25/3 Fatty Acid Catabolism, Ketone Bodies, Ethanol Metabolism & De Novo Lipogenesis – Comprehensive Lecture Notes
1. Mobilisation & Oxidation of Fatty Acids (β-Oxidation)
• Fatty acids enter cytoplasm from circulation ➜ must be “activated”
• Activation reaction (outer mitochondrial membrane/cytosol interface):
Fatty acid+CoA+ATP →Fatty acyl-CoA+AMP+PPi
• Carnitine shuttle shortens acyl-CoA, ferries it across the inner mitochondrial membrane. • Inside the matrix each β-oxidation cycle removes a two-carbon unit as acetyl-CoA and yields reduced co-enzymes:
1 FADH2 (from acyl-CoA dehydrogenase
1 NADH (from β-hydroxy-acyl-CoA dehydrogenase)
1 Acetyl-CoA ➜ feeds TCA cycle or other pathways (e.g. ketogenesis).
Electrons on FADH2/ NADH move to the electron-transport chain (detailed next semester) to drive ATP synthesis.
2. Fate of Acetyl-CoA When Glucose is Low – Ketogenesis
• Problem: The brain cannot utilise long-chain fatty acids directly & hepatic glycogen is exhausted ≈ overnight.
• Solution: Liver converts surplus acetyl-CoA ➜ ketone bodies (water-soluble):
Acetoacetate
D-β-hydroxybutyrate
Acetone (spontaneous decarboxylation by-product → nail-polish-remover breath)
• Pathway (mitochondria of hepatocytes):2 Acetyl-CoA → Acetoacetyl-CoA
+Acetyl-CoA → HMG-CoA →acetoacetate (cleaved from HMG-CoA)
→ D-beta-Hydroxybutyrate/acetone
• Export: Ketone bodies cross blood–brain barrier; peripheral tissues reconvert to acetyl-CoA via succinyl-CoA transferase ➜ enter TCA.
• Concept image: “Transportable acetyl-CoA.” Cannot circulate acetyl-CoA itself, but ketone bodies can.
ketone bodies then enter the TCA cycle
3. Real-World Tie-In – The Ketogenic Diet
• Carb-near-zero diet (high fat/protein) forces chronic ketogenesis.
• Reported uses: weight loss, glycaemic control (e.g. diabetic student anecdote).
• Side effect: acetone breath.
• Ethical note: not medical advice; individual variability is high.
4. Ethanol Metabolism & Its Metabolic Consequences
• Occurs primarily in liver cytosol/mitochondria.
