Order of Reactions - Experimental Results
(b) How Reaction Order is Found from Experimental Results
Initial Rate Method: The rate of reaction is measured for different initial concentrations of reactants. The order is determined by examining how the rate changes with concentration.
Half-life Method: If half-life is constant, the reaction is first-order; if it changes, other orders may be considered.
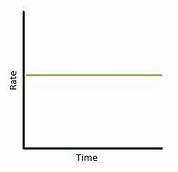
Zero order reaction
Plot of concentration vs. time gives a straight line as the concentration of this species has no impact on the rate.
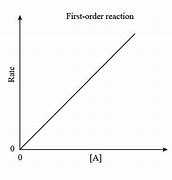
First order reaction
Plot of ln(concentration) vs. time gives a straight line as the concentration of the species and rate are directionally proportional
Doubling the conc. doubles the rate.
Second order reaction
Plot of 1/concentration vs. time gives a straight line as the rate is proportional to the conc. squared
c) General Rate Equation and How It Is Used
Rate equation: r = k[A]n[B]m
r = rate of reaction
k = rate constant
[A], [B] = concentrations of reactants
n , m = reaction orders with respect to each reactant (determined experimentally)
Usage:
Predicts how reaction rate changes with concentration.
Helps in determining reaction mechanisms.
Enables calculation of rate constants under different conditions.