Chapter 17: Acids and Bases
Properties of Acids
General Characteristics:
Acids are commonly used in laboratory and industrial settings.
Example of laboratory use: Dissolving metals in soil using acids like nitric acid.
Common Acids
Acetic Acid: Key component of vinegar, contributes sour taste.
Hydrochloric Acid:
Used in metal cleaning and cleaning products.
Major acid in the human stomach, responsible for acid reflux and heartburn.
Sulfuric Acid: Used in fertilizer production and batteries.
Nitric Acid: Widely used in fertilizers.
Citric Acid: Found in fruits, contributes to tartness.
Carbonic Acid: Present in carbonated drinks; derived from carbon dioxide in water.
Hydrofluoric Acid: Extremely corrosive, can etch glass, dangerous to handle.
Phosphoric Acid: Commonly used in fertilizers.
Ammonium ( ): It acts as an acid by donating a proton ( ) to a base, reverting to ammonia ( ).
Naming Acids:
Distinctions based on suffixes:
Acids ending in -ate lead to the suffix -ic in the acid name.
Acids ending in -ite lead to the suffix -ous.
Example: Sulfate → Sulfuric Acid; Sulfite → Sulfurous Acid.
Monoprotic: has one H
Diprotic: has two H
Triprotic: has three H
Properties of Bases
General Characteristics:
Bases typically have a bitter taste (e.g., coffee, chocolate) and feel slippery (due to soap-like consistency when mixed with water).
Bases are present in cleaning products (e.g., drain cleaners).
Common Bases:
Sodium Hydroxide: Frequently used in laboratories.
Potassium Hydroxide: Similar applications as sodium hydroxide.
Sodium Bicarbonate (Baking Soda): Used in cooking, neutralizes stomach acid.
Sodium Carbonate: Used to soften water and in glass production.
Ammonia (NH₃): Acts as a base but does not contain hydroxyl ions in its natural form.
Definitions of Acids and Bases
Three definitions: Arrhenius, Bronsted-Lowry, Lewis.
No individual definition of acids and bases is correct, each is useful in a given instance
Arrhenius Definition:
Acid: Substance that produces hydrogen ions (H⁺) in aqueous solution.
Base: Substance that produces hydroxide ions (OH⁻) in aqueous solution.
Example: Hydrochloric acid (HCl) dissociates in water to form H⁺ and Cl⁻.
Acids and bases combine to form water, thereby neutralizing each other
acid + base → salt + water
Example: HCl (𝑎𝑞) + NaOH (𝑎𝑞) → NaCl (𝑎𝑞) + HO (𝑙)
Bronsted-Lowry Definition:
Acid: Proton (ion) donor.
Base: Proton (ion) acceptor.
Example: In the reaction of ammonia with water, ammonia accepts a proton to become ammonium (NH₄⁺).
Lewis Definition:
Lewis Acid: Electron pair acceptor.
Lewis Base: Electron pair donor.
This definition expands the scope of acids and bases beyond just protons.
Arrhenius Acid-Base Reactions
Neutralization Reaction:
Reacting an acid with a base yields water and a salt.
Example: The reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) produces sodium chloride (table salt) and water (H₂O).
Hydronium Ion
The ions produced by the acid are so reactive that they cannot exist in water.
Formation of the hydronium ion (H₃O⁺) from hydrogen ions and water:
When protons (H⁺) combine with water molecules, hydronium ions are formed, indicating an acidic solution.
Chemists often use H+ (aq) and H3O+ (aq) interchangeably to mean the same thing – an H+ ion that has dissolved in water
Bronsted-Lowry Definition
Acid: Proton (H+ ion) donor
Base: Proton (H+ ion) acceptor
All Arrhenius acids and bases are acids/bases under the Brønsted-Lowry definition. However, some B-L acids and bases are not Arrhenius acids/bases.
NH3 does not inherently contain OH ions but it still produces OH ions in solutions.
According the B-L definition, is a base because it accepts a proton from water
Amphoteric Substances
According to the B-L definition, substances (such as H2O) can act as acids or bases
H2O as donor (acid):
H2O as acceptor (base):
Acid-Base Pairs and Conjugates
Conjugate Acids and Bases:
A conjugate acid is the species formed after a base gains a proton.
A conjugate base is the species that remains after an acid has donated a proton.
Example: Ammonium (NH₄⁺) is the conjugate acid of ammonia (NH₃).
Strong vs. Weak Acids and Bases
The dissolved ions act as charge carriers, thereby conducting electricity
Substances that completely dissociate into ions in water are strong electrolytes
Compounds that do not dissociate into ions in water are nonelectrolytes
Strong Acids: Completely dissociate in water and are strong electrolytes ()
Examples: Hydrochloric acid (HCl), hydrobromic acid (HBr), sulfuric acid (H₂SO₄, diprotic).
All hydrohalic (derived from halogens) acids are strong acids that completely ionize in water except hydrofluoric acid (HF)
Weak Acids: Do not completely dissociate in water and are weak electrolytes (⇌).
Examples: Hydrofluoric acid (HF), acetic acid (CH₃COOH), carbonic acid (H₂CO₃, diprotic).
