Light, Energy, and Electrons Chapter Test
Excited/Ground State
- Ground State
- Lowest energy state
- This means that e- are found in shells closer to the nucleus
- n =1
- Excited State
- Higher potential energy of an atom
- n = 2 or higher
- A form of heat, light, electrical, or mechanical energy is needed to go from the ground to an excited state
- As electrons increase in energy, they move away from the nucleus and into outer shells
Absorption/Emission
- Absorption (take in)
- Energy moves electrons from a ground state to a higher energy state
- Heat, light, electrical, chemical mechanical energy
- Emission (give off)
- Lets electrons fall back down to a lower energy state
- Usually light
- Energy must be absorbed for an electron to move to a higher state (one with a higher n value)
- Energy is emitted when the electron moves to an orbit of lower energy (one with a lower n value)
- The overall change in energy associated with "orbit jumping" is the difference in energy levels between the ending (final) and initial orbits
Wavelength/Frequency/Energy (ROY G BIV) (Both equations)
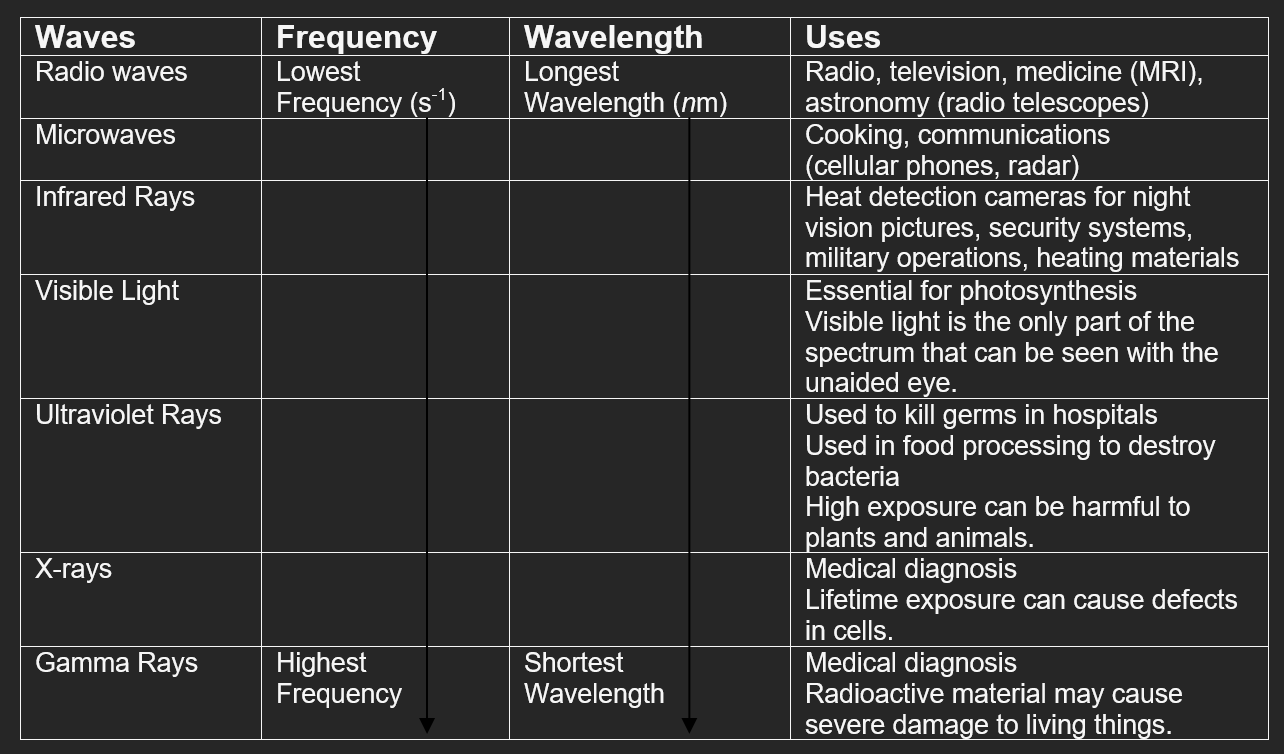
- The wavelength (λ) of light is defined as the distance between the crests or troughs of a wave motion.
