Science
Biology
2.1 Plant and Animal Cells
What is a cell?
A cell has the following characteristics..
A lifespan
Can sense and respond to changes in their environment
Grow, reproduce, and repair themselves
Require energy
Produce waste
The Cell Theory
All living things are composed of one or more cells
The cell is the most basic and functional unit of life
All cels come from pre-existing cells
Cell Structure
Organelles: a cell structure that has a specific function for the cell
Cytoplasm: jelly-like liquid that contains many important substances and hold the organelles working the cells
Organelles and their functions (2.1)
Cell Membrane: separates the inside of the cell from the outside environment, controls the flow of materials in and out of the cell (semi permeable) and is flexible and double layered.
Nucleus: surrounded by a nucleus membrane (thick, double membrane, that separates the nuclear contents from the cytoplasm), the “control centre” of the cell, directs the cell's activities, and contains genetic information (DNA).
Chromosomes: made up of chromatin (DNA), which coils up into chromosomes, thread-like structures in the nucleus, and contains genetic information (DNA).
Mitochondria: the "power plant”, releases energy from glucose through a process called cellular respiration, cells requiring more energy have more mitochondria.
Endoplasmic Reticulum: canals and tubes connected to the nucleus to help transport materials within the cell, continuously from cell membrane to nuclear membrane, important in the production and release of hormones in the brain, has two types: smooth and rough ER.
Ribosomes: small organelles that make protein, required for all of the cell activities, they can be attached (to RER) or free floating in the cytoplasm.
Golgi Apparatus: they sort and package proteins and other materials for transport out of the cell, creating sac-like structures called vesicles that will travel outside the cell, cells that secrete a lot of mucus such as the lining of intestines have many golgi bodies.
Vacuole: main functions include storage and support, the membranous sac containing water, food, and wastes, animal cells that have many small vacuoles, and plant cells usually have one large vacuole. When filled with water, it keeps the cells plump, keeping the plant stems and leaves firm (called keeping turgor pressure).
Lysosome: enzymes inside break down wastes, invading bacteria, and damaged cell organelles.
Cell wall: an additional covering for plant cells outside of the cell membrane, provides rigid shape, protection, and structure for the cell.
Centrioles: helps us with cell division.
Chloroplasts: contains chlorophyll, the green pigment found in plants, the site of photosynthesis - chlorophyll - traps light energy.
2.3 The Importance of Cell Division
Cell Division for Reproduction
Sexual reproduction → producing offspring by the fusion of two gamete cells.
Gamete cells have half the DNA
Cell Division for Growth
If the cell needs more chemicals, the area of higher concentration will move the chemicals to the area of lower concentration. This is called diffusion.
Osmosis
This occurs when a fluid (water) travels through the membrane from the solute of higher concentration to the solute of lower concentration
Cell Division for repair
Body sheds millions of dead skin cells, red blood cells are replaced every 120 days
2.5 The Cell Cycle
Mitosis:
Interphase
90% of cell life is spent here.
DNA is duplicated
Stages of interphase
Growth 1
Cells grows and preforms like regular
Synthesis
Cell makes an entire copy of its DNA
Growth 2
Cell approaches maximum size
Produces the organelles for mitosis
Prophase
Chromosomes coil and thicken
Nuclear membrane disappears
Centrioles start to move to opposite poles
Spindle fibres start to form
Metaphase
Centrioles are located at opposite poles
Spindle fibres attach at the centromere and align the chromosomes at the centre of the cell
Anaphase
Chromosomes split up
Sister chromatids go towards opposite poles
Telophase
Clear membrane forms around the nucleus
Chromosomes stretch out (become thin) and form chromatids
Cytokines
Cytoplasm and other organelles are distributed
Cell membrane forms
Plants
Cell well and chloroplasts and one large vacuole
Animals
Lysosomes and centrioles and many small vacuoles
2.7 Cell division gone wrong
Malfunctions in the DNA
Cell keeps dividing despite the messages from the nucleus
Benign tumor
Not harmful
will not metastasize
rarely lethal
Limited growth
Non-cancer
Malignant Tumor
Invasive
Uncontrolled growth
May metastasize
Often lethal
Cancerous
Characteristics of cancer cells
Cancer cells use up
Oxygen and nutrients that other healthy cells need
Can lead to crowding
Cancer cells can divide Isolated from others
Inefficient
Causes of cancer
When DNA isn’t replicated properly
Genetic mutations
Carcinogens (smoking and UV radiation, etc.)