The degree to which an acid is strong or weak depends on the attraction between the anion of the original acid (the conjugate base) and the H+, relative to the attractions of these ions to water.
If the attraction between H+ and A is weak the forward direction is favored and the acid is strong
If the attraction between H+ and A is strong the reverse direction is favored and the acid is weak
Dissociation Constants ( value)
Ka (Acid Dissociation Constant):
A measure of the strength of an acid in solution.
Higher Ka indicates a stronger acid.
The equilibrium expression for dissociation is given by: , where [HA] is the concentration of the undissociated acid.
[ ] is not included in calculations as it is a pure liquid.
Amphoteric Water ()
Water is amphoteric: can act as both an acid and a base.
Acts as a base when mixed with an acid (hydrofluoric acid in this case).
Produces hydronium ions (H₃O⁺) when acting as a base.
Acts as an acid when mixed with a base, producing hydroxide ions (OH⁻).
Water can autoionize, producing both hydronium and hydroxide ions on its own.
Reaction:
Identifying the ion product/dissociation constant of water ( ) at 25°C: no division by reactants as it is just pure liquid water
or
At neutral pH:
Concentration of H₃O⁺ and OH⁻ is equal:
or
Importance of : Provides baseline for acid-base reactions.
At neutral pH, both H₃O⁺ and OH⁻ equal each other.
Calculating Hydroxide Ion Concentration using
For a given , calculate using:
If :
Example Calculation:
Identify if acidic, basic, or neutral:
If [H_3O^+] > [OH^-], solution is acidic.
If [H_3O^+] < [OH^-], solution is basic.
pH (hydronium concentration)
pH is defined as the negative log of hydronium concentration:
Example: If , then:
Sig Figs and pH
Sig figs are essential for pH reporting.
If has:
Two significant figures: pH should have two decimal places.
Three significant figures: pH should have three decimal places.
Characteristics of pH scale
Less than 7 (pH < 7): acidic
More than 7 (pH > 7): basic
Exactly 7 (pH = 7): neutral
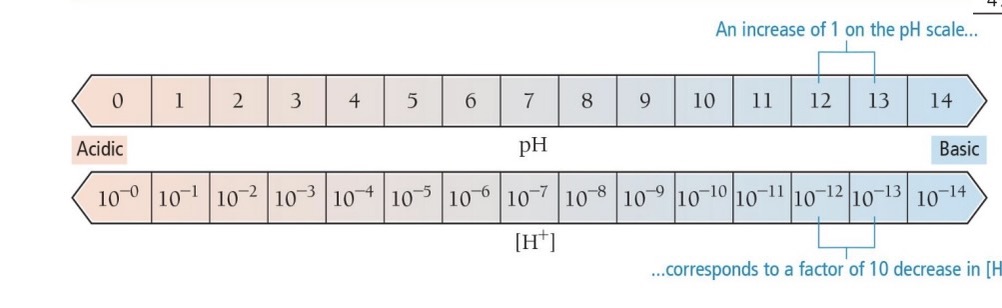
Each unit change in pH represents a tenfold change in acidity:
Example: pH 2 (Lime) is 10x more acidic than pH 3 (Plum).
pH changes from 2 to 4 represent a 100x change in acidity.
Example Problem 1
Determine pH from given hydronium concentration
= 3.74 < 7 = acidic
Example Problem 2
Calculate the pH if .
First solve for concentration
H
Then use pH formula
> 7 = basic
Example Problem 3
Calculate concentration for a solution with a pH of 4.80
using this:
$
pOH (hydroxide concentration)
When pOH is less than 7: solution is acidic.
When pOH is greater than 7: solution is basic.
pH + pOH = 14 (at 25°C)
Strength of Acids ()
Acid strength is determined by dissociation constant (Kₐ).
Large Kₐ = strong acid.
pKₐ is calculated as:
Stronger acids have larger but smaller values.
Bases have the same: and
Two Sources of Hydronium
Hydronium can come from:
Dissociation of the acid
Autoionization of water.
Except in extremely dilute acid solutions (< 10-5 M), the autoionization of water contributes a negligibly small amount of H3O+ compared to the ionization of the acid
The addition of the H3O+ from the acid actually shifts the autoionization reaction to the left, producing even less H3O+
Strong and Weak Acids
Strong acids fully dissociate in water (e.g. HCl).
The concentration of H₃O⁺ equals the concentration of the added acid.
Weak acids only partially dissociate (e.g. HF); their pH must be calculated using equilibrium expressions.
Strong Acids: 100% dissociation
Hydrochloric Acid (HCl)
Hydrobromic Acid (HBr)
Hydroiodic Acid (HI)
Upper of polyatomic series
Weak Acids: <5%
Hydrofluoric Acid (HF)
Acetic Acid (CH₃COOH)
Citric Acid (C₆H₈O₇)
Lactic Acid (C₃H₆O₃)
Formic Acid (CH O)
Carbonic Acid (H CO)
Hydrogen Cyanide (HCN)
Lower of polyatomic series
Rules
Large organic compounds are going to be weak acids
In binary acids, all the halogens are strong acids except HF
In oxyacid series, the number of oxygens determines the acid strength
Per-ic and ic acids are strong ( / )
Hypo-ous and ous acids are weak ( / )
Strong Acids
We simply assume:
[H+] = initial acid concentration
So in practice:
Strong acids are not analyzed using equilibrium calculations.
