The Periodic Table
What is the periodic table?
- The periodic table is the elements organised in order of
- Arranged into and
- : regular intervals
Groups
Columns going
Same group: similar
Same number of on their
8 Groups
- i:
- ii:
- iii:
- vii:
- viii:
Periods
Rows going
Labelled => N=1 to N=7
Same period: Same number of /shells
Important terms
Isotope
Atoms of the that have
Example: Carbon
Stability
Elements that have a full outer shell => very
Octet Rule
Atoms must have a full outer shell ()
Molecules
A molecule is a group of atoms joined together. It is the smallest particle of an element or compound that can exist independently
Example: Hydrogen (H2) and Water (H2O)
Chemical bond: force holding atom together
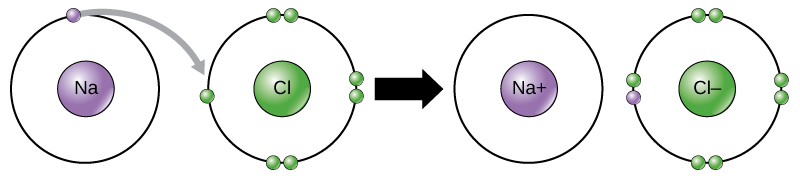
Ionic bonding
Ion: charged atom or group of atoms
Ionic bonding steal or give an electron
- If an atom gives away an electron => positively charged +
- If an atom receives/steals an atom => negatively charged -
Example: Sodium chloride => NaCl
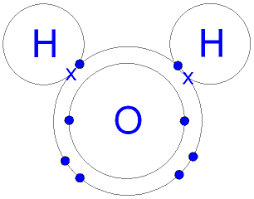
Covalent Bonding
Sharing electrons
Example: water => H2O