- Wavelengths found in the electromagnetic spectrum (range of light) can be measured in units as large as 103 meters (radio waves) to 10-11 meters (gamma waves).
- For the wavelengths of visible light (the light we see in color) the most common units used are nanometers (10-9 meters) and Angstroms (10-10 meters).
- Frequency (ν) is the number of occurrences of a repeating event per unit time.
- In the case of light, frequency refers to the number of times a wavelength is repeated per second. The unit used most often to describe frequency is Hz which means "per second" or /s.
- The relationship between wavelength and frequency is related through the speed of light.
- c = λν
- c = 3.00 x 10^8 m/s
- c is the speed of light
- v is frequency
- λ is wavelength
- E=hv
- h = 6.63x10^-34 J.s
- E stands for energy (in Joules)
- v stands for frequency [in reciprocal seconds – written s^-1 or Hertz (Hz)- 1Hz = 1 s^-1)
- h is Planck’s constant.
- If the frequency is known, it can easily be converted to wavelength using the speed of light and vice versa.
- The wavelengths and frequencies of the light emitted by an atom (its emission spectrum) is determined by its electronic structure.
- As each electron moves from a higher energy level (orbit) to a lower one, a different color is emitted.
- Each shade of color has a unique wavelength based on the unique distance and energy.
- As a wavelength increases in size, its frequency and energy (E) decrease.
- As the frequency increases, the wavelength gets shorter.
- As the frequency decreases, the wavelength gets longer.
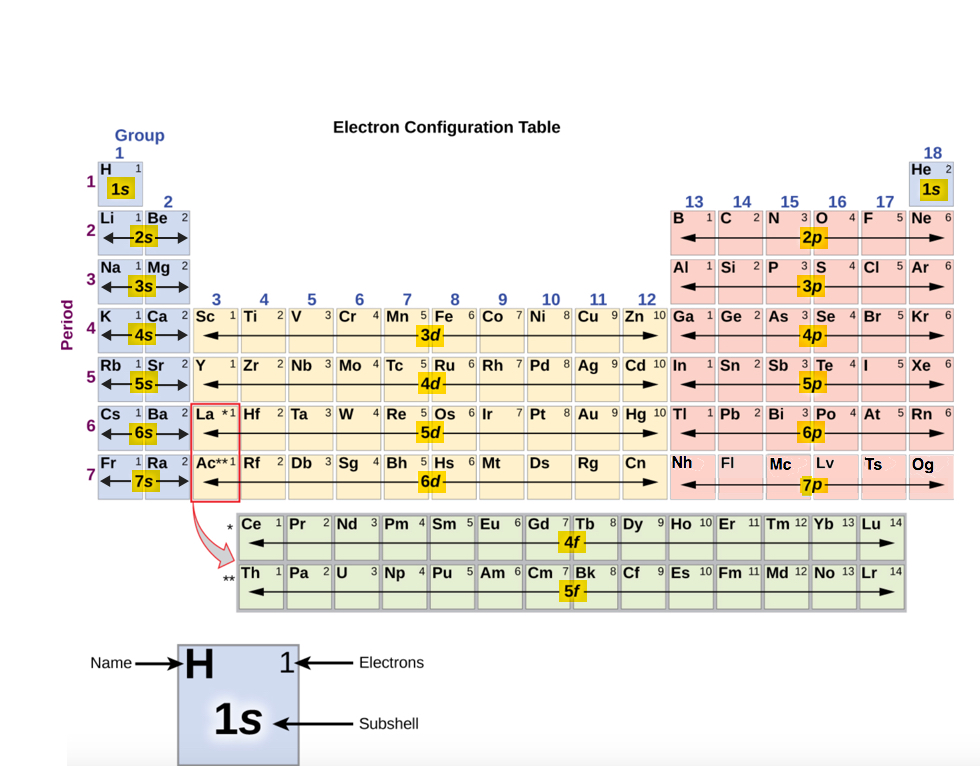
Quantum Numbers (names and their meaning only)
- Principle Quantum Number (n)
- Indicates the main energy level (shell) occupied by the e- (distance from the nucleus)
- Shell number (1st shell is closest to nucleus, 2nd is further, and so on)
- Come from the Bohr Model
- Values of n can only e positive integers (1, 2, 3, etc.)