Screening examples
Self examination
Pap test
Blood test
Colonoscopy
Mammograms
ABCD’s of Moles
Asymmetrical border colour diameter
Benign tumours are symmetrical, even uniform and less than six
Malignant tumours are asymmetrical, uneven, not uniform colour and the diameter is greater than 6
Cancer treatments
Surgery
Chemotherapy
Radiation
Specialized cell is a cell that can perform a specific function such as white blood cells which can destroy infected cells and fight infection and bacteria
Stem cells
All cells in our bodies originally came from a small group of stem cells
Scientist believe if they study them, they will develop a better understanding In cell specialization
They believe the stem cells may be used to treat injuries and diseases by regenerating organ
Stem cells are capable of becoming any cell in the human body
Embryonic cells are out of the embryo and they are able to differentiate into other cell types. Tissue stem cells are involved in the replacement of damaged tissues. Stem cells are found in the skin blood and neural tissue.
3.1 Hierarchy of Animal structure
Cell (simple)
Tissue
Organ
Organ Systems
Organisms (complex)
Difference between cell, tissues and organs
Cells are the most basic unit of structure and function of living things
Tissues are a group of cells with similar structure and function
Organs are a group of two or more types of tissue that performs a specific task
4 types of tissue
Epithelial tissue (skin, lining of digestive system)
Nerve tissue (Brain, nerves)
Connective tissue (bone,blood)
Muscle Tissue (heart,hamstrings,biceps, digestive system)
Digestive system
Function
Breakdown of food
The absorption of nutrients
Elimination of waste
Digestive Tract
In humans: mouth → esophagus → stomach→ small intestine →liver →gall bladder → pancreas→ large intestines→ anus
