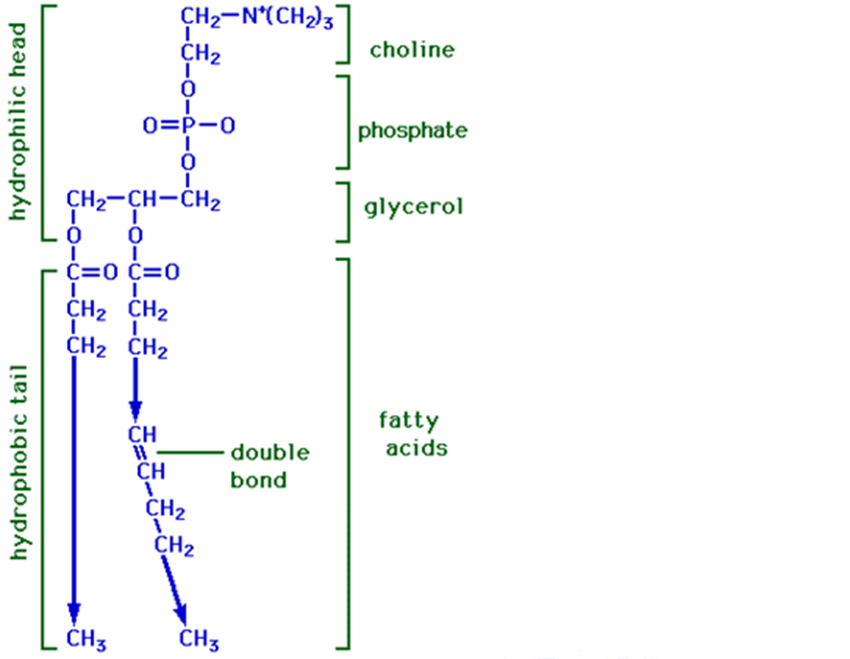
Prevent cell membrane from dissolving
Polar head, nonpolar tail
Phosphate group
Glycerol head & 2 fatty acid tails
STERIOIDS
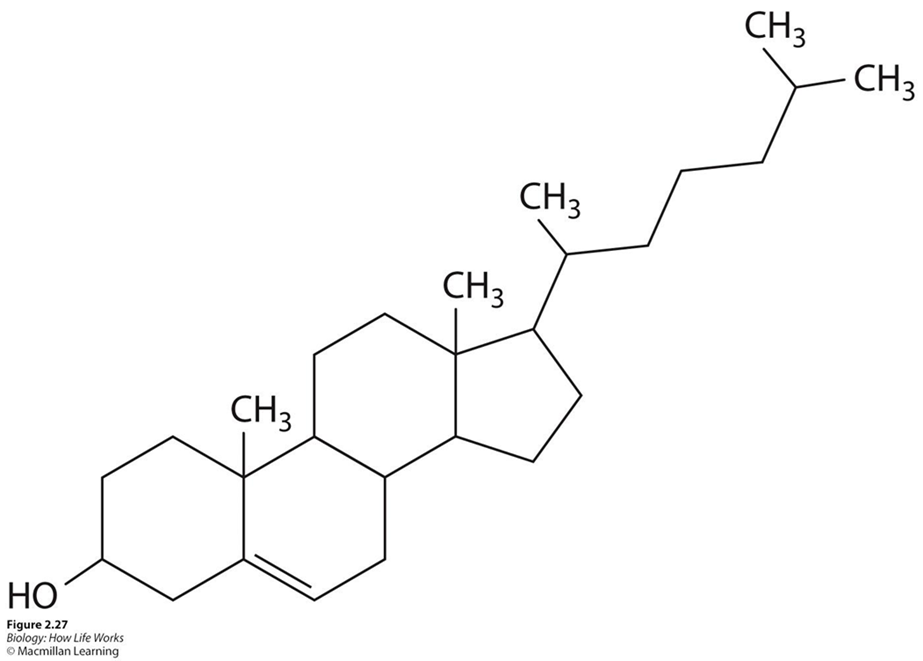
4 carbon rings distinguish it
Methyl group
Acts as a bio tag for gene expression
Diff chem placements for diff steroids
Hormones & Cholesterol