Oxidative Phosphorylation
Coenzyme Oxidation and Redox Potentials:
NADH(reductant) + H+ + 1/2O2(oxidant) → NAD+ + H2O
Oxidation
NAD+ + 2H+ + 2e- → NADH + H+, E0 = -0.32V.
Reduction
1/2O2 + 2H+ + 2e- → H2O, E0 = 0.816V
ΔE0 = E0(reduction/acceptor) - E0(oxidation/donor)
ΔE0 = 0.816 -(-0.32) = 1.136V.
The nernst equation shows that ΔG0 = -nFE0.
ΔG0 = -(2) x (96.485kJ/Vmol) = -219.21kJ/mol
A reaction with a net positive ΔE0 yields a negative ΔG0 which results in a spontaneous exergonic reaction.
Molecules along the ETC have reduction potentials between the values of the NAD+/NADH couple and the O2/H2O couple.
So the electrons move down the energy scale towards progressively more positive reduction potentials.
OXPHOS-ETC Complexes:
There are four protein complexes in the ETC:
NADH-Coenzyme Q Reductase (I)
Succinate-Coenzyme Q Reductase (II)
Coenzyme Q-Cytochrome C Reductase (III)
Cytochrome C Oxidase (IV)
+ ATP Synthase
— — — — — — — — — — — —
Molecular Components of Complexes:
Flavoproteins:
Tightly bound FMN or FAD (prosthetic groups) may participate in 1 or 2 e- transfer events.
Coenzyme Q (Ubiquinone; CoQ; UQ):
1 or 2 e- transfer reactions.
Cytochromes (b, c, c1, a and a3):
Haem (prosthetic groups) are 1e- transfer agents (i.e. Fe3+ → Fe2+).
Fe-S Proteins:
1e- transfer (Fe2+ and Fe3+ states).
Protein-bound Cu2+:
1e- transfer sites (Cu+ → Cu2+).
— — — — — — — — — — — —
Overview of the Complexes:
Complex I:
Accepts 2e- from NADH (links between glycolysis, TCA, B-oxidation and ETC).
Complex II:
Includes succinates dehydrogenase (links between TCA and ETC).
Entry point for FADH2e- from TCA.
Complexes I and II produce a common product (reduced coenzyme Q (UQH2)) which is a substrate for complex III.
Complex III:
Oxidises UQH2 while reducing cytochrome c (substrate for complex IV).
Complex IV:
Reduces molecular oxygen.
Coenzyme Q:
Coenzyme Q is a mobile electron carrier.
It is highly hydrophobic.
Diffuses freely in the hydrophobic core of the inner-mitochondrial membrane.
Can take part in 1e- or 2e- reactions.
Complex I: NADH-Coenzyme Q Reductase:
~900kDa
More than 30 polypeptide chains.
1 molecule of FMN.
As many as 7 Fe-S clusters.
Containing a total of 20-26 iron atoms.
Step 1:
NADH binds to complex I on the matrix side of the IMM.
Transfer of 2e- from the NADH to FMN:
NADH + [FMN] + H+ → [FMNH2] + NAD+.
Step 2:
Transfer of 2e- from the [FMNH2] → series of Fe-S proteins.
Step 3:
2e- are transferred from Fe-S clusters to coenzyme Q.
Some of the energy liberated by the flow of e- through this complex is used in a ‘coupled’ process to drive protons across the membrane.
As 2e- flow from NADH to Co-Q, 4H+ are pumped out across IMM.
Complex II: Succinate-Coenzyme Q Reductase:
The only TCA enzyme that is an integral membrane protein in the IMM.
~100-140kDa.
FAD is covalently bound to a histidine residue in a 68kDa flavoprotein.
3 Fe-S clusters in 29kDa protein.
2 small subunits, with haem b that binds UQ.
Step 1:
Succinate → Fumarate (reduction of bound FAD to FADH2).
Step 2:
FADH2 transfers e- immediately to Fe-S centres → UQH2.
The small ΔG0 (-5.6kJmol-1) is insufficient to transport H+ across the IMM.
Cytochrome C:
It is a mobile e- carrier that is H2O soluble.
It is globular, meaning that the haem group lies in the centre of the protein.
Associates along the membrane surface in its reduced state.
Carries e- to the 4th complex (cytochrome c oxidase).
Complex III: Coenzyme Q-Cytochrome c Reductase:
It is a dimer consisting of 11 proteins, 248kDa per monomer.
Fe-S Rieske protein (iron sulphur protein (ISP)).
3 Different Cytochromes:
Cyt b (2 haem groups, bL and bH).
Cyt c1.
Cyt c (loosely associated).
Passage of e- through the complex is accompanied by proton transport across the IMM, which is a complicated 2 step Q cycle*, with 2 QH2 molecules.
