2.1.1: Reaction Rates:
13.1: Factors that Affect the Rate of Chemical Change
Five factors that influence the rate of a reaction:
Chemical nature of the reactants: Different substances react at different rates depending on their chemical properties, such as bond strength and molecular structure.
Ability of the reactants to come in contact with each other: The greater the surface area of the reactants, the more collisions can occur, leading to an increased reaction rate.
Concentrations of the reactants
Temperature
availability of catalysts
Factors
Some reactions are faster due to certain elements losing electrons easier than others, which can significantly influence the overall reaction rate. For example, metals tend to lose electrons more readily, leading to faster oxidation reactions compared to nonmetals.
Reactions are carried out in liquid and gaseous states so as to let the molecules react and collide. When both reactants are in the same phase, it is a homogenous reaction, a heterogenous mixture is two different phases.
Concentrations example: if air were 30% O2 instead of 21%, it would be impossible to stop forest fires.
Temperature of system: Increasing the temperature of a system generally increases the reaction rate, as higher temperatures provide more energy to the molecules, allowing them to collide more frequently and with greater intensity.
Prescence of a catalyst: A catalyst lowers the activation energy required for a reaction to occur, thereby increasing the reaction rate without being consumed in the process.
13.2: Measuring Reaction Rates
Rate is a change in quantity over time.
Change in concentration of X with respect to time =
(Concentration of X at timeFinal - concentration of X at timeInitial) / (tFinal - tInitial)
When measuring rates, there is no negative.
units for rates: mol / (L s), mol L^-1 s ^-1, or M s^-1
Reaction rates are not constant, but usually change as the reactants are used, due to the concentration of the reactants decreasing, thus decreasing the reaction rate.
The rate at which a substance is being consumed at a certain moment is the instantaneous rate.
The instantaneous rate can be measure by observing a graph and by subtracting the initial concentration from the point measured, over the time chosen - the initial time.
13.3: Rate Laws
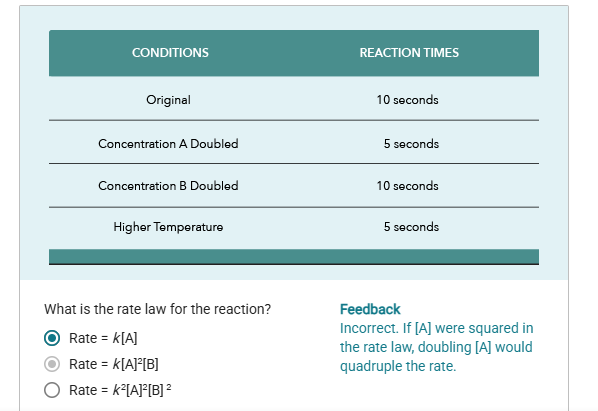
Suppose reaction A + B = Product
It’s rate of reaction can be expressed as: rate = k[A]^m [B]^n
Where k is the rate constant, m and n are the reaction orders with respect to reactants A and B, respectively, which indicate how the rate is affected by the concentration of each reactant.
The rate constant changes with temperatureand is influenced by the activation energy of the reaction. Typically, as the temperature increases, the rate constant k also increases, leading to a faster reaction rate.
The value of k is large for fast reactions and small for slow reactions.
The exponents identify the reaction order for each reactant
The overall reaction order is equal to the sum of the exponents.
In a 0-Order reaction,the rate of reaction is constant and independent of the concentration of the reactant, meaning that changes in concentration do not affect the speed of the reaction.
in a first-order reaction, the rate of reaction is directly proportional to the concentration of one reactant, indicating that as the concentration increases, the reaction rate also increases correspondingly.
in a second-order reaction, the rate of reaction is proportional to the square of the concentration of one reactant or to the product of the concentrations of two different reactants, meaning that if the concentration doubles, the reaction rate increases by a factor of four.