They are treated as 100% dissociated.
HCl → →
Weak Acid Calculations
The concentration of H3O+ in a weak acid is not equal to the concentration of the weak acid - < 5% dissociation.
To solve for concentration:
Set up ICE table:
Initial concentrations, equilibrium changes, and final concentrations.
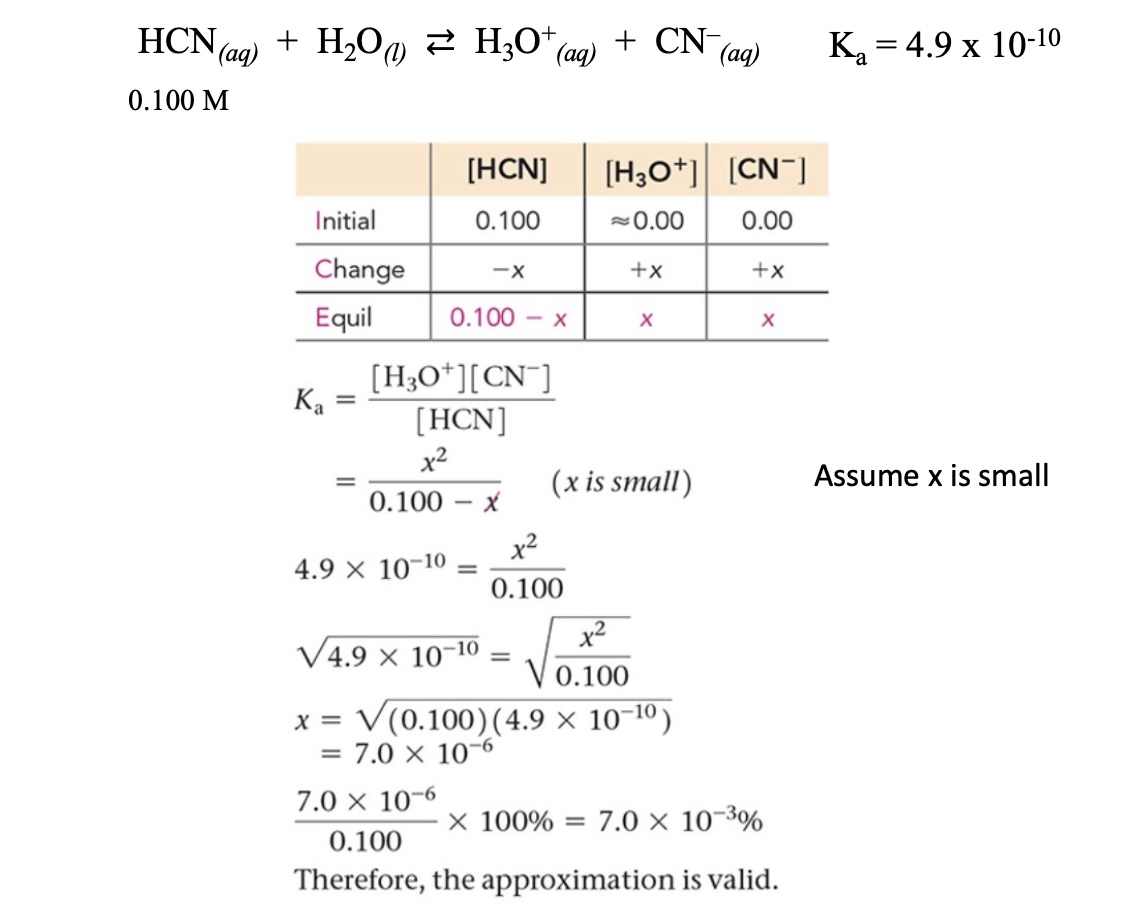
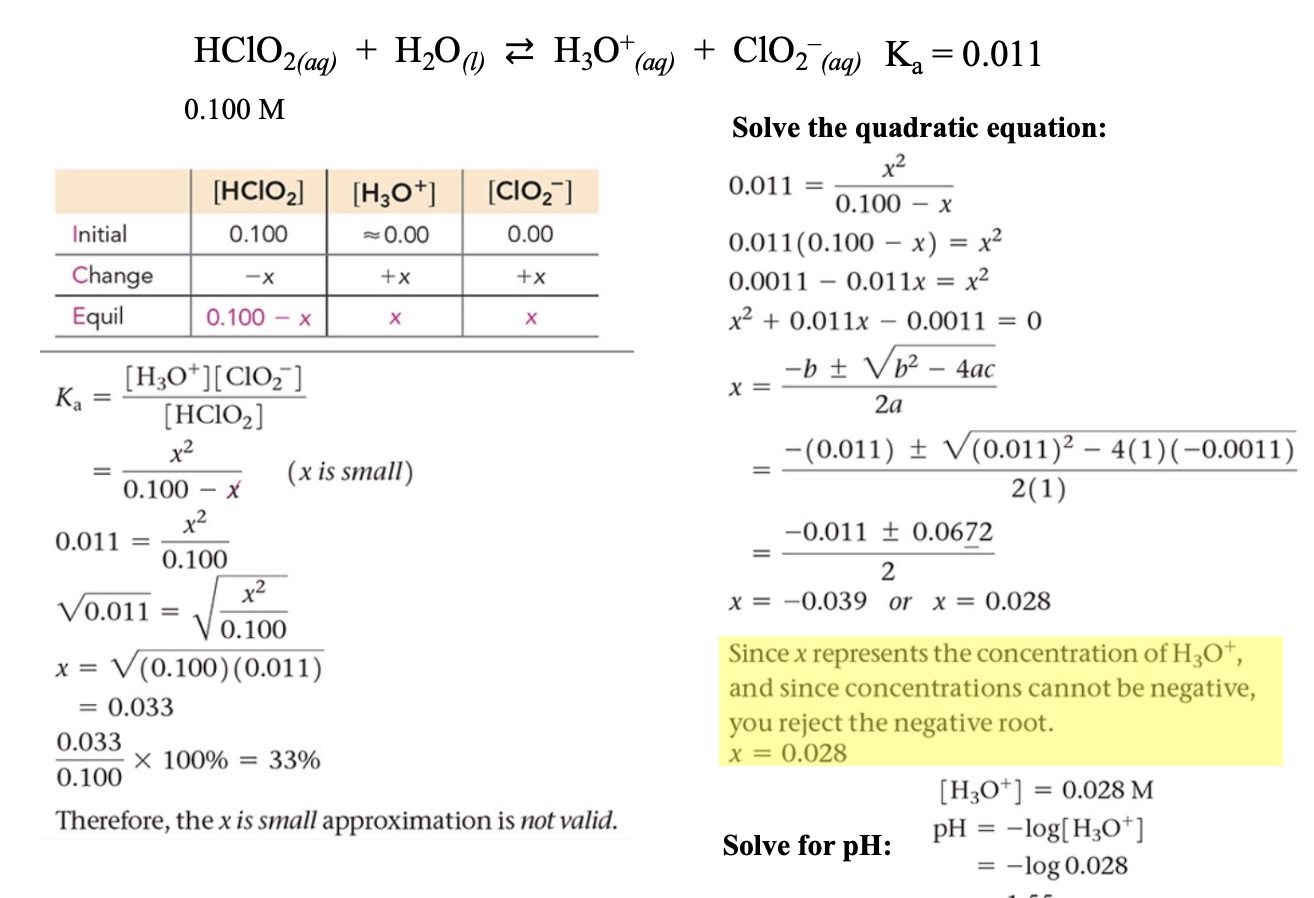
Apply the equilibrium constant expression () and solve.
Use the ‘x is small” assumption if valid
Valid if < 5%
If using the assumption that x is small is invalid, apply the quadratic formula
Solving for concentration of
,
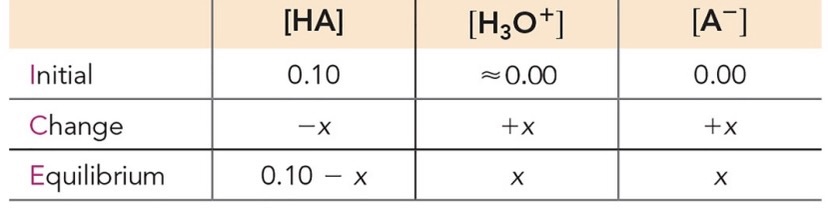
is a pure liquid and therefore not included in the constant calculation
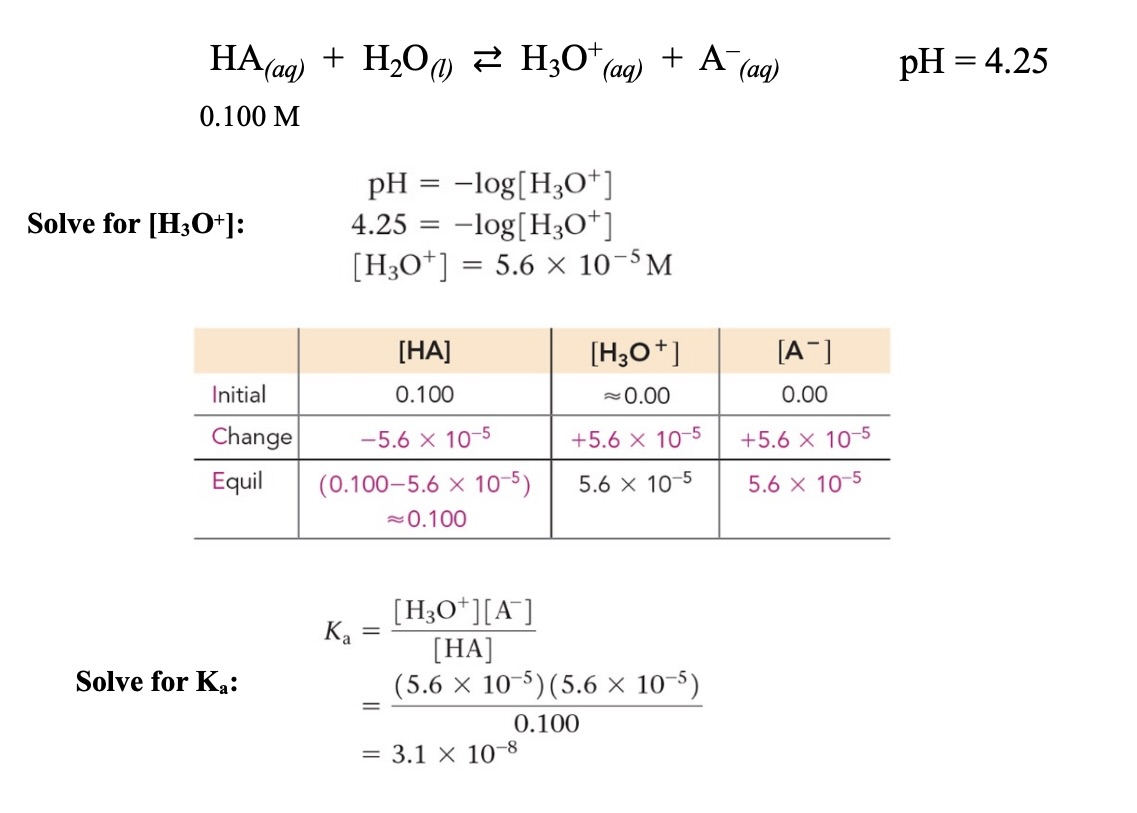
Example: Solving for the concentration of H3O+ of a weak acid solution (x is small)
Example: Solving for the concentration of H3O+ of a weak acid solution (x is not small)
Solving for from pH of a weak acid
