Diffusion
Diffusion is the random movement of particles from an area of high concentration to an area of low concentration, until an equilibrium is reached. The difference between the high concentration of molecules in one area and the low concentration of the same molecules in another area, is known as the concentration gradient.
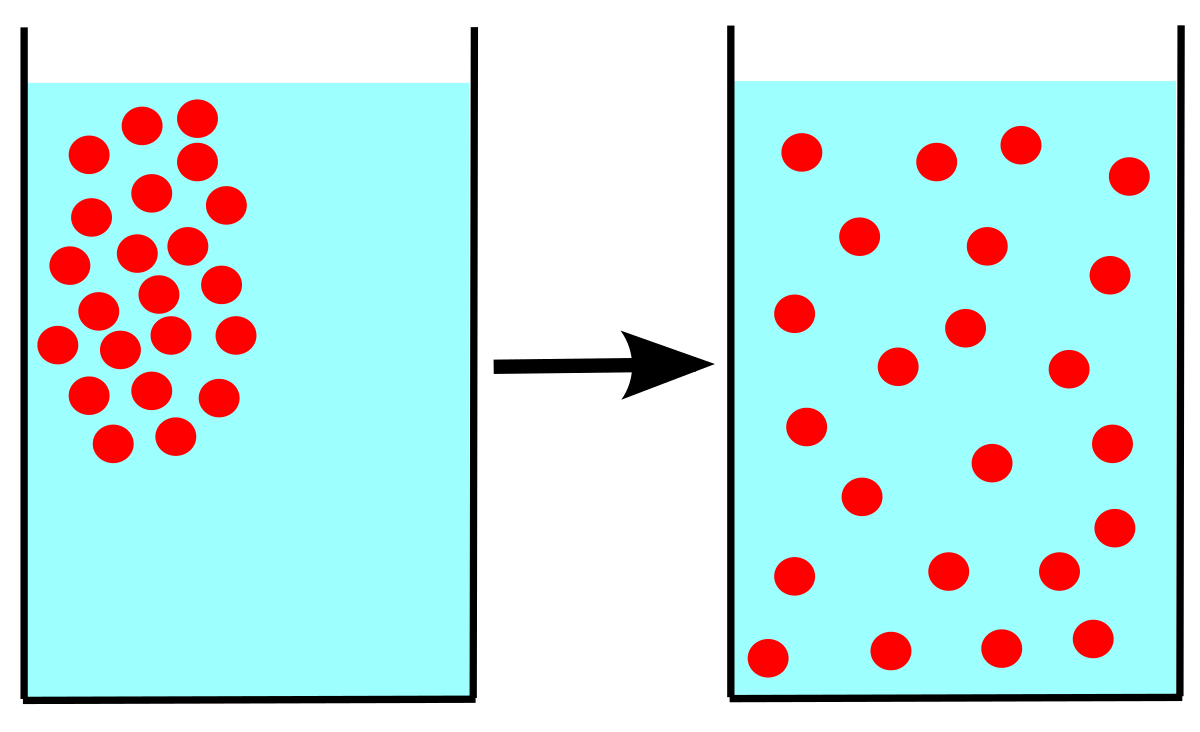
Factors Affecting the Rate of Diffusion:
1. Concentration Gradient:
The greater the difference between two areas of concentration, the steeper the gradient, and the faster diffusion will occur.
2. Temperature:
An increase in temperature will increase the rate of diffusion.
3. Pressure: An increase in pressure will also increase the rate of diffusion.
4. Energy Requirement: No kinetic energy is required for diffusion to occur when the gradient is steep, making it a passive form of transport.
Importance of Diffusion in the Body:
- Oxygen Transport: Oxygen diffuses from the lungs into the blood.
- Carbon Dioxide Excretion: Carbon dioxide diffuses from the blood to the lungs to be excreted.
- Waste Removal: Nitrogenous wastes diffuse from the blood into the kidneys to form urine.
- Nutrient Absorption: Food particles absorbed from the blood diffuse into the small intestine for digestion and absorption.