3.1.2 - Carbohydrates
Thursday 14th September ‘23
Monosaccharides & Disaccharides
Monosaccharides: Sweet tasting; soluble; general formula: CH2On (n is between 3 and 7); Gluten, Galactose, Fructose
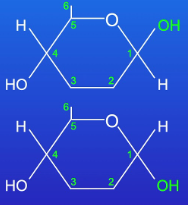
Glucose: Hexose sugar with two isomers. Alpha: Hydroxyl under the carbon ring; Beta: Hydroxyl above carbon ring.
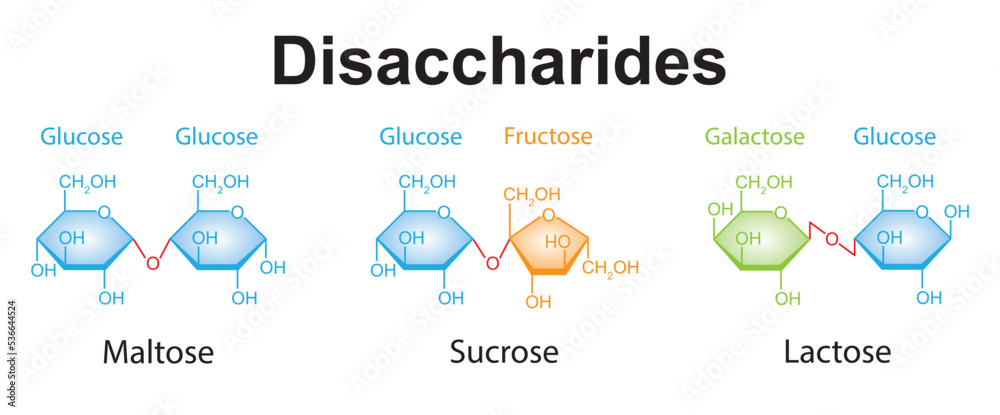
Reducing sugars: Lactose and Maltose
Non-reducing sugars: Sucrose
Dissacharides: In glycosidic bonding, two monosaccharides join in a condensation reaction; Sucrose, Lactose, Maltose
 Polysaccharides
Polysaccharides
Glycogen
Storage form of carbohydrates (excess glucose) in mammals.
Found in large quantities in liver and muscle cells.
Can be hydrolysed to release glucose quickly when it is needed for respiration.
Polymer of alpha glucose joined by 1,4 glycosidic bonds. It forms 1,6 glycosidic bonds at branch points.
Starch
Storage form of carbohydrates (excess glucose) in plants.
Insoluble but can be hydrolysed to release glucose for respiration.
Amylose - alpha glucose; 1,4 glycosidic bonds; unbranched (compact helix).
Amylopectin - alpha glucose; 1,4 glycosidic bonds; 1,6 glycosidic bonds at branches (every 25-30 molecules).
Cellulose
Storage form of carbohydrates (excess glucose) in plants.
Linked by hydrogen bonds to form a microfibril, macrofibril and cellulose fibre (forms cell walls)
Unbranched polymer of beta glucose which forms 1,4 glycosidic bonds by flipping.