Using given pH, the concentration of hydronium ions can be calculated
Then using an ICE table, can be calculated
Percent Ionization
Definition: The percentage of the initial weak acid that ionizes in solution:
Percent Ionization:
Where is the initial concentration of the weak acid.
Characteristics: Most weak acids ionize between 1%-5%, indicating their weak nature because only a small fraction completely dissociates into ions.
Percent ionization decreases as the acid concentration increases.
If [HA] increases (acid becomes more concentrated- removal of water) it shifts the reaction to the left and less acid ionizes
If [HA] decreases (acid becomes more dilute- adding more water) it shifts the reaction to the right and more acid ionizes
Acid Mixture: Strong Acid + Weak Acid
Three sources of H3O+:
from strong acid
from weak acid
from auto ionization of water
The formation of H3O+ by the strong acid suppresses the formation of additional H3O+ formed by the auto ionization of water and USUALLY by the weak acid. However, this needs to be checked:
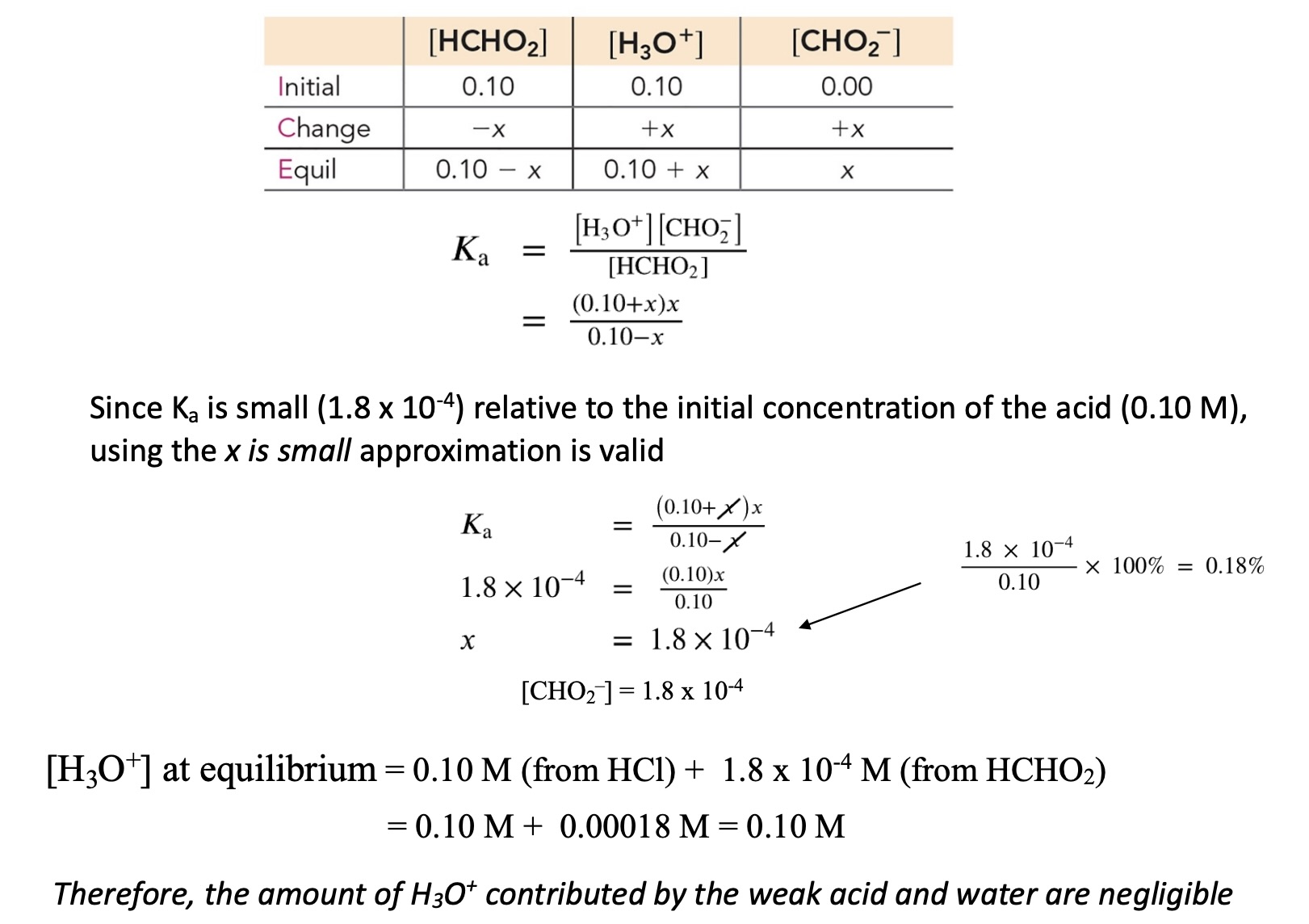
Example
0.10 M + 0.10 M (formic acid)
From auto ionization of water: [H3O+ ] ≈ 0.00 (can assume negligible)
From Strong Acid: [H3O+] = 0.10 M since HCl is a strong acid
For weak acid: need to solve for [H3O+] or [CHO2- ] (same value at equilibrium)
Weak Acid Equation:
Since the is a strong acid and already dissociated 100% of itself into , the intial is 0.10 M, along with the given initial
Acid Mixture: Weak Acid + Weak Acid
Three sources of H3O+:
from strong acid
from weak acid
from auto ionization of water
To calculate the for a mixture of two weak acids:
Compare the values and perform the standard weak acid calculation using only the weak acid with the larger
The stronger of the weak acids acts as the dominant source of ions; its dissociation creates a Common Ion Effect that suppresses BOTH the ionization of the weaker acid and water auto ionization
