Mole Concept
📐 Precision & Dimensional Analysis Notes
🎯 Precision vs Accuracy
Key Definitions
Precision - Degree of "exactness" in measurement
Accuracy - How close data is to the "true" or "accepted" value
Understanding the Difference
Precision relates to consistency of measurements
Accuracy relates to correctness compared to known values
When running multiple trials, accuracy refers to how close the average is to the true value
Visual Examples 🎯
The dartboard analogy helps illustrate measurement errors:
Bull's-eye = "true" value
Darts = experimental measurements
Scenarios:
(a) Both accurate and precise: Darts clustered near center
(b) Precise but inaccurate: Darts clustered away from center
(c) Imprecise but accurate average: Darts scattered but average position is near center
🔍 Measurement Precision in Practice
Reading Instruments
When measuring with laboratory instruments:
Always read to the last place you can see
Make a "good faith estimate" on the next decimal place
Use metric units for precision
Pencil Measurement Example 📏
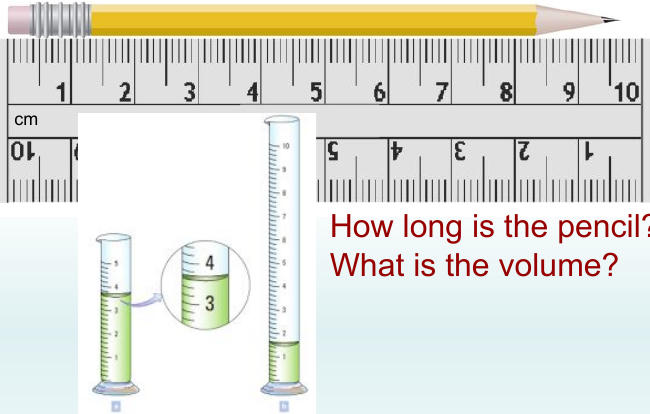
For a pencil measuring 9 cm with additional precision:
Visible: past 0.6 cm mark, not quite on 0.7 cm mark
Acceptable answers: 9.66 cm - 9.69 cm
Most appropriate: 9.69 cm (includes good faith estimate)
Volume Measurement Examples 🧪

Key Principle
Graduated cylinders provide more decimal places than beakers, making them more precise instruments.
Meniscus Reading
The meniscus is the curved surface of liquid in glassware
Always read at the bottom of the curve
Example: 7.3 mL reading when curve appears closer to 7.4 mL
Why 7.3 mL and not 7.38 mL?
Instrument increment is 0.2 mL
Between lines, only odd numbers can be reported
Answer: C (Both A and B are correct)
Instrument Selection
Instrument | Precision Level | Use For |
|---|---|---|
Beaker | ±5% estimation | Approximate volumes |
Graduated Cylinder | 0.1-0.2 mL increments | Precise measurements |
Digital Instruments | Computer-estimated last digit | Exact readings |
Digital Readouts
Important: Never estimate beyond what's displayed on digital instruments
The computer already provides the "good faith estimate"
Write exactly what appears on screen
Example: Digital scale showing 5.67 g → record as 5.67 g
🔄 Dimensional Analysis
Fundamental Concepts
Dimensional analysis = analyzing and converting units while maintaining the same quantity
Basic Principle
When converting units, we multiply by 1 in the form of conversion ratios:
(therefore )
(also valid)
Why Division Works
Example: Convert 144 inches to feet
The units factor out, leaving only the desired unit.
Multi-Step Conversions
Example: How many seconds in 1.0 years?
Continue through minutes to seconds
Chemical Application
Example: Given 3 moles H₂ produces 2 moles NH₃, how much NH₃ from 4.65 moles H₂?
Key Takeaways for Dimensional Analysis
Start with given quantity
Choose conversion ratios that eliminate unwanted units
Multiply numerators and divide denominators
Units must cancel appropriately
Never changes the actual quantity - only the units expressing it
📚 Mole Math Essentials (Plain‑Text Version)
1. The Mole Concept – A Chemical Counting Unit
Definition: The mole (mol) is the SI unit for amount of substance. It works like a “dozen” but on an astronomical scale.
Avogadro’s Number: 6.022 × 10²³ particles (≈ 602,000,000,000,000,000,000,000).