— — — — — — — — — — — — —
*Q Cycles:
The Q cycle takes place in Complex III. In the first half of the cycle, two electrons of a bound QH2 are transferred.
One to cytochrome c.
One to a bound Q in the second binding site to form the semiquinone radical anion Q.
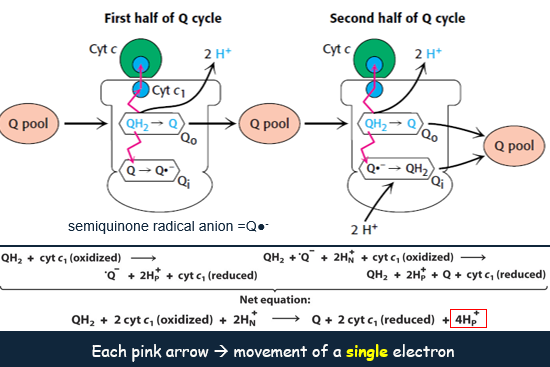
The newly formed Q dissociates and enters the Q pool.
In the second half of the cycle, a second QH2 also gives up its electrons to Complex III.
One to a second molecule of cytochrome c.
One to reduce Q to QH2.
This second electron transfer results in the uptake of two protons from the matrix.
Complex IV: Cytochrome c Oxidase:
Consists of 13 subunits, with a size of 204kDa.
2 Cu centres (2 x CuA and 1 x CuB), which associate with cytochromes a and a3 respectively.
Centres contained within core subunits I, II and III, which induce redox centres and a proton channel.
Reduction of O2 requires the passage of 4e- through this complex.
— — — — — — — — — — — — —
Some of the unused free energy released on e- transfer from cytochrome c to O2 is harnessed by pumping 4H+ out through a proton channel.
4Cytcred + 8H+insitu + O2 → 4Cytcoxi + 2H2O + 4H+out
Complex I, III and IV Drive H+ Across the IMM:
Stores the energy of electron transport in an electrochemical membrane potential.
H+ is driven out of the matrix, causing pH to rise, and the matrix becomes negatively charged.
Both charge and concentration difference tend to attract H+ back into the matrix.

ATP Synthase:
It is a complex that carries out ATP synthesis.
ATP synthase/F1F0-ATPase.
It harnesses H+ flux to drive ATP synthesis.
— — — — — — — — — — — — —
ATP Synthase Structure:
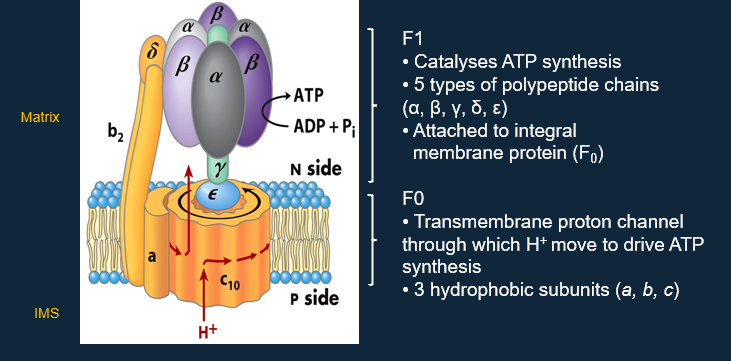
— — — — — — — — — — — — —
ATP Synthesis is driven by Conformational Changes:
Translocation of H+:
Carried out by F0, causing rotation of the c complex and rotation of the γ/ε stalk.
Catalysis of ATP Synthesis:
Carried out by F1, causing conformational changes in β-subunits which alter binding affinity for ATP/ADP, and stabilise ATP.
Energy drives ATP dissociation.
Coupling of H+ Gradient Dissipation with ATP Synthesis:
H+ leakage through F0 drives the molecular rotor (γ/ε), the interactions of γ with the β-subunit drive conformational change.
The Electron Transport Chain:
The synthesis of ATP results in the translocation of 3H+ from the IMS to the matrix via F0.
4H+ (1H+ to bring substrate Pi) are transported to the matrix per ATP synthesised and moved to the cytosol.
Therefore 25% of the energy derived from ETC + OxiPhos is consumed as the electrochemical energy devoted to mitochondrial ATP-ADP transport.
Phosphorylation and Oxidation are Tightly Coupled:
P/O Ratio:
The ratio of phosphate incorporated into ATP to oxygen atoms reduced to water.
It is a measure of the efficiency of coupling of phosphorylation to oxidation, and in turn is a measure of ATP production efficiency.
10H+ are transported out of the matrix per 2e- passed from NADH → O2.
4H+ are transported to the matrix per ATP generated.
P/O ratio for NADH = 10/4 = 2.5
P/O ratio for FADH2 = 6/4 = 1.5