The contribution from the second acid becomes so small that it does not significantly change the total concentration or the final pH.
Example
0.300 M + 0.100 M
From auto ionization of water: [H3O+ ] ≈ 0.00 (can assume negligible)
Strongest weak acid HF ( )
Stronger Weak Acid Equation:
Remember pure liquids are always excluded

Strong and Weak Bases
Strong Bases: Bases that completely dissociate in solution, producing hydroxide ( ) ions directly:
Ex: 1.0 M [ ] = 1.0 M and [ ] = 1.0 M
Strong bases are typically alkali and alkaline earth metals.
Weak Bases: Do not completely dissociate; must calculate hydroxide concentration similarly to weak acids using ICE tables:
Strong bases ‘contain’ OH- and dissociate in water
Weak bases ‘produce’ OH- by accepting a proton from water to form OH-
Ex: Ammonia (base) Ammonium (conjugate acid)
Weak bases such as ammonia contain an amine group with a N with a lone pair that acts as the proton acceptor.
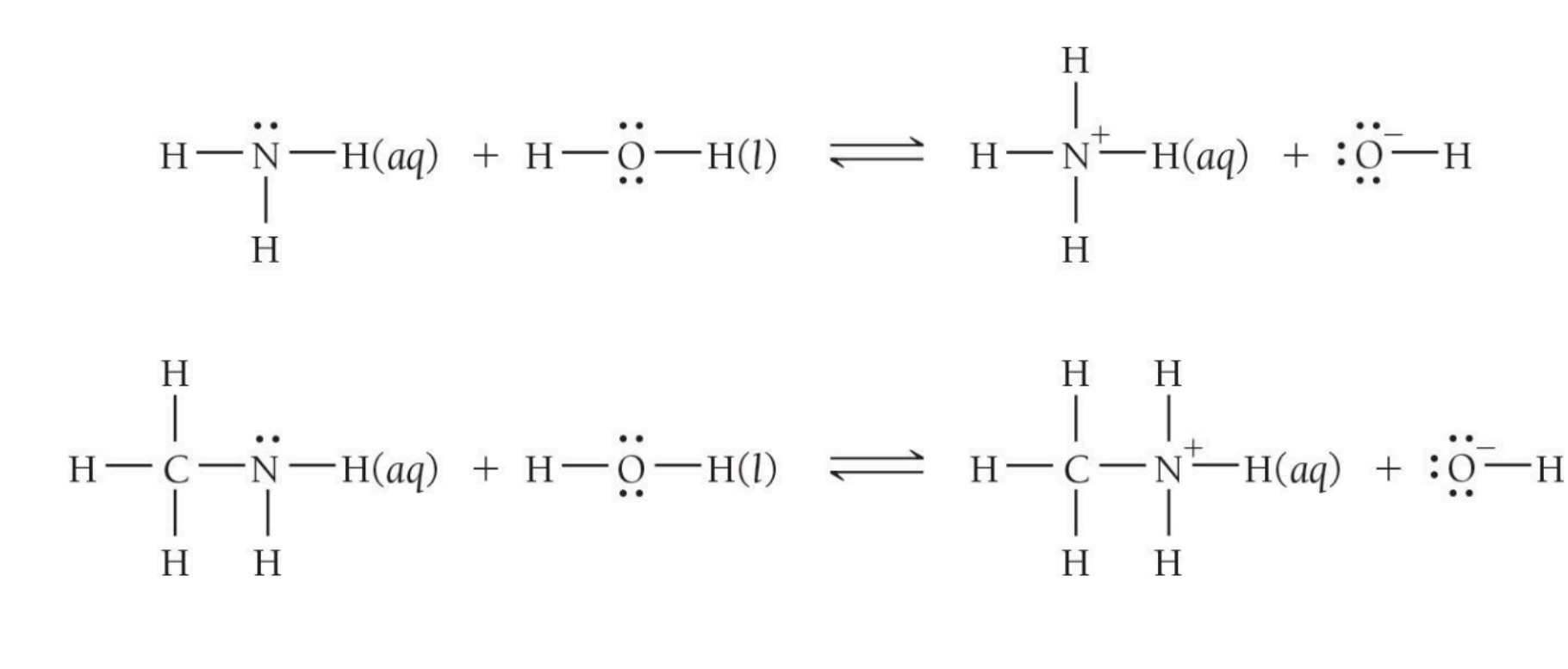
Amines are generally weak bases because the nitrogen lone pair, while available for protonation, is not extremely eager to accept a proton, especially when stabilized by nearby atoms or resonance
- Base Ionization Constant
The smaller the , the weaker the base
-log ()
Strong Base Calculation
For strong bases, the ionization is assumed to be complete, so the concentration of the base equals the concentration of the hydroxide ions produced (with stoichiometry)
To find hydronium ion concentration, we need to use the formula
Then we can use the pH formula:
pH = -log ()

Weak Base Calculation

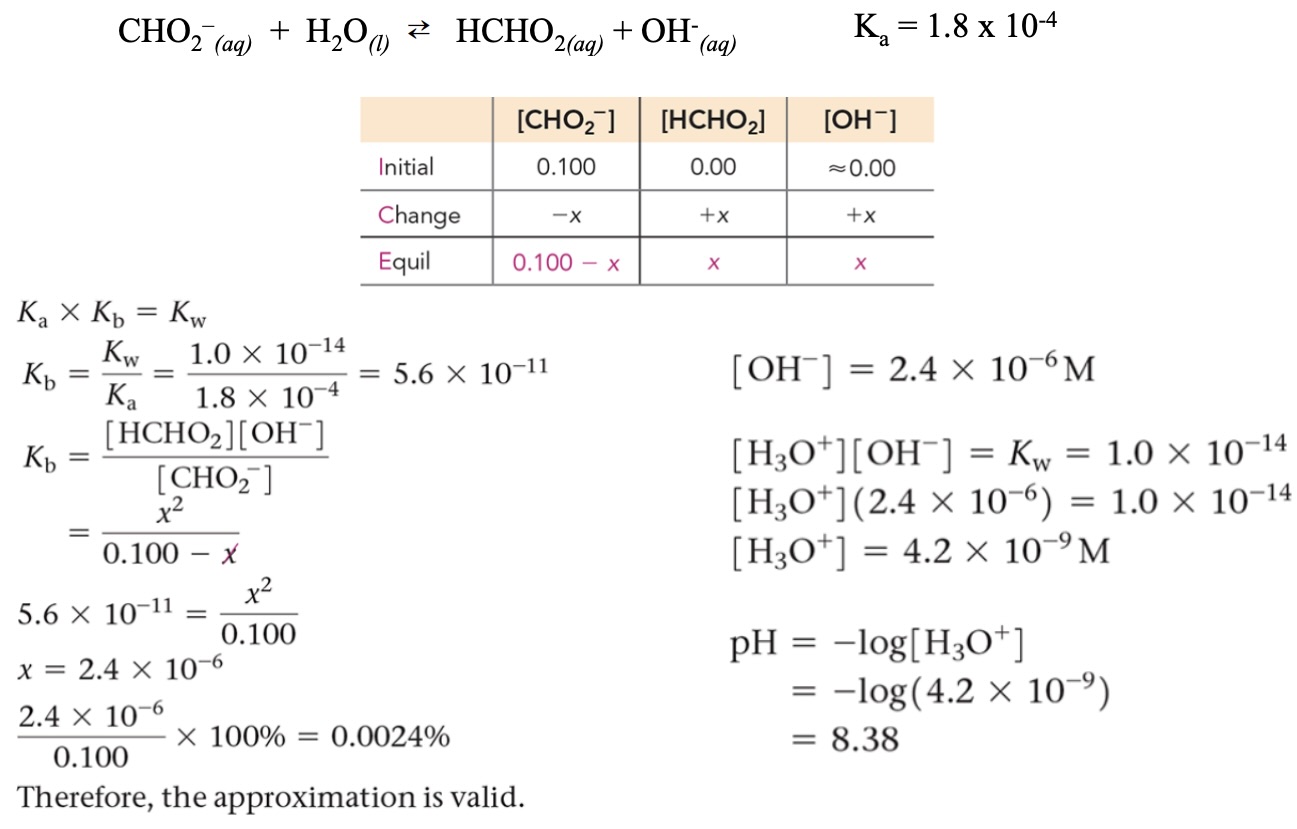
Anion Behavior (Conjugate Bases)
Conjugate bases can influence solution properties. The strength of a conjugate base depends on the strength of its parent acid:
From Strong Acids: Their conjugate bases are neutral (do not affect pH).
