CRYSTALLINE SOLIDS
Energy - the ability to do work and heat
Work - directed energy change resulting from a process
Potential Energy -
Kinetic Energy -
UNITS OF ENERGY
joule (J)
calorie (cal)
Calorie (Cal) 1 Cal = 1000 cal = 1kcal
British Thermal Unit (BTU) 1 BTU = 1055 J
Heat - transfer of thermal energy between two bodies that are different temperature
Endothermic - heat supplied to the system by the surroundings
Exothermic - gives off heat or transfers thermal energy to the surroundings
First Law of Thermodynamics - energy cannot be created or destroyed
Specific Heat Capacity - amount of heat required to raise the temperature of 1g of a substance by 1 °C.
Formula: 𝑄=𝑚CpΔ𝑇
Calorimetry - measurement of heat flow
Calorimeter - apparatus that measures heat flow
Constant-Pressure Calorimetry
ΔH measurement
carried out in a coffee-cup calorimeter
Constant-Volume/Bomb Calorimetry
carried out in a bomb calorimeter
combustion reaction
ΔE measurement
Thermochemical Equations - represents chemical reactions and the heat involved in it consists of balanced chemical equation (with physical states) and value of ΔH (constant-pressure reaction) or ΔE (constant-volume reaction)
enthalpy is an extensive property
ELECTROCHEMISTRY
Redox Reactions - reduction and oxidation half-reactions occurring for electron transfer
Voltaic Cell - generate electricity from chemical reaction
Electrolytic Cell - consumes electricity to cause a chemical reaction
Electrochemical Cell Parts:
Circuit - where electrons flow
Anode - electrode that oxidizes
Cathode - electrode that reduces
Anode Half-Cell - where oxidation takes place
Cathode Half-Cell - where reduction takes place
Salt Bridge - keeps solutions electrically neutral
Electrical UNITS:
Electrical Charge - coulombs. C Faraday’s constant: 1mol e- = 9.65×10^4 C
Current - ampere, A 1A = 1 C/s
Electrical Energy - joule, J and kilowatt-hour, kWh 1J = 1V * C
Power - watt, W
Electrolysis - aids in separation of elements of a compound in a solution
Electroplating - technique of allowing metal ions to deposit as solids to form a coating
CRYSTALLINE SOLIDS
Solid
has definite mass, volume, and shape
intermolecular distances are short
intermolecular forces are strong
incompressible and rigid
Crystalline Solids
regular arrangement of particles
long range order
sharp melting points
anisotropy ( polarizes light)
when cut, new surfaces are plain and smooth
Examples:
diamond, graphite, NaCl, sugar
Classification of Crystalline Solids
Molecular:
orderly collection of molecules held together by van der Waals force
gases solidify only at low temperatures
easy to deform and compress
poor conductors
Covalent-Network:
3D collection of atoms bound by shared valence electrons
difficult to deform because bonds are directional
high melting point
poor electrical conductor
opaque
Ionic:
individual atoms act like closed-shell
commonly salts that are held together by the strong force of attraction between ions of opposite charge
tight packed arrangement
poor electrical conductors
hard and high melting points
Metallic:
constructed of atoms that have very weakly bounded outer electrons
large number of vacancies in orbitals
electrons aren't tightly bound to individual atoms and are free to migrate through the metal.
good conductor of electricity and heat
Amorphous Solids
random arrangement of particles
only short-range order
broad melting points
isotropic (does not polarize light)
when cut, new surfaces have an irregular surface
Examples:
coal, coke, glass, plastic, rubber
Allotropes - one or more forms of a chemical element that
occur in the same physical state.
CRYSTALLINE STRUCTURES
Crystalline lattice - atoms in a crystal are in a regular repeating
pattern (unit cell)
Simple Cubic (SC)
very inefficient and rarely seen in nature due to low packing density,
atoms touch each other along the cube edge
Body-Centered Cubic (BCC)
atoms touch each other along cube diagonals
contains 1 center atoms in contact with 8 corner atoms
Face-Centered Cubic (FCC)
atoms touch each other along face diagonals
contains 6 face atoms and 8 corner atoms
Coordination Number – the number of atoms touching a particular atom, or the number of nearest neighbors
Number of atoms in a unit cell – based on the total contribution of the atoms composing the unit cell
Atomic Radius - ½ of edge length
Atomic Packing Factor (APF) - the fraction of space occupied by atoms assuming that atoms are hard spheres
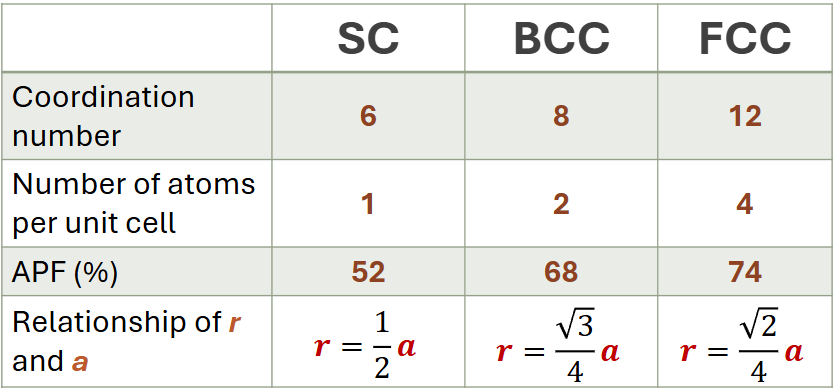
Note: density of a crystal lattice is equal to the density of its unit cell
Metals - close packing, often large atomic masses
Ceramics - less dense packing, often lighter elements
Polymers - low packing density (often amorphous)
Composite - intermediate values