LO16 notes: VSEPR shape and polarity
TERMINOLOGY: VSEPR, electron regions, polarity, molecular geometry, bond polarity, and electronegativity
Lewis Strucure
What is missing from a lewis structure?
Its shape!
VSEPR (Valence Shell Electron Pair Repulsion)
VSEPR helps us find the shape of molecules from the lewis structure
Each electron groups around a central atom repels one another, this repulsion defines the geometry of the molecule
This results is 5 electrom group arrangements of minimum energy seen in a large majority of molecules and polyatomic ions

Electron center geometry is NOT (always) the same as molecular shape
To determine the shape of a molecule consider only atoms not the lone pairs
Electron center vs molecular geometry (shape)
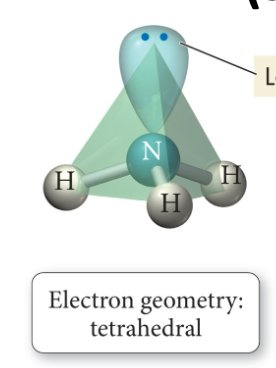
Electron center geometry: lone pairs are considered (which are the 2 dots in the picture), and bonded pairs that are part of a bond