Topic 5: Formulae, Equations and Amounts of Substance
The Mole:
1 mole of a substance = 6.0×10²³ number of particles. This is known as avogadro number Example: 1 mole of CH4 contains 6.02 x 10 23 particles 1 mole of NH3 contains 6.0×10²³ particles
Molecule: two or more atoms joined together by a covalent bond
Ion: charged particle formed from the loss or gain of electrons
Molar Mass:
The molar mass (Mr) of a substance is the mass per mole of the substance in g mol 1
It is equal to the relative atomic mass (for an atom) or the relative formula mass (for a compound or molecule).
mass = Mr x moles
Topic 5: Formulae, Equations and Amounts of Substance
Empirical Formulae
Molecular formula: the actual number of atoms of each element in a molecule
Empirical formula: simplest whole number ratio of atoms of each element in a compound
Determining Molecular Formula
Step 1: Work out the Mr of the empirical formula: Mr(C3H7) = (3 x 12) + (7 x 1) = 43 g mol 1 Step 2: Divide the given molar mass by the Mr of the empirical formula: 86 / 43 = 2 This tells you how many times larger the molecular formula is than the empirical formula. Step 3: multiply the empirical formula by the answer from step 2 to get the molecular formula: 2 x C3H7 = C6H14
Empirical determinations: rounding
Rounding to find the ratio:
Number is 0.1 away from a whole number: round it off to the nearest whole number Number is not 0.1 away: multiply it by a factor that makes it a whole number or 0.1 away e.g. if 1.5 then x by 2 and 1.7 x 3.
Combustion Analysis
Combustion Equations: an organic compound composed entirely of hydrogen and carbon atoms
Organic Compound: a chemical compound that contains carbon, typically bonded to hydrogen, and often to other elements like oxygen, nitrogen, sulfur, or phosphorus
Burning a hydrocarbon (or carbohydrate) completely in oxygen produces carbon dioxide and water only.
Combustion Analysis:
When a compound is completely burned, it is possible to collect and measure the masses of carbon dioxide and water formed. This allows us to work out the moles of carbon, hydrogen and oxygen (if present) in the original compound and therefore the empirical formula.

Rule of Co2 with carbon, and H2o with hydrogen.
Same moles of carbon for c02
Double the moles of hydrogen in h20 e.g


Topic 5: Formulae, Equations and Amounts of Substance
Balancing Equations C3H6 + O2 > CO2 + H2O Success criteria 1. Write out the number of each atom on both sides
2. Balance each element. Leave until the end anything whi
ch occurs more than once on either side
3. Use numbers in front of the compounds to aid balancing
4. Don't touch small numbers within compounds You can use fractions to balance
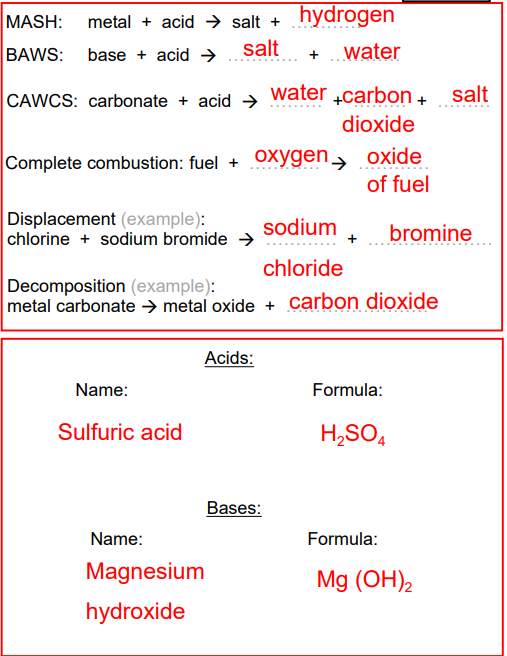
Reactions Recap:
Ions to learn: