19.2 The equilibrium constant Kp
Mole fraction and partial pressure
Equilibria involving gases are usually expressed in the terms of Kp, the equilibrium constant in terms of partial pressure, for gases it makes it easier to measure pressure than concentration.
Concentration and pressure are proportional to one another and Kp has a direct relationship to Kc.
Mole fraction
Under the same conditions of temperature and pressure, the same volume of different gases contains the same number of moles of gas molecules.
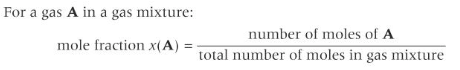
 Partial pressure
Partial pressure
In a gas mixture, the partial pressure p of a gas is the contribution that the gas makes towards the total pressure P. The sum of the partial pressures of each gas equals the total pressure.
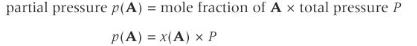

Dissolving oxygen in the blood
The amount of oxygen that can dissolve in the blood is proportional to the partial pressure of oxygen breathed into the lungs. At equilibrium in the lungs, each cubic decimeter of the blood can dissolve about 3cm3 of gaseous oxygen.
An adult human only has about 5dm3 of blood and so 15cm3 doesn’t seem like much, as quickly as oxygen forms, it combines with haemoglobin in the blood, which allows more than 200cm3 of oxygen to effectively dissolve in each dm3 of blood.
Normal air at 1 atmosphere pressure has a pressure of 101 kPa. The 21% of oxygen in the air has a partial pressure of just over 21 kPa.
On earth, as altitude increases, the atmospheric pressure decreases.
People are able to survive at a higher altitude as the body compensated by producing more haemoglobin, however at very high altitudes the partial pressure of oxygen may be so small so very little may dissolve. Oxygen tanks are used and therefore pure oxygen is breathed in.
The equilibrium constant Kp
Written similarly to Kc but partial pressures replacing the concentration terms.

p is the partial pressure
Suitable units are kilopascals (kPa), pascals (Pa) or atmospheres (atm) but the same unit must be used for all gases.
The power for the partial pressure is the balancing number in the chemical equation.
Kp only includes gases as they have partial pressure, any other species must be ignored.

