Unit 1: Chapter 5
5.1
Macromolecules are polymers built from monomers
enzymes: catalysts which speed up reactions
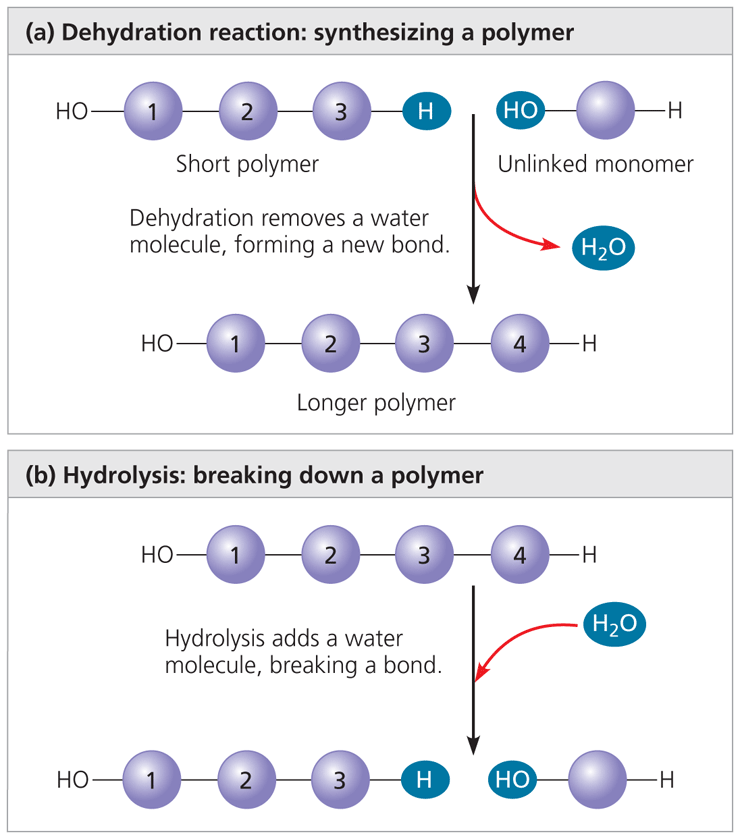
Dehydration reaction: two monomers join together by releasing water, one gives an OH and the other gives a H. Together, which can create a polymer.
Hydrolysis: Polymers being disassembled
- the bond between monomers is broken in addition to water
- one monomer gets the OH group and the other gets a H
- this is used in our digestive tract when we break down food
5.2
Carbohydrates: Sugars and polymers of sugars
- monosaccharides, disaccarides, polysaccarides
- Glucose is the most common monosaccharide
- can be aldoses or ketoses
what binds two monomers together is a glycosidic linkage
Polysaccharides
- storage material
* plants use starch to store energy
* many animals also use starch for this same reason and access to glucose - provide sugar for cells
- protection of the cell
* cellulose is a major component of the cell wall in plants
5.3
Lipids: smallest macromolecule, fats, phospholipids, and steroids
- mix poorly with water
Fats: (triglycerol) glycerol + 3 fatty acids
- the nonpolar C--H bonds make fats hydrophobic
- made from dehydration reaction
