Mole Concept
What is a Mole?
A mole is the amount of substance that contains 6.02 x 10 particles of that substance.
OR
The amount of substance in grams that contains the same number of particles as there are in 12 grams of carbon - 12 / C - 12. (These particles can be atoms, molecules, and ions etc)
Avogadro’s Constant /
The Avogadro’s Law is 1 mole of Carbon has a mass of 12 grams and contains 6.02 x 10 atoms.
Avogadro’s Law
Avogrado’s Law States that equal volumes of all gases under the same conditions at room temperature and pressure contain the same number of molecules.
Atomic Mass
The average mass of one molecule of an element or compound compared to the mass of an atom of carbon-12.
Carbon is the standard which is the atomic mass unit.
Molar Mass
Molar is mass is the mass of 1 mole of a substance.
Unit: g/mol or gmol
Relative Molecular Mass
Relative molecular mass is the ratio of the mass of a compound compared to mass of a carbon 12 atom.
Molecular → Covalent compounds
Formula mass → Ionic compounds
Molar mass has a unit of grams per mole but relative molar mass has no unit, however, they are numerically equal.
To calculate the relative molar mass and the relative formula mass you are adding the atomic masses of the elements in the compound.
Calculating Molar mass
Add the atomic masses of the elements in the compound.

R.T.P and S.T.P
R.t.p is room temperature & pressure.
S.T.P is standard temperature & pressure.
1 mol of gas occupied 24 at r.t.p = 24,000
1 mol of gas occupied 22.4 at s.t.p = 22,400
CONVERSIONS
When converting from one thing to another you must state what you have and what you want to go to.
Quantity → what it is being converted to
Cross multiply
Moles → other units, you multiply
Other units → moles, you divide
Write your answers to 2 dp
Mass → moles
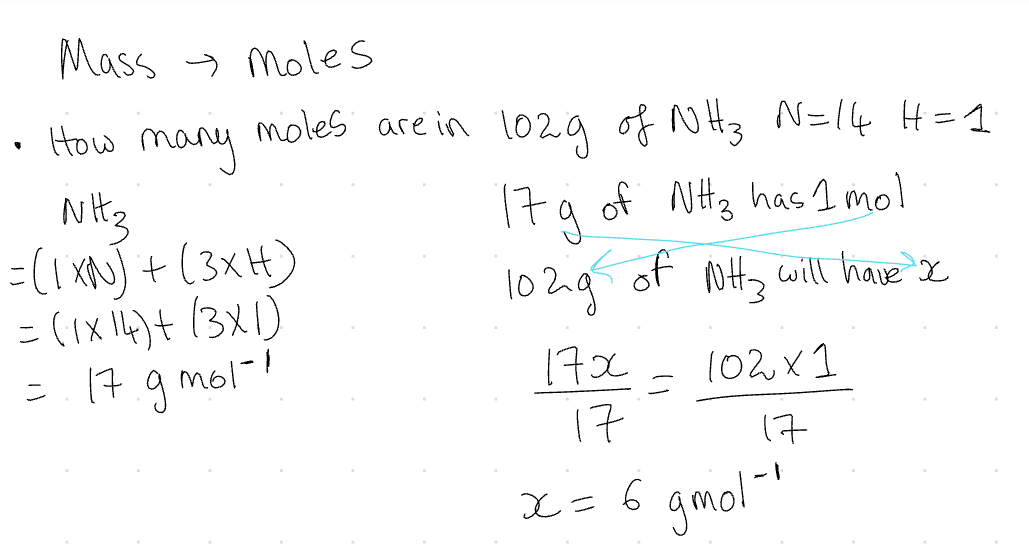
Moles →Mass
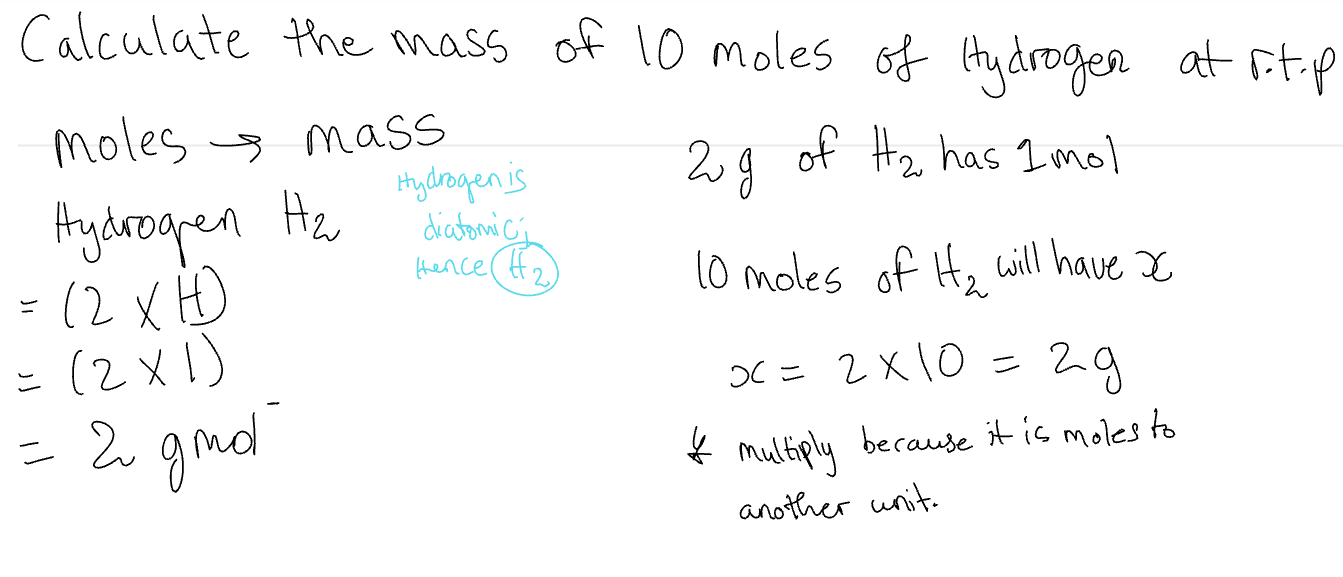
Volume → mols
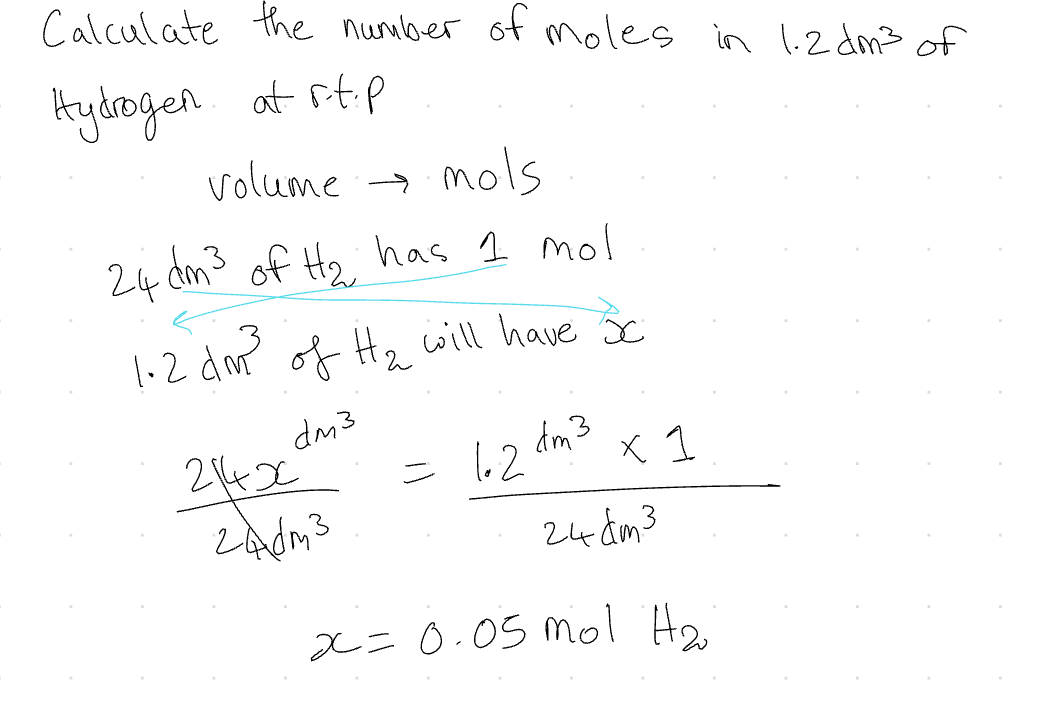
