Test Sighhhhhh
Bonds
Ionic bonds
Metal + Nonmetal
If the difference between 2 atoms electronegativity is >2.0 , higher electronegativity atom steals electron from lower electronegativity atom
Particles of opposite charge (+ and -) have electrostatic attraction and forms ionic bond
Covalent bonds
Nonmetal + Nonmetal
Difference in electronegativity <1.7, share electrons
Polar vs. Nonpolar
Polar covalent bond - difference in electronegativity > 0.5 - electrons skew towards one atom
Nonpolar covalent bond - difference in electronegativity <0.5
<0.5 - Nonpolar Covalent
0.5 to 1.7 - Polar Covalent Bond
>2.0 - Ionic Bond
Valence Electrons and Lewis Structures
Valence electrons count valence electrons by each dot
Fill each side of letter up before pairing
Covalent bonds represented by a line
Formal Charge
Occurs if the # of electrons an atom contributes to the structure is different from typical valence (ion)
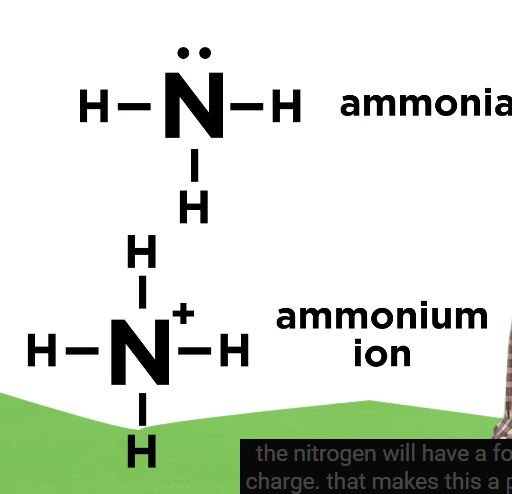
Octet Rule and Exceptions
Allows us to predict how atoms will form chemical bonds
8 valence electrons = octet
Many exceptions
Molecular & Electron Geometries and Bond Angles
VSEPR - Valence Shell Electron Pair Repulsion
Electron clouds repel eachother, therefore molecules automatically adopt a geometry so atoms can be as far away as possible
Hybridized orbitals
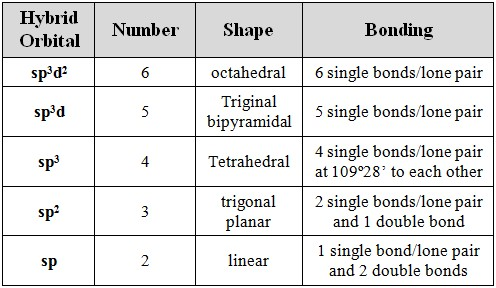
number of electron domains → hybridization of central atom → electron domain geometry
Lone pairs connected to central atom count as electron domains but don’t take up much space - Angles represent a higher amount of atoms but there are less (H2O)
Acids
All acids start with H
Binary acids: HYDROGEN + NONMETAL ELEMENT
Oxyacids: HYDROGEN + OXYGEN + ONE OTHER ELEMENT
Hydrogen bonded to oxygen
Naming: Hydro + Stem + -ic acid
Ionic compounds with oxygen added
ate ending - ic acid
ite ending - ous acid
hypo - ite ending becomes hypo - ous acid
per - ate ending becomes per - ic acid
Ionic Compounds
H+ PO 3- 4 → H3 PO4
H+, H+