Lab Techniques- Separations
5.1 Separations
Extractions
Extraction allows for the chemist to separate one substance from a mixture of substances by adding a solvent in which the compound of interest is highly soluble. If the solution containing the compound of interest is shaken with a second solvent (completely immiscible with the first) and allowed to separate into two distinct phases, the bound of interest will distribute itself between the two phases based on its solubility in each of the individual solvents (liquid-liquid extraction).
distribution (partition) coefficient: the ratio of the substance’s solubilities in the two solvents
Solubility largely depends on two things: polarity of the solute and polarity of the solvent
LIKE DISSOLVES LIKE
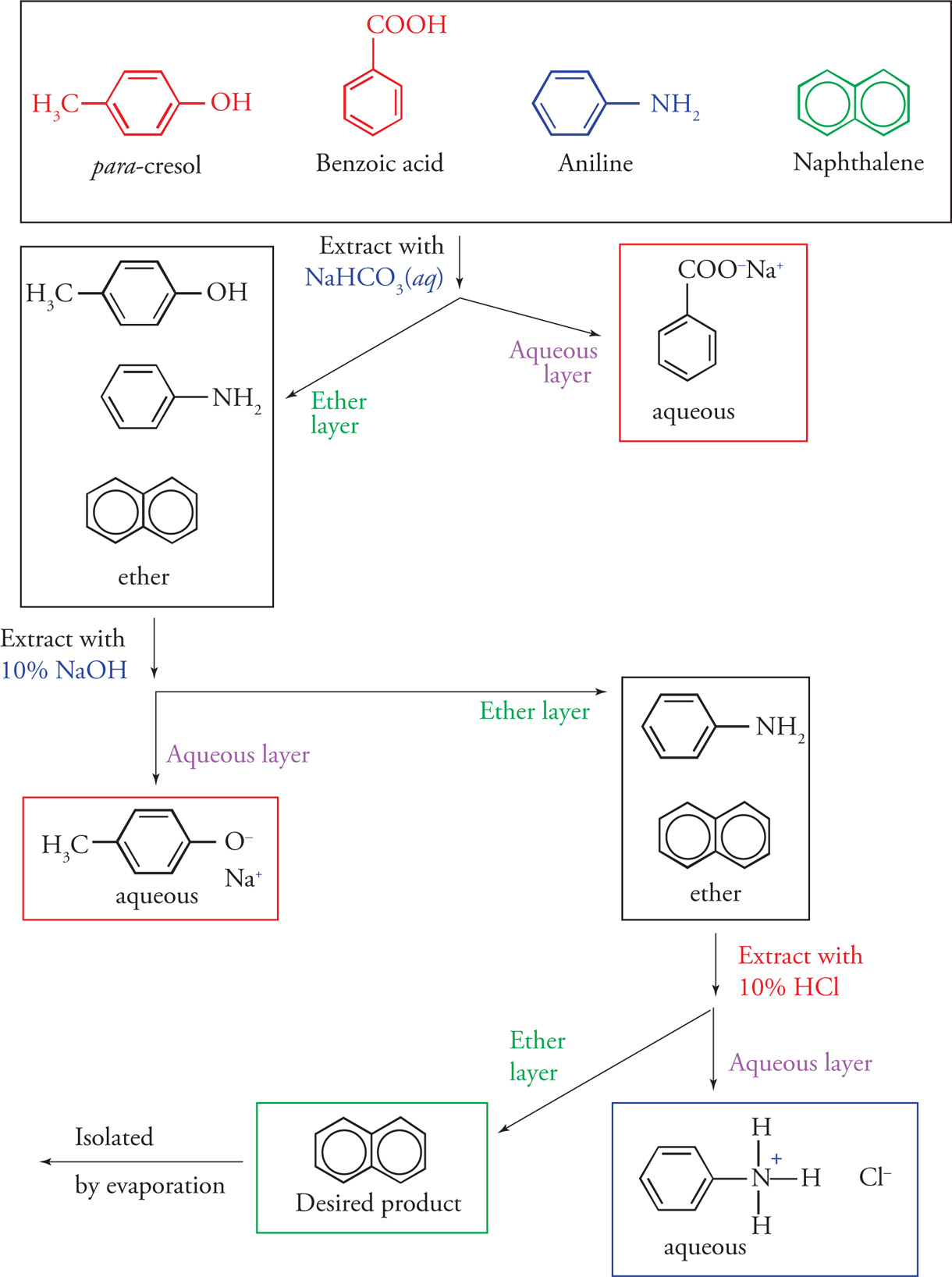
The simplest liquid-liquid extraction is accomplished when an organic compound is extracted with water. (can remove substances that are highly polar or charged including inorganic salts, strong acids and bases, and polar, low molecular weight compounds that are less than five carbons like alcohols, amines, and carboxylic acids)
Extraction of Organic Amines
Organic compounds that are basic (e.g amines) can be extracted from mixtures of organic compounds upon treatment with dilute acid (5-10% HCl).
