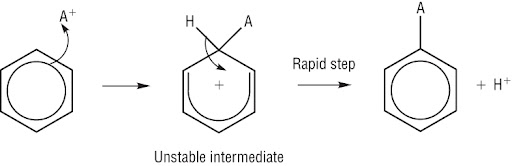
Electrophilic substitution in benzene
benzene is a cyclic aromatic compound
in benzene one electron from each carbon atom is in a p orbital - these p orbitals overlap sideways and the electrons delocalise over the six carbon atoms
this forms a ring of electron density above and below the plane of the carbon atoms - called a pi bonding system
this system of delocalised electrons is energetically very stable - requires lots of energy to break
because of this electron density benzene reacts with electrophiles
because of the delocalised electrons above and below the ring, the electron density between any two adjacent carbon atoms is less than in the double bond in alkenes because of this benzene cannot polarise a bromine molecule and induce a dipole
catalysts are used to create an electrophile