To describe how the rate of a reaction varies with the concentrations of reactants, use the differential rate law.
Rate = -d[A]/dt = k[A]
It gives the initial rate of the reaction, or the instantaneous rate at any given time.
the addition of the differential rate law related tothe concentration of reactants to time produces the integrated rate law:
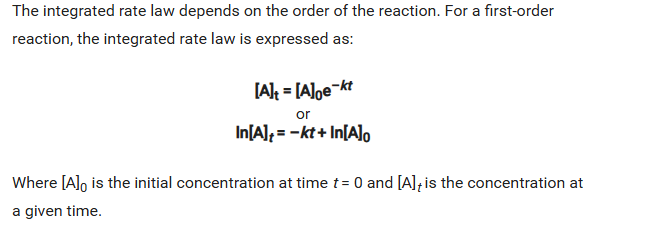
It can be represented graphically as so:
Steps of collecting and analyzing kinetic data:
Experimental design
data collection
data analysis (graphing and interpreting data)
writing the rate law for reaction
The units for rate constant are just variations of the rate law
0-Order: rate = k[A]^0 = K; M/s
1-Order: rate = k[A]; with units of k times molar (A), equals Molar per second, but the unit of k is 1/s
A better way to find this is by solving for k: K = rate/[a](concentration) = M/s / M = 1/s
2-Order: rate = k[A]^2; on the left side we have M/s, while the right side has M². So, molar squared times something equals molar per second.
To ensure seconds remains on the bottom, add 1/s for units for K.
one of the concentration terms must be removed, so it becomes 1/(m*s)
So the unit for k is 1/ m*s
Reactions and Collisions
Reaction rate = Change in concentration / change in time.
Zero-order reaction: The rate of the reaction is constant as the reaction progresses, regardless of the concentration of the reactant.
First-order reaction: The rate of the reaction is directly proportional to the concentration. As the reactant is consumed, the rate of reaction decreases in a linear relationship with reactant concentration.
Second-order reaction: The rate of the reaction is proportional to the square of the concentration. For this type of reaction, the rate of the reaction decreases rapidly (faster than linearly) as the concentration of the reactant decreases.
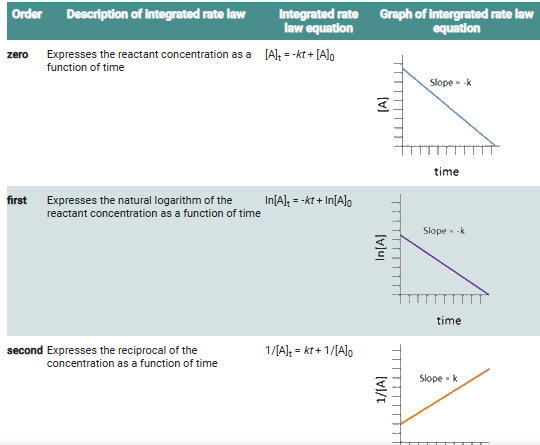
The integrated rate laws, with some adjustments of terms, can be plotted as straight lines to determine the order of the reaction and the rate constant, allowing for a clear visualization of how concentration affects the rate over time.
these are the quantities you should plot versus time for each type of reaction in order to get a straight line:
Zero order: [A]t
First order: ln[A]t
Second order: 1/[A]tTwo methods of finding the reaction order:
The method of initial rates: Determine how the initial reaction rate changes as the initial reactant concentration changes, and then compare this to Rate = k[A]t.
The integrated rate law method: Plot the reactant concentration data versus time in three different ways, [A]t, ln [A]t, and 1/[A]t, to see which plot is linear.
The time for half of the reactant in a first-order reaction to turn into product is given by the half-life, t1/2:
t1/2 = ln(2)/k
This reaction only depends on the rate constant, k.
In radioactive decay, the half-life of a radioactive element is considered a constant
The previous equations do not find the rate of reaction, to find it find the change in concentration over change in time