Scale Illustration: If you poured 6.022 × 10²³ water drops, Earth’s oceans could be filled about 45 times.
Key Relationships
1 mol = 6.022 × 10²³ atoms, molecules, ions, or other particles.
This number is the same for any substance.
2. Molar Mass Calculations
Molar mass (gram‑formula mass) = mass in grams of 1 mol of a substance.
How to find it from the periodic table:
Locate each element in the compound.
Note the atomic mass (the bottom number in each element’s box).
Multiply each atomic mass by its subscript (the number of atoms of that element).
Add all the values together.
Example – Water (H₂O)
H: atomic mass ≈ 1.008 g mol⁻¹, 2 atoms → 2 × 1.008 = 2.016 g mol⁻¹
O: atomic mass ≈ 16.00 g mol⁻¹, 1 atom → 1 × 16.00 = 16.00 g mol⁻¹
Total molar mass: 2.016 + 16.00 ≈ 18.015 g mol⁻¹
3. Mole‑Gram‑Particle‑Liter Conversions (The “Bridge” Method)
From | To | Conversion Factor |
|---|---|---|
moles | particles | 1 mol = 6.022 × 10²³ particles |
moles | grams | use molar mass (g / mol) |
moles | liters (gas at STP) | 1 mol = 22.4 L |
Dimensional‑Analysis Example
Problem: How many calories are in 5.2 Mcal?
Solution (plain text): 5.2 Mcal × 1 × 10⁶ cal / Mcal = 5.2 × 10⁶ cal.
4. Stoichiometry with Moles
Reading a balanced equation as mole ratios:
2 H₂ + 1 O₂ → 2 H₂Oreads as “2 moles of H₂ react with 1 mole of O₂ to give 2 moles of H₂O.”Mole ratios come directly from the coefficients and are used to convert between reactants and products.
Example – Combustion of Propane
Question: How many moles of CO₂ are produced from 98.0 g of C₃H₈?
Steps (plain text):
Molar mass of C₃H₈:
C: 12.01 g mol⁻¹ × 3 = 36.03 g mol⁻¹
H: 1.008 g mol⁻¹ × 8 = 8.064 g mol⁻¹
Total = 44.094 g mol⁻¹
Convert grams to moles:
98.0 g / 44.094 g mol⁻¹ ≈ 2.22 mol C₃H₈Use the mole ratio from the balanced equation (C₃H₈ + 5 O₂ → 3 CO₂ + 4 H₂O):
2.22 mol C₃H₈ × (3 mol CO₂ / 1 mol C₃H₈) ≈ 6.66 mol CO₂
5. Quick Reference Table (Conversion Factors)
Quantity | Symbol | Value |
|---|---|---|
Avogadro’s number | Nₐ | 6.022 × 10²³ particles / mol |
Molar volume (STP) | Vₘ | 22.4 L / mol |
Gram‑to‑mole factor | — | Molar mass (g / mol) |
📚 Dimensional Analysis & Stoichiometry
🧪 Dimensional Analysis & Mole Ratios
Understanding Dimensional Analysis
Dimensional analysis converts between units by multiplying fractions where units cancel out. When converting 144 inches to feet:
The key principle: units factor out when a unit is divided by itself, leaving only the desired unit
Setup:
Result:
Why divide by 12: The conversion factor creates a fraction equal to 1
Mole Ratios: The Foundation
Three essential mole relationships form the backbone of stoichiometric calculations:
Relationship | Equation | Source |
|---|---|---|
Particles to moles | Avogadro's number | |
Mass to moles | Periodic table | |
Reaction stoichiometry | Balanced chemical equation |
Worked Example: Decomposition of Potassium Chlorate
Given the reaction:
Problem: Calculate moles of produced from 465 g
Solution pathway:
Convert mass to moles using molar mass
Use mole ratio from balanced equation
Calculate final moles of product
Laboratory Application: Aluminum Foil Experiment
Objective: Determine length of aluminum foil containing exactly 1 mole of aluminum
Key insights:
Expect calculations yielding small numbers in intermediate steps
Final answer will exceed 200 cm
Success requires persistent calculation through multiple conversion steps
Approach mirrors the potassium chlorate example: convert between mass, moles, and physical dimensions using dimensional analysis