- bonded together by an Esther linkage
- saturated: C--C
* can pack together closely and therefore are solid at room temp ex. butter - unsaturated: C=C
* cannot pack closely and are liquids at room temp ex. olive oil
Phospholipids: similar to a fat molecule but instead of 3 fatty acids, only 2 are attached and the third attached to the OH group from the glycerol is a phosphate group. Additionally, a small charged or polar molecule is attached to the phosphate group
- Major constituents of cell membranes
- Has a hydrophilic head (glycerol, phosphate group, polar molecule) but a hydrophobic tail (fatty acids)
- Phospholipids create a bilayer which shields their hydrophobic tails from water
- The bilayer also forms a boundary between the cell and its environment
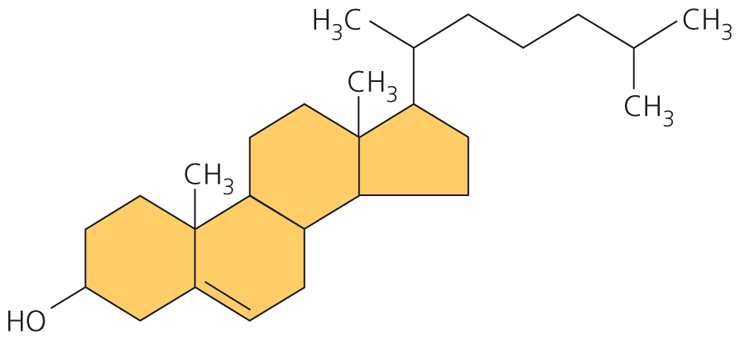
Steroids: consist of a carbon skeleton of four fused rings
ex. cholesterol
- a common component in animal cell membranes
- sex hormones
cholesterol:
5.4
Protein: made up of many polypeptides (amino acids) (possibly thousands)
- help speed up chemical reactions - enzymes
- storage
- transport - hemoglobin transports oxygen
- structural support - keratin, found in hair that makes it stronger
- defence - antibodies help inactivate and destroy viruses
