Semester 2 guide
Unit 6
Day 2 - Ionic Bond
Ionic bonds form from the attraction of + and - ions
Ionic compound - Made of crystals
A 3D repeating pattern of alternating + and - ions
Properties of Ionic Bond
Strong
solid at room temperature
High melting and boiling point
Made out of metal and non-metal
Does not conduct electricity in a solid state
Conduct electricity in an aqueous (dissolved in water) and liquid state (melted or dissolved)
Empirical Formula - The formula for the Ionic bond where it is arranged in the smallest whole-number ratio
ex. Mg3N2
Day 3 - Covalent bond
Sharing electron
Non-metals only
Single bond - sharing 1 pair of electrons (2 total)
Double bond - sharing 2 pairs of electrons (4 total)
Triple bond - sharing 3 pairs of electrons (6 total)
One electron is donated to the bond from each atom
Molecular compounds/molecules - Atoms are covalently bonded.
Properties of molecular compound
Weaker than an ionic bond
Much lower boiling and melting point
Solid, liquid, gas at room temperature
Non-conductors in any state
Molecular formula - Chemical formula for a covalently bonded group of atoms
Unit 7
Day 1
Ions can be monatomic or polyatomic:
Monatomic: Made up of a single atom
Polyatomic: Made up of multiple atoms
Monatomic cations have the same name as the element
Example: Na+1 = Sodium ion, Ca+2 = Calcium ion
Monatomic anions have the ending of the element name changed to“-ide”
Example: Cl-1=chloride ion, O-2=oxide ion
Polyatomic ions: (See polyatomic ion sheet) are made up of two or more elements covalently bonded with an overall positive or negative charge.
Day 4
Acids are ionic compounds that contain H+1 as their cation.
Acids are named based on their anion.
If the anion ends in…
“-ide” 🡪 hydro ___ ic acid
Example: H2S → Hydrogen sulfide → (Hydrosulfuric acid)
“-ate” 🡪 ___ ic acid
Example: H2SO4 → Hydrogen chromate → Sulfuric acid
“-ite” 🡪 ___ ous acid
Example: H2SO3 →Hydrogen sulfite → Sulfurous acid
Day 5
Naming molecular compounds (non-metals only)
mono – 1 hexa – 6
di – 2 hepta – 7
tri – 3 octa – 8
tetra – 4 nona – 9
penta – 5 deca - 10
Unit 8
Day 1
A Brief History of the Mole…
Avogadro: (1811) An Italian scientist who studied the behavior of gases.
Theorized: “The volume of a gas at a specific temperature and pressure contains equal numbers of atoms or molecules regardless of the nature of the gas.”
Loschmidt (1865): Estimated the average diameter of the molecules in air and was able to calculate the number of particles in a given volume of gas.
Millikan (1910): Measured the charge on an electron. He divided the two from the charge on a mole of electrons and obtained Avogadro’s number.
Perrin (1926): Earned the Nobel Prize for computing Avogadro’s number using many different methods and named this constant in honor of Avogadro. Used oxygen as a standard and proposed “Avogadro’s number is the number of molecules in exactly 32-grams of oxygen.”
The standard was later changed to the carbon-12 isotope.
The presently accepted definition of the mole is:
“The amount of any substance that contains as many elementary entities as there are in 12 grams of pure carbon-12.”
The mole (mol) as a unit in chemistry serves as a bridge between the atomic and macroscopic worlds.
In Latin, mole means “huge pile.”
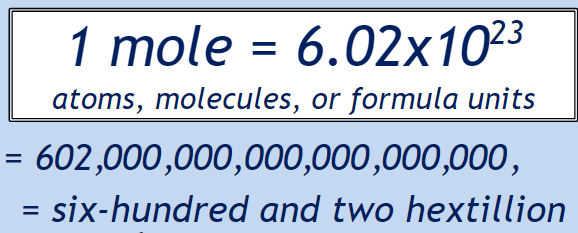
Day 2
The atomic mass on the Periodic Table is the mass of a single atom in amu (atomic mass units).
Example: 1 carbon atom= 12.01 amu
The molar mass (MM) = the mass of one mole of a substance. It is equal to the atomic mass in grams.
Day 5
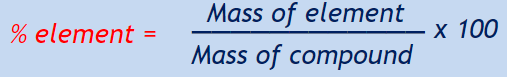
Example: 1 mole of carbon atoms = 12.01 grams

Unit 9
Day 1
A chemical equation represents a chemical reaction.
Reactants → Products
“Yield”
Reactants and products are separated by a plus (+) sign.



Day 3
Law of Conservation of Atoms: There must be the same number of each type of atom before the reaction as after the reaction.
Coefficients: These are numbers that go in front of each substance to indicate the
number of atoms or molecules that are reacting or being produced.
Day 4
Synthesis: two elements to form a compound
Decomposition: breaking down a compound into elements/smaller compounds
metal chlorate 🡪 metal chloride + oxygen
metal carbonate 🡪 metal oxide + carbon dioxide
metal hydroxide 🡪 metal oxide + water
Single Replacement: change by activity series
fluorine + magnesium iodide 🡪 magnesium + iodine fluoride
Double Replacement: changing the metal
Combustion
C - CO2
H - H2O
S - SO2
N - N2
Day 6
A neutralization reaction is a type of double replacement reaction.
Neutralization reaction: Is a reaction between an acid and a base (base – a metal hydroxide, ex: NaOH)
