3. Non-covalent interactions
Covalent bonds can be polarised due to varying electronegativities
Over short distances atoms and molecules experience attractive forces collectively known as Van der Waals interactions
Dipole-dipole
Dipole-induced dipole
Temporary dipole-induced dipole
Over shorter distances atoms and molecules repel one another.
Van der Waals interactions and steric hindrance can be described by the Lenard-Jones potential.
In order of increasing strength: dispersion forces < dipole-dipole =(~) pi-pi stacking < hydrogen bonds < ionic interactions.
Polar and non-polar molecules
A dipole moment measures a molecules’s net polarity from unequal electron sharing and its molecular geometry, resulting in a separation of positive and negative charge. It is measured in Debye and is the sum of the individual bond dipoles
A dipole can be represented using an arrow pointing towards the electronegative atom (electron rich region).
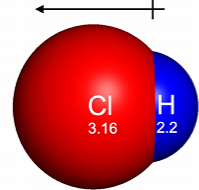
Red = electronegative (electron rich regions)
Blue = electropositive (electron poor regions)
If the molecule is symmetric, the dipoles cancel out, and the molecule is non-polar.
(London) dispersion forces
Electrons are mobile and at any point in time can be unequally distributed leading to temporary dipoles.
If atoms/molecules are close, a temporary dipole is induced in the proximal atoms/molecules (induced dipole)
These are weak interactions and attraction drops off rapidly as distance increases. They are also short-lived (transient)
These interactions occur between all atoms and molecules (polar and non-polar)
Dipoles in non-polar molecules can also be induced by permanent dipoles. (dipole-induced dipole)
Steric repulsion
Steric repulsion is a repulsive force that occurs when atoms or molecules get too close to each other, causing their electron clouds to overlap and leading to an increase in energy.
Repulsive forces increase rapidly as the distance decreases.
The minimum distance that 2 atoms can approach one another is the sum of their Van der Waals radii.
Lenard-Jones (12-6) potential
The Lenard-Jones potential plots the potential energy between 2 atoms as a function of the distance between them.
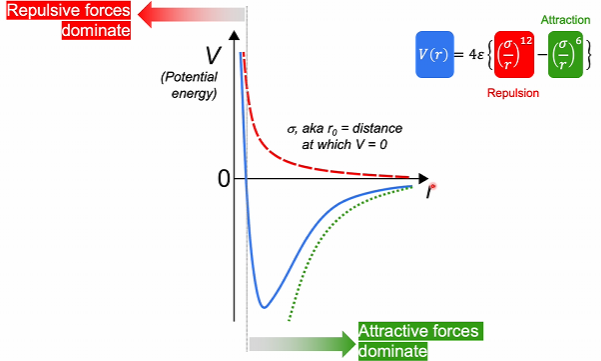
This shows the sum of the attractive forces and the repulsive forces.
Van der Waals distance is the minimum distance atoms can be apart before repulsive forces increase in energy and dominate (where V = 0).
Attractive forces display a dependence while repulsive forces display a dependence (r is distance).
- interactions
These are attractive interactions between aromatic molecules.
Aromatic molecules can arrange themselves in a face-to-face, parallel staggered, or T-shaped orientation with the T-shaped orientation being the most favourable and the face-to-face orientation being the most unfavourable.
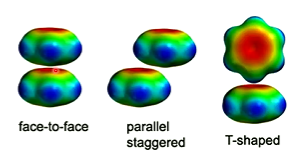
These occur in DNA base stacking and between aromatic amino acid side chains.
Hydrogen bonds
The hydrogen atom covalently bound to an electronegative atom interacts with another electronegative atom with a lone pair.
The electronegative atom can be O, N, or F, but O and N are the most biologically relevant.
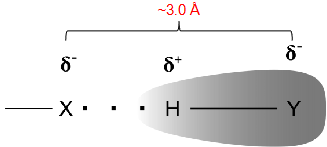
X is the H-bond acceptor (electronegative atom with a lone pair of electrons).
Y is the H-bond donor. The Y-H bond is highly polarised.
H bonds are just a stronger and special form of dipole-dipole interactions.
Heteronuclear (anything other than H) distance is very consistent (~ 3 angstroms) due to steric hindrance.
Usually linear as this is the minimum energy conformation for hydrogen bonds to form.
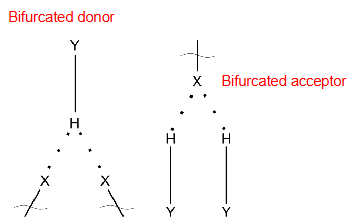
Can be bifurcated (typically seen with oxygen as it has 2 lone pairs of electrons). These 3 centre H-bonds are weaker than 2 centre H-bonds.
H-bonds have partial covalent character (around 10%)
H-bonds occur between complementary base pairs in DNA and within the polypeptide backbone to form helices and strands (secondary structure), and between amino acid side chains (tertiary structure).
Hydrophobic effect
The hydrophobic effect is not a force but a result of the hydrogen bonding in bulk water.
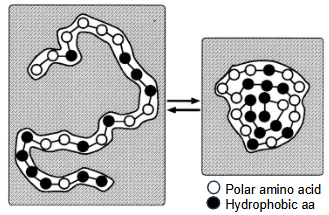
In the unfolded state, the polypeptide has increased surface area and disrupts the dynamic hydrogen bonding within bulk water as the non-polar amino acid residues are unable to form H-bonds. This decreases entropy.
In the folded state, the polypeptide has a reduced surface area and H-bonds in bulk water are not disrupted so this is entropically favourable. Polar amino acid residues can also form hydrogen bonds with water molecules. Non-polar amino acid residues can form dispersion forces with each other within the core.
Ionic interactions
Attraction between oppositely charged groups.
Ionic bonding occurs if there is a large difference in electronegativity (>1.7) between the atoms. A complete transfer of an electron from one atom to another will occur creating ions which are held together by electrostatic forces.
In reality, no pure ionic bonds exist - all ionic bonds possess some covalent character. For example, in LiF, Li’s shared 2s electron is closer to the Li nucleus in the compound than it was in elemental Li, so there must be orbital overlap and some covalent character.
Ionic interactions occur in protein tertiary and quaternary structure between oppositely charged amino acid side chains. These are called salt bridges.