- Made of many amino acids linked by peptide bonds which are referred to as polypeptide chains
Amino Acids: 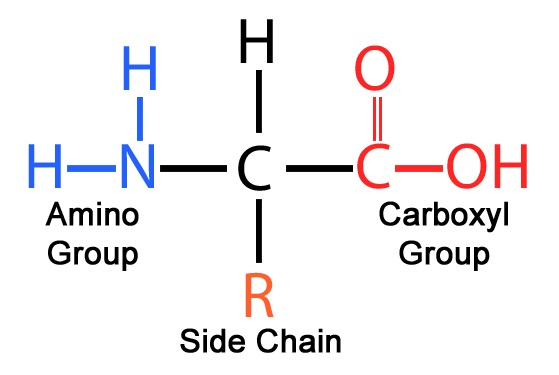
Hydrophobic amino acids:
- Glycine - H
- Alanine - CH3
Hydrophilic amino acids:
- Serine - OH -- CH2
- Cysteine - SH -- CH2
Polypeptides: amino acid polymers
- amino acids are joined together by a peptide bond
- amino end (Nitrogen and 2 hydrogens) is called N-terminus
- carboxyl end (carboxyl group) is called C-terminus
- the polypeptide backbone is the main chain
connecting smaller branches are called side chains 1
Four levels of protein structure:
- Primary Structure - its sequence of amino acids which is specific to each protein
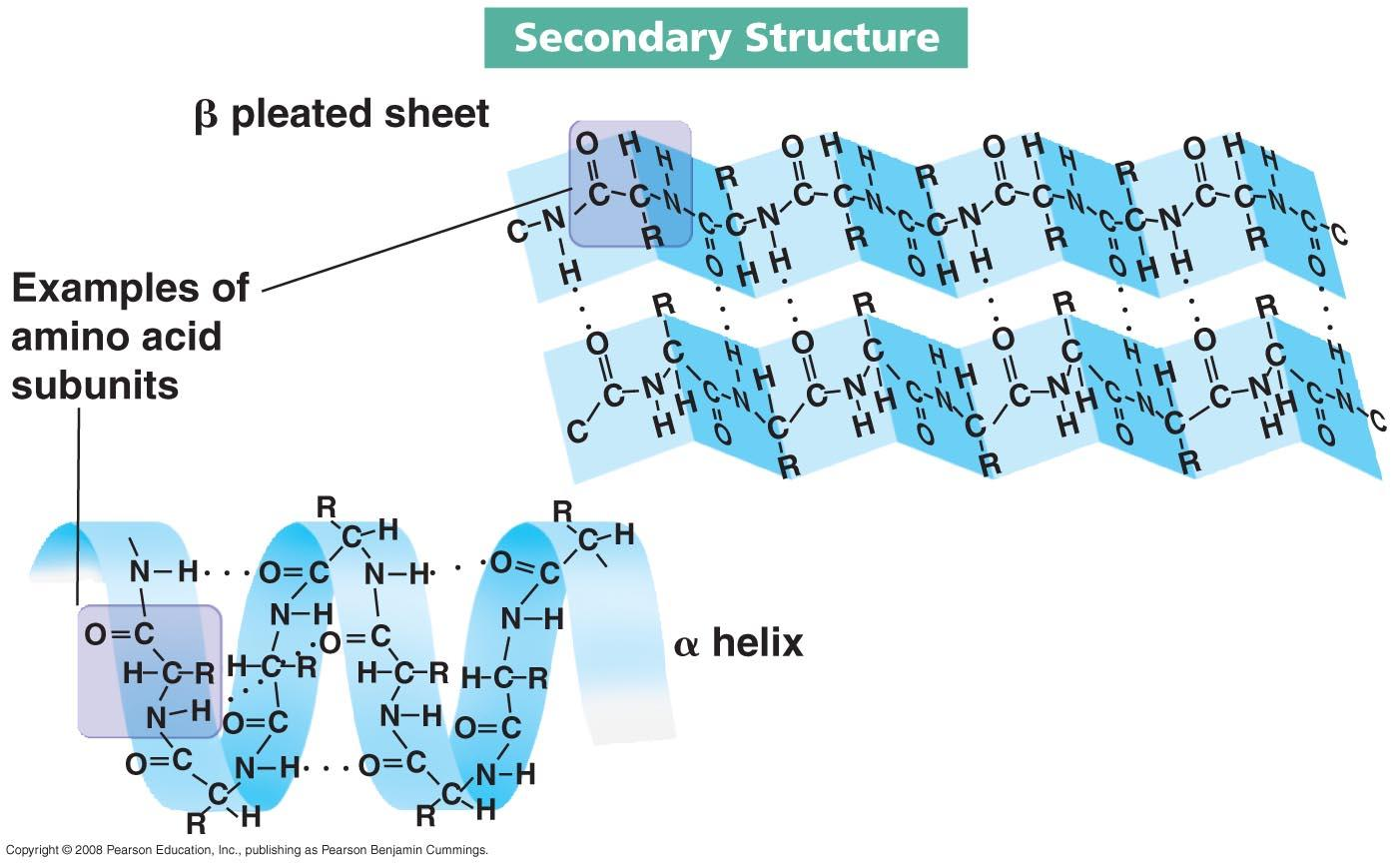
- Secondary Structure - Segments of their polypeptide chains are either coiled or folded
* are a result of the hydrogen bonds between the polypeptide chain
* ^^A helix coil^^ is found on every ^^four^^ hydrogen bonds. Some proteins will only have one while others have multiple A helix coils
* ^^B helix is a pleated sheet^^, two or more pleated sheets side by side are connected by ^^hydrogen bonds^^. The hydrogen bonds make the bonds stronger