Volume →Mass
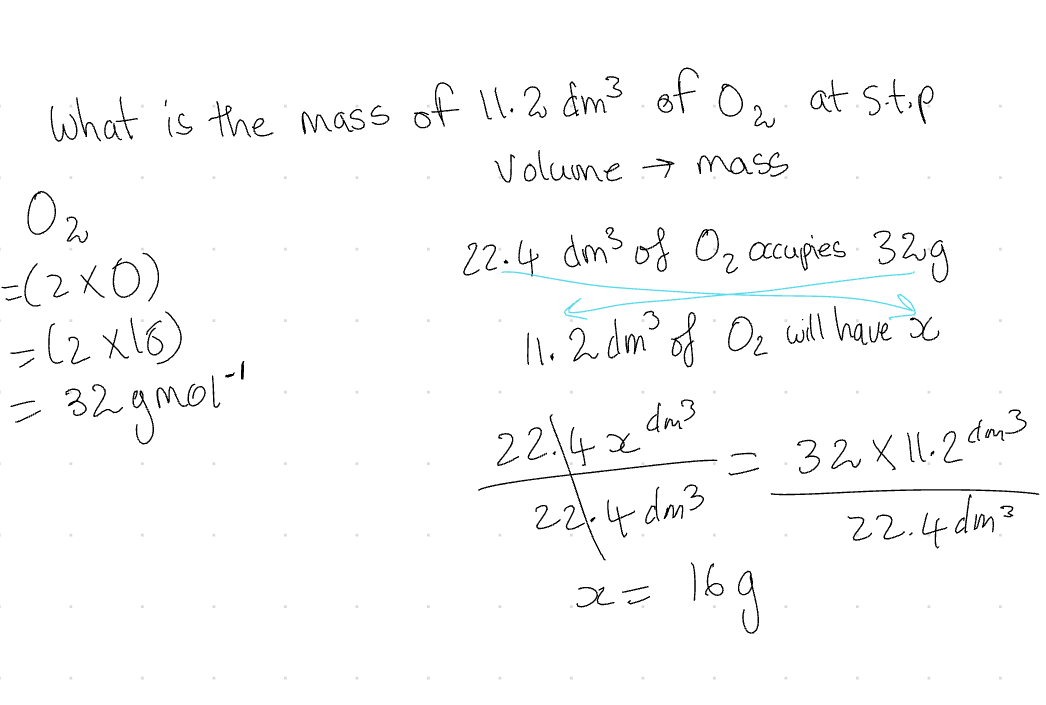
Mass → Molecules
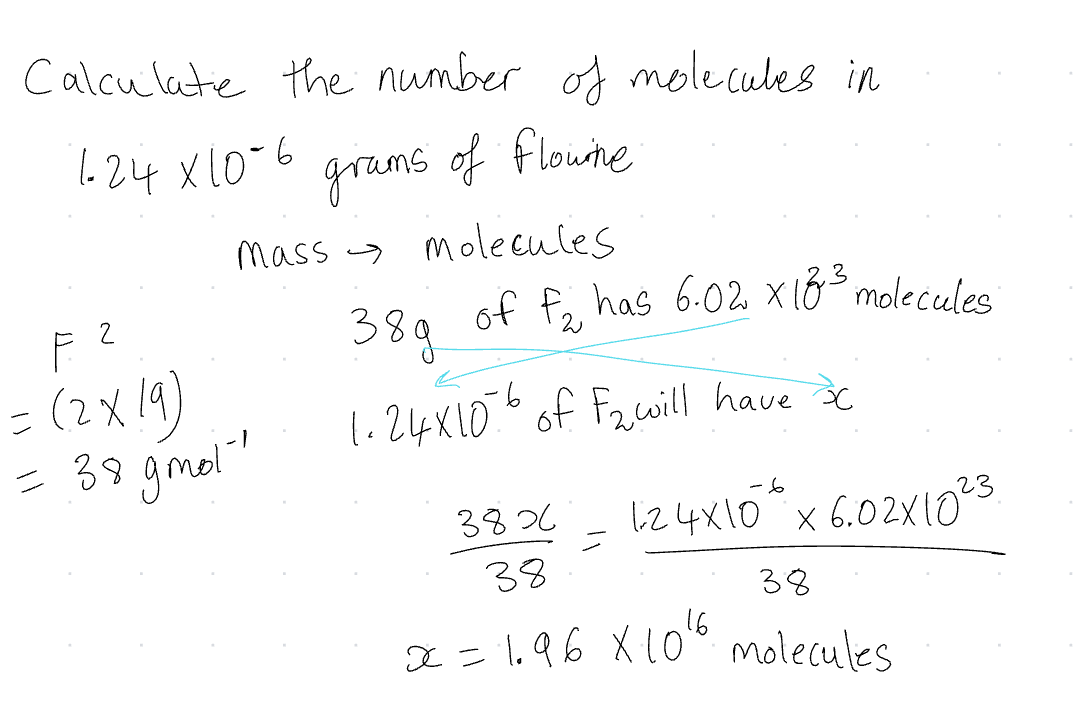
Molecules → Volume
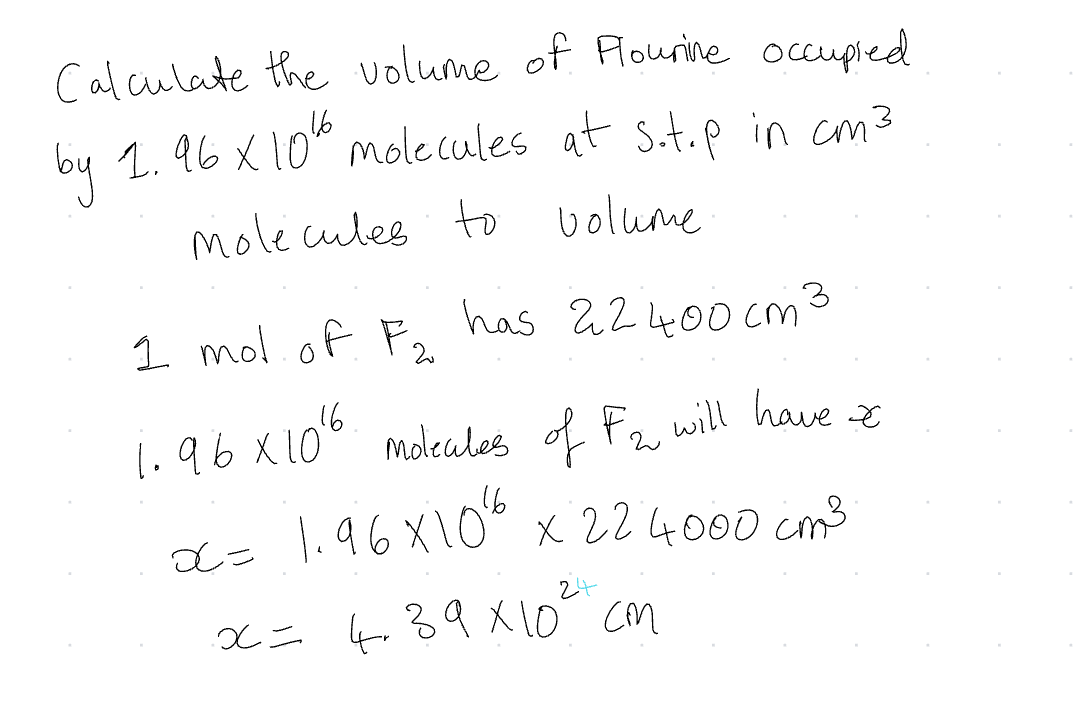
Moles → Volume
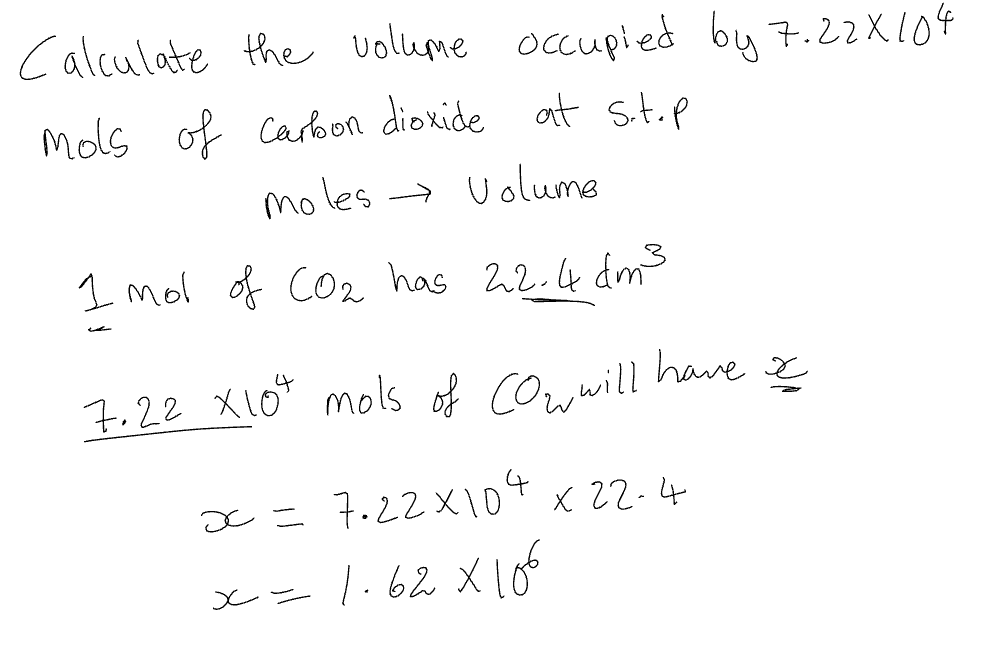
Mass →Atoms
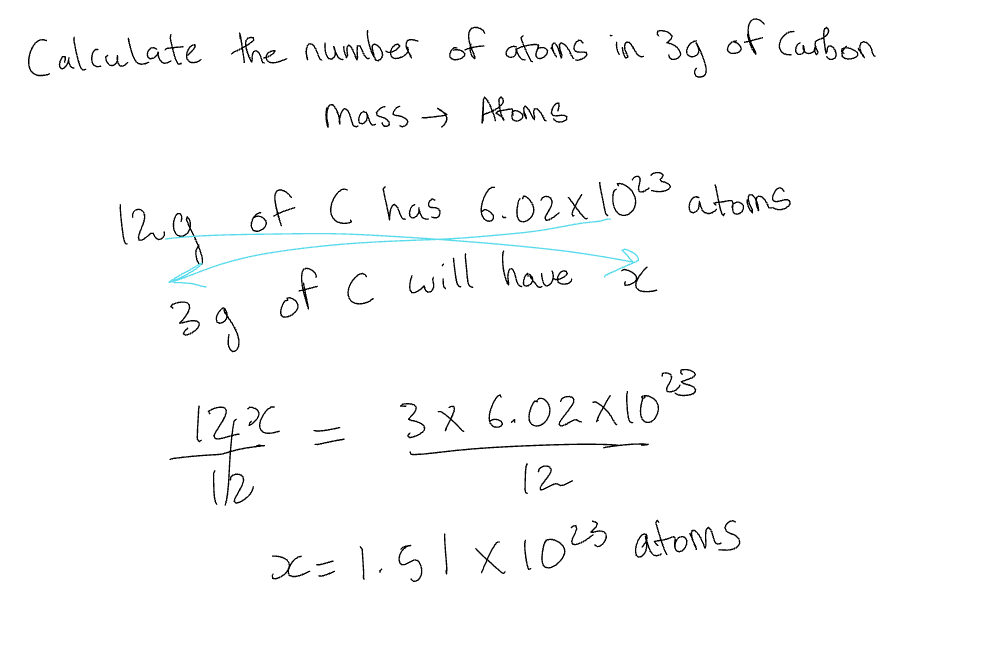
Concentration
Concentration is the amount of solute dissolved in a given volume of solution. (It is a solution because it has already dissolved)
Concentration can be expressed in terms of:
Mass Concentration → mass of solute dissolved in
1 or 1000 of solution.
Unit - g/ or g
Molar Concentration →moles of a solute dissolved in 1 or 1000 of solution.
Unit - mol/ or mol
Mole Fraction → 5 mol
= 5 moles are in 1
1 has 5 moles
Standard Form
Step 1: Write the first number.
Step 2: Add a decimal point after this and write the remaining non-zero numbers.
Step 3: Now count the number of digits after the 1st number. or count the amount of places you moved the decimal point. Now the put the number of digits to the power of 10.
EGS: 14,300,000 in the standard form is 1.43 × 107.
3000 in the standard form is 3 × 10³.
1.98 ✕ 10¹³ and 0.76 ✕ 10¹³.

General steps
Calculate the number of mols in the compound that the information was given for
Write a balanced equation
Mole ratio
Calculate the final answer