Diseases/illnesses/disorders of the digestive tract
Stomach ulcers, appendicitis,heart burn
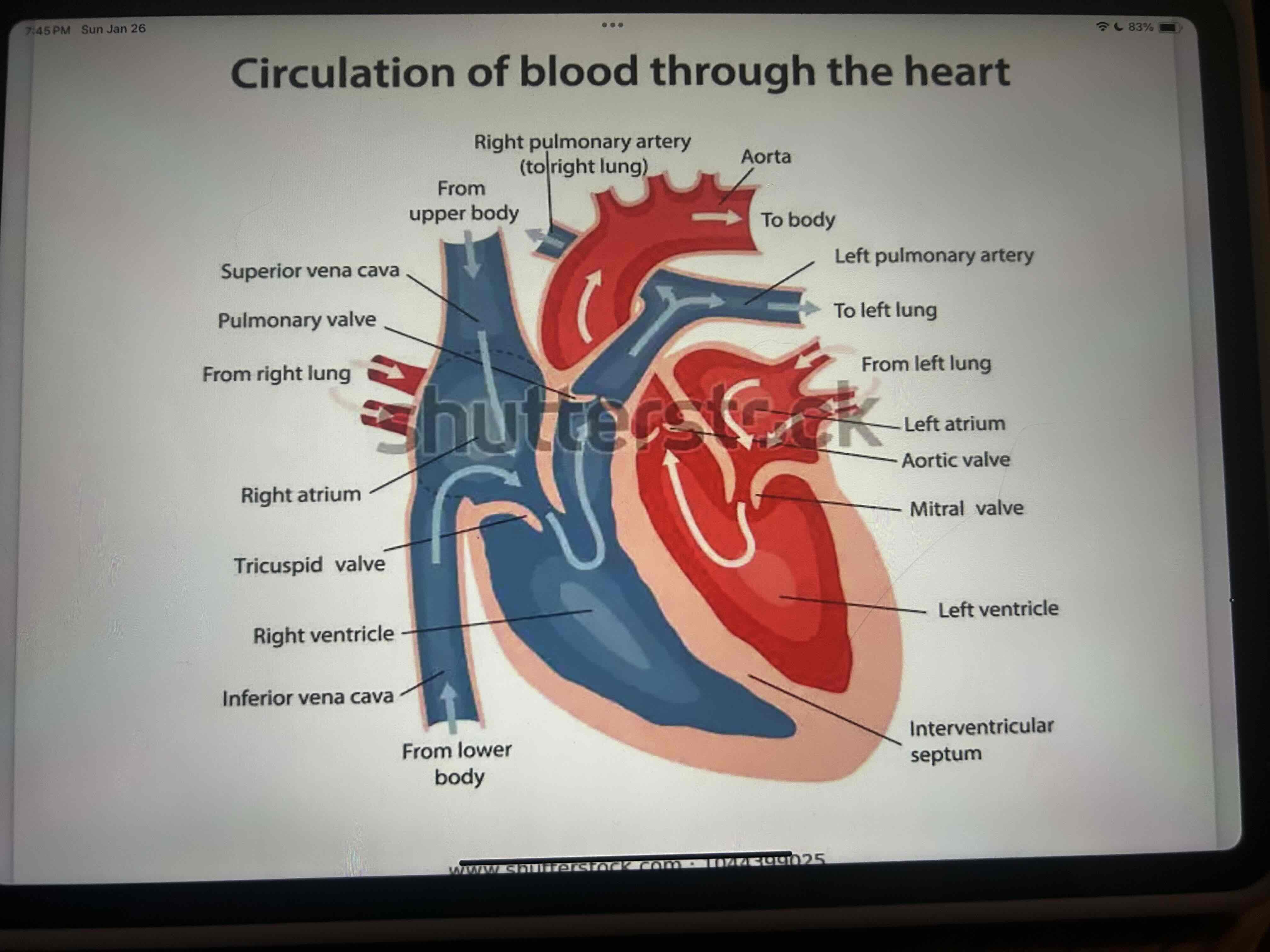
Circulatory System
Function
Transport substances around the body, such as nutrients, oxygen hormones and waste
Regulation of body temperature
Parts of the circulatory system
Blood (transports various materials), heart ( a pump that pushes blood through blood vessels) and blood vessels (network of tubes where blood moves through).
flow of O2 and CO2 in the system
right side of the heart carries deoxygenated blood to the lungs, blood picks up oxygen from the lungs and moves it to the left side of the heart, left side of the heart carries oxygenated blood to body tissues ( Arteries and capillaries), tissues use oxygen and return deoxygenated blood to the right side of the heart (capillaries and veins)
Diseases
Stroke, heart attack, high blood pressure
Respiratory system
Function
Providing oxygen required by the body and removal of carbon dioxide produced by the body as it uses energy
Parts of the system
Nasal cavity, pharynx, nose, mouth, bronchus, trachea, lungs, ribs, bronchiole, alveolus, diaphragm.
Flow of O2 and CO2 in the system
O2 comes in as co2 comes out. Oxygen diffuses into the blood and is carried to the tissues, oxygen then diffuses from the blood into the tissues at the same time carbon dioxide from tissues are picked up by the blood and transported to the lungs where it diffuses out into the air in the alveoli and is expelled into the external environment.
Breathing:
Inhale: Chest expands, diaphrasm contracts.
Exhale: chest contracts, diaphragm relaxes
Diseases: cancer & tuberculosis
Musculoskeletal System
Function
Supports the body, protects delicate organs, makes movement possible, store minerals and makes blood cells.