From Weak Acids: Their conjugate bases can act as weak bases (increase pH).
Example:
acid, base, conjugate acid, conjugate base
(conjugate base) has an affinity for ions removing from water molecules
Weaker the parent acid = stronger the conjugate base
Solving for pH of a Weak Acid Anion Solution
Find Kb:
Solve for x using ICE table concept: use approximation if valid, or quadratic if needed.
Find [H₃O⁺]:
pH = −log[H₃O⁺]
Cation Behavior (Conjugate Acids)
Conjugate acids can influence solution properties. The strength of a conjugate acid depends on the strength of its parent base:
From Strong Bases: Their conjugate bases are neutral (do not affect pH).
From Weak Bases: Their conjugate acids can act as weak acids (decrease pH).
Example:
acid, base, conjugate acid, conjugate base
(conjugate acid) donates ions to water turning it back into and producing
Weaker the parent base = stronger the conjugate acid
Cations that are small, highly charged metals form weakly acidic solutions
Cations such as and
The hydrated form of the ion acts as a Brønsted-Lowry acid:
The smaller and more highly charged the cation, the more acidic its behavior
The alkali and alkaline earth metals do not ionize water in this way
Salt Solution Behavior
Split the salt:
Salt→ Cation + Anion
Anion (conjugate base from acid)
Add H⁺ to recreate the acid:
Strong acid → neutral anion (Cl⁻, NO₃⁻)
Weak acid → basic anion (F⁻, CH₃COO⁻)
Cation (conjugate acid from base)
Try removing H⁺:
If it gives a weak base → cation is acidic
Example: NH₄⁺ → NH₃ → acidic
If no H or removal not possible:
Group 1/Group 2 metals / Ca²⁺ Sr²⁺ Ba²⁺ → neutral
Other small highly charged metals → acidic (Al³⁺, Fe³⁺)
Cation | Anion | Solution |
|---|---|---|
neutral | neutral | neutral |
acidic | neutral | acidic |
neutral | basic | basic |
acidic | basic | compare Ka (cation) vs Kb (anion) → stronger wins |
Polyprotic Acids
Definition: Acids that can donate more than one proton (eg,
First dissociation ( ) is typically stronger than subsequent dissociations ( , etc.) because the first proton separates from a neutral molecule, while the second proton separates from an anion that holds the proton more tightly
Either
The second proton contributes a negligible amount of H3O+ and is ignored
The second proton is significant and therefore you use the amount of original acid for the second reaction’s acid and resulting hydronium
Example: Sulfuric Acid
First dissociation (strong acid):
Assume complete dissociation for the first reaction
If the initial concentration is 0.0100 M, then at the end of this step:
[HSO₄⁻] = 0.0100 M
[H₃O⁺] = 0.0100 M

Acid Strength and Molecular Structure
The strength of binary acids depends on bond strengths and the polarity of the bond between hydrogen and the bonded atom.
Strong vs Weak Acid Example HF is weak - HF is polar and the positive charge is on the proton, which makes it easier for the release of
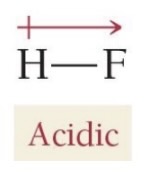
The strength of the bond affects the strength of the corresponding acid
Stronger bond = Weaker acid (harder for the H+ to be released)
Stronger electronegativity = Stronger acid
. If atom A is very electronegative, it pulls electron density away from hydrogen, making the hydrogen more positively polarized, easier for to be released
Oxyacids
Oxyacids contain a H atom bonded to an O atom. The O atom is bonded to another atom Y
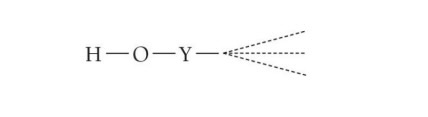
The more electronegative Y the more it weakens and polarizes the H-O bond the more acidic the oxyacid
The more O atoms bonded to Y the stronger the acid
is stronger than
is stronger than
Lewis Acids and Bases
Lewis Acids: Electron pair acceptors
Lewis Bases: Electron pair donors
accepts a proton donates an electron pair – acting as a base
donates a proton accepts an electron pair – acting as an acid
Molecules that can act as Lewis Acids/Bases include
Molecules with incomplete octets that can accept an electron pair
Molecules that can rearrange their electrons to create empty orbitals to be able to accept an electron pair
Some cations can act as Lewis acids because of their empty orbitals