Tertiary structure - overall shape of the polypeptide according to the interactions of its side chains (R)
- an example of tertiary structure is hydrophobic interactions
* as a polypeptide folds into its shape, hydrophobic amino acids will cluster in the core of the protein - another example can be disulphide bridges
* two cysteines (SH) groups can interact with each other when close
* from that, the sulphur will bond together (S--S) creating a bridge
Quaternary structure - two polypeptides aggregated together to form one macromolecule
- some proteins only
denaturation - a process where a protein loses its shape due to weak chemical bonds and interactions
5.5
2 types of nucleic acids:
DNA - deoxyribonucleic acid
- applies directions for its own replication
RNA - ribonucleic acid
- synthesis
the flow of genetic information: DNA → RNA → Proteins
Nucleic acids as polymers are called: polynucleotides
a monomer of nucleic acid: nucleotide
What is in a nucleotide: 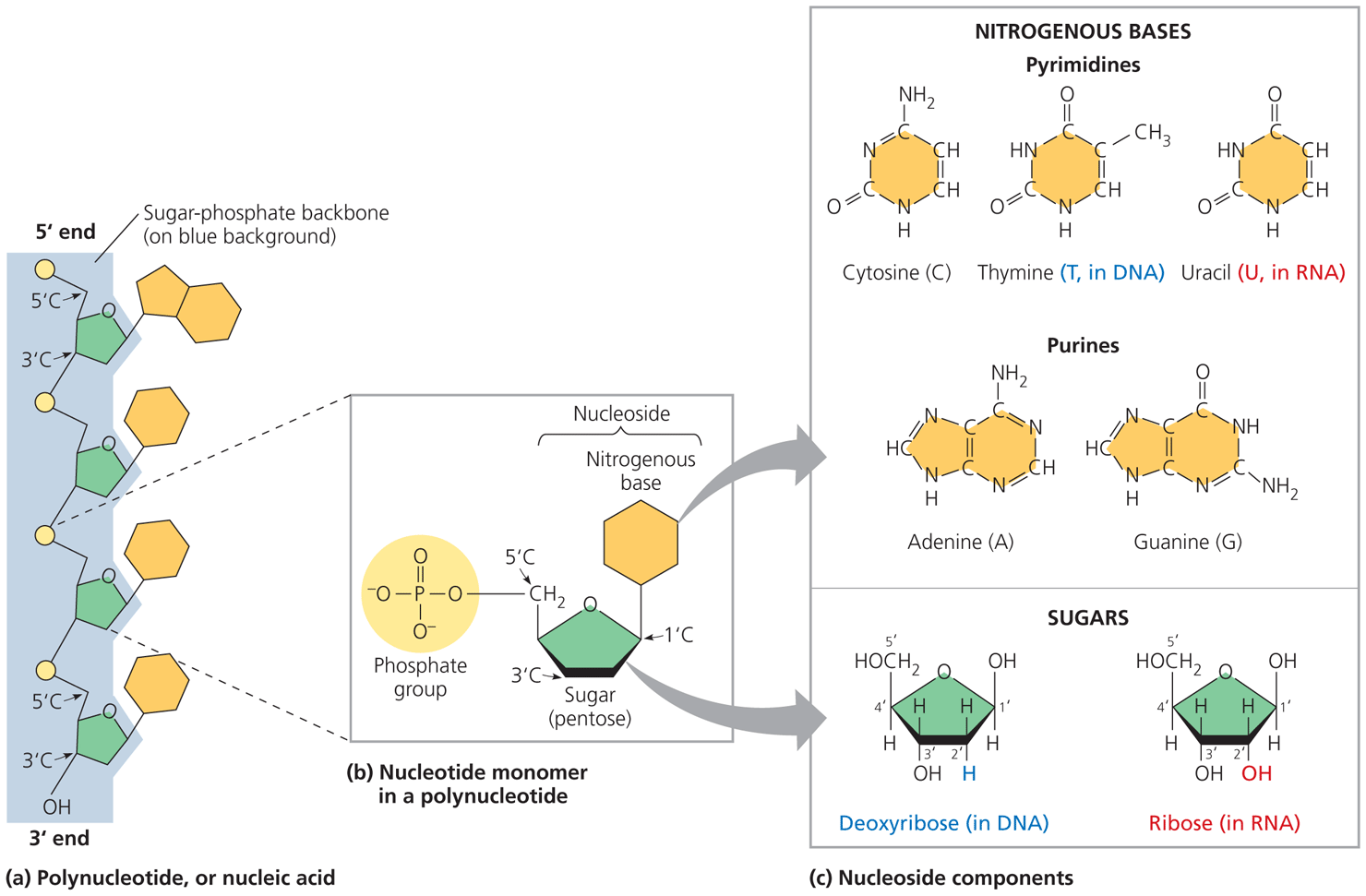 adenine, guanine, and cytosine are found in DNA and RNA while thymine is only found in DNA and uracil is only found in RNA
adenine, guanine, and cytosine are found in DNA and RNA while thymine is only found in DNA and uracil is only found in RNA