Parts: bones, ligaments, cartilage
Tendons vs ligaments
Ligaments are tough, elastic tissue that holds the bones together at the joints and skeletal muscle is connected to the bones by tendons
Muscles can only pull they cannot push and so skeletal muscles always work in opposing pairs.
Diseases: osteoporosis, tears in ligaments
Nervous System
Function
Senes the environment and coordinates appropriate responses
Parts
Made of the peripheral nervous system (PNS), peripheral nerves, central nervous system (CNS) brain and spinal cord
Sensory vs motor neurons
Sensory neurons carry signals from sensory receptors to the central nervous system, while motor neurons transmit signals from the central nervous system to muscles for action.
Diseases: multiple sclerosis, brain injuries
Interaction of systems
Nervous and Muscular Systems: The nervous system sends signals to muscles to control movement and reflexes.
Circulatory and Respiratory Systems: The circulatory system transports oxygen from the lungs (respiratory system) to cells and removes carbon dioxide.
Digestive and Circulatory Systems: The digestive system breaks down food, and nutrients are absorbed into the bloodstream for distribution by the circulatory system.
Physics
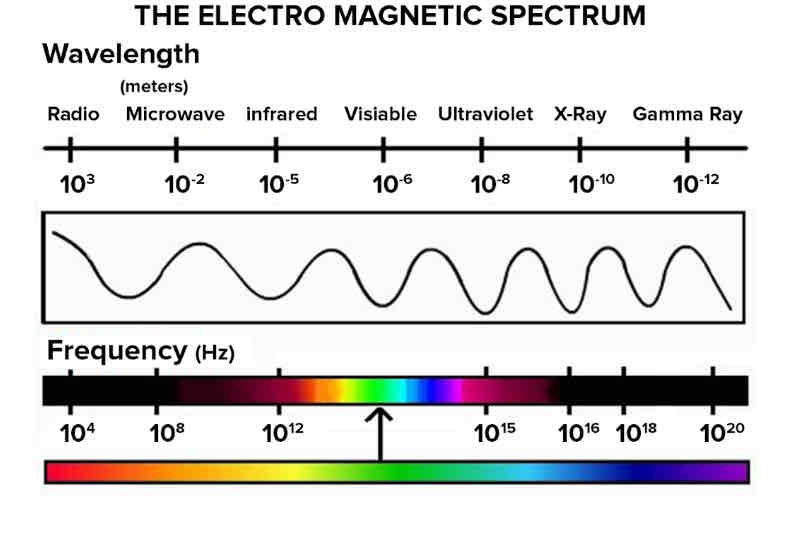
Light is an electromagnetic wave it travels very fast (3.0. X 10 8 m/s) 300 000 000
The electromagnetic spectrum is organized in order from the longest wavelength/lowest amount of energy to the shortest wavelength and highest amount of energy.
 The sources of light are…
The sources of light are…
Bioluminescence: Emission of light by a living organism (fireflies)
Chemiluminescence: The production of light from chemicals (glow sticks)
Luminescence: Emission of light from materials that are relatively cool.
Fluorescence: production of visible light by an object that has absorbed ultraviolet light. (Highlighters & fluorescent light bulbs)
Phosphorescence: When a special material phosphors absorb UV light and emit that light over a period of time. (Glow in the dark items)
Incandescence: The production of light energy when an object is heated to a high temperature. (Regular light bulbs and flame)
Triboluminescence: When two crystals are rubbed or crushed together and emit light. (two sugar cubes rubbed together & two quarts stones rubbed together)
Electric discharge: an electric current passes through gas, causing gas to flow. The electricity causes the gas to glow. (Lighting & neon lights)
Light emitting Diode (LED) : An electronic device that allows an electric currents to flow in only one direction using semiconductors (silicon), are energy, efficient, and produce less heat (LED Christmas lights)
Transparent materials , lets lights pass through it easily
Translucent materials let some light pass through it
Opaque material does not let any light pass through it
Laws of reflection
The angle of incidence must equal to the angle of reflection
The incident ray, reflected ray & the normal all lie in the same plane.