This treatment will protonate the basic functional group, forming a positively charge ion. The resulting cationic salts of these basic compounds are usually freely soluble in aqueous solution and can be removed from the organic compounds that remain dissolve in the organic phase
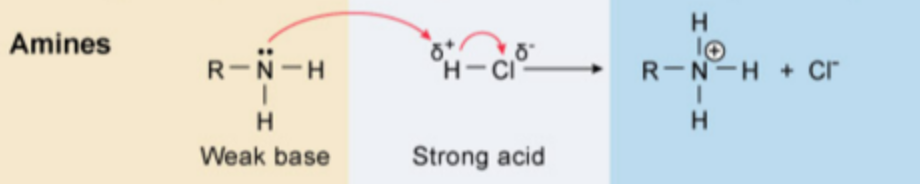
==Organic basic compounds (amines) + 5-10% HCl → Cl- + NH3R==
Extraction of Carboxylic Acids
Extraction with a dilute weak base (typically 5% sodium bicarbonate, could use NaOH) results in converting carboxylic acids into their corresponding anionic salts
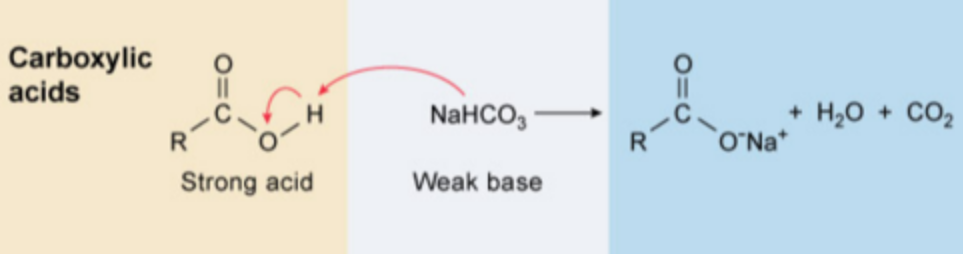
==Carboxylic acids + 5%NaHCO3 → R-COO-Na+ + H2O +CO2==
Extraction of Phenols
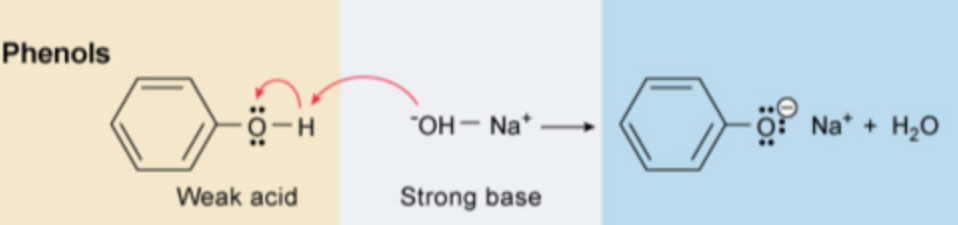
Amnionic salts are generally soluble in aqueous solution and can be removed from the organic compounds that remain dissolved in the organic phase. Diluted sodium hydroxide could also be used for this extraction.
When phenols are present in a mixture and need to be removed, dilute sodium hydroxide solution (about 10%) will succeed in converting phenols into their corresponding anionic salts. Anionic salts of phenols are generally soluble in aqueous phase and therefore can be removed from the organic phase
Separatory funnel is the apparatus that is used to carry out these extractions