- As n increases, the orbital becomes larger; the electron has a higher energy and is farther away from the nucleus
- Angular Momentum Quantum Number (l)
- Indicates the general type of shapes of the orbitals
- Nickname is subshell of n
- Designated s, p, d, f
- Values of l are zero and all positive integers less than equal to n-1
- Magnetic Quantum Number (ml)
- Indicates which exact orbital the electron is in
- Describes the orientation of the orbital
- Because an s orbital is spherical, it only has one orientation (ml = 0)
- p orbitals can have three different orientations, one along the x-axis, one along the y-axis, and one along the z-axis
- Spin Quantum Number (ms)
- Indicates the two spin states of an e- in an orbital
- Only 2 e- fit in each orbital, and they spin in opposite directions (up and down)
- Possible m, values are -1/2, + 1/2
- Spin is represented by dashes inside circles
- Orbital notation
Shells
- Distance from the nucleus (principle quantum number)
- Represent ranges in energy
Subshells
- Represent shapes (s, p, d, f)
- One or more orbitals with the same set of n and l values
- Each shell is divided into the number of subshells equal to the principal quantum number, n, for that shell.
- The first shell consists of only the 1s subshell; the second shell consists of two subshells, 2s and 2p; the third of three subshell, 3s, 3p and 3d, and so forth.
- Each subshell is divided into orbitals. Each s subshell consists of one orbital; each p subshell of three orbitals, each d subshell of five, and each f subshell of seven orbitals.
- Angular momentum quantum number
Number of subshells in a shell
- The number of subshells in a shell is equal to the shell number
- 1st shell - 1 subshell
- 2nd shell - 2 subshells
- 3rd shell - 3 subshells
Electron Filling Order: 1s 2s 2p…
- Electron filling tree
- 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f
- Also known as the Aufbau principle
Orbitals
- Three dimensional space that electrons most probably occupy
- Defined by n, l, and ml
- The math equation treats electrons like waves
- You can solve the equation to get the shape in space in which electrons are
- Shapes look like “clouds” of probability
Number of orbitals per subshell: S P D F/ Number of electrons per orbital and per subshell
- S subshell
- Spherical shaped
- 1 orbital, 2 e-
- P subshell
- Peanut shaped
- 3 orbitals, 6 e-
- D subshell
- Double peanut shaped
- 5 orbitals, 10 e-
- F subshell
- Flower shaped
- 7 orbitals, 14 e-
- Each subshell’s name comes from the old spectroscopic description of the lines corresponding to these orbitals
- 1st subshell in a shell = s subshell → sharp
- 2nd subshell in a shell = p subshell → principal
- 3rd subshell in a shell = d subshell → diffuse
- 4th subshell in a shell = f subshell → fundamental
Aufbau Principle
- “Building up”
- An electron occupies that lowest energy possible
- The levels follow a pattern of increasing energy
- Fill starting at nucleus (Bohr Models)
- P subshell → 3 orbitals
- Fill left to right
Pauli Exclusion Principle
- No 2 electrons have the same spin if they are in the same orbital
Hund’s Rule
- Electrons do not pair up until there are no more empty orbitals in that subshell
Orbital Notation
- Representation of electron configuration in which orbital is represented by a circle and dashes
- Each dash represents the number of electrons in each subshell
Electron Configuration
- The correct order electrons are filled in
- The most stable, or ground, electron configuration of an atom is that in which the electrons are in the lowest possible energy level
- All subshells contain a certain number of orbitals
- May be occupied by a single e- or by 2e- having opposite spins
- Like cups
- Shells don’t always get filled from 1 to 2 to 3 etc. because some subshells overlap
Valence/Core electrons
- Valence = outermost
- Valence electrons are electrons in the outer shells
- Core electrons are electrons in the inner shells
- Count the total electrons in the highest shell number
- Do not count electrons in d subshells
- Do count s and p
The Periodic Table and ordering of electrons
- Rows (periods)
- All of the elements in the row have the same number of orbitals
- Columns (groups)
- All of the elements in the column have the same number of (valence) electrons
- Share similar chemical and physical properties because they possess the same # of valence electrons