Images are described using SALT
1.Size (is the image bigger or smaller than the object)
2. Attitude (is the image upright or inverted)
3. Location ( is the image located in front of or behind the mirror (real) or behind the mirror (virtual)?
4. Type: Is the image virtual or real?
Refraction:
Refraction is the bending of light as it travels from medium to another medium of different density.
Light changes its speed when it travels through different media.
There are two rules of refraction…
The incident ray, refracted ray and the normal all lie in the same plane.
If a light ray travels from a fast -> slow medium it bends toward the normal, but if it travels from a slow -> fast medium it bends away from the normal
Partial refraction occurs when light passes through a boundary between two different materials and bends as it enters the new material
The index of refraction is a quantitative physical property that can distinguish the different types of media,
The index of refraction is the speed of light in a vacuum\the speed of light in a medium.
Total internal reflection:
When light travels from a slow to fast medium, light bends away from the normal. It speeds up at the boundary between the media it is travelling through.
There are two conditions for total internal reflection..
Light has to travel from a slow to fast medium
The incident ray has to be greater than the critical angle so no refraction occurs.
Cameras and Binoculars
Cameras and binoculars use glass prisms that have a high index of refraction and place
them in a particular angle within a device in order to reflect 100 % of the light internal.
Critical Angle
When the refracted ray is = to 90 degrees, the incident ray this occurs at is referred to as the critical angle
If the angle of incidence increases beyond the critical angle, the refracted ray will be reflected back into the media and the refracted ray will then disappear and only the reflected ray will be visible in the medium
Chemistry
Physical properties
Color,
luster,
ductility
texture,
boiling condensation point
melting, freezing point
solubility,
conductivity
odor,
crystal formation
states of matter viscosity
malleability
clarity
Chemical Properties
Combustibility
reaction with acid
Evidence of a chemical change
changing colour,
changing odour
bubbles without heat,
new white precipitate, solid forms,
change in temperature and light
Describing Chemical Reactions
Reactants —> Products
Release energy: Exothermic = Product
Absorbed Energy: Endothermic= reactant
Types of chemical reactions
Synthesis: A + B = AB
Decomposition AB —> A + B
Single Displacement: AB + C —> AC + B
Double Displacement : AB + CD —> AD + CB
Combustion look for the hydrocarbon.
Acids and Bases
H Ions = hydrogen ions (acids)
OH Ions = hydroxide ions (bases)
An example of an acid is HCI which is stomach acid and it is also in a swimming pool, H2, S04, which is in car, batteries and detergent
An example of a base is NAOH which is in soap, CAOH which controls the acidity of soil
ionic compounds (metal + non metal)
Properties of ionic compounds
solid at room temperature
Hard and brittle
High meltingPoint
Electrons transfer
Molecular and covalent bonding
nonmetal plus nonmetal and they share electrons
properties
Solid liquid gas at room temp
Low melting and boiling points
Nonconductive
The law of conservation mass
The mass of the reactants is equal to the mass of the products
Properties of an acid
Sour taste
Soluble in water
Electrolytes
Reacts with Metals
React with carbonate
Turns blue litmus red
Turn red litmus red
Phenophallen —> clear
Properties of Bases
Bitter
Soluble in H2O
Electrolytes
Slippery
Turns blue litmus to blue
Turns red litmus blue
phenophalien — > pink
Naming acids and bases
Binary or Oxy (oxygen)
Oxy; drop hydrogen, “ate” —> “ic”, + add acid
Binary: hydrogen becomes hydro, non metal: “ic”
Bases; name like ionic plus add hydroxide
Complete combustion
Excessive O2
Products are CO2 in H2O
Blue flame
Incomplete combustion
O2 was limited
Products are CO2, CO, CI, H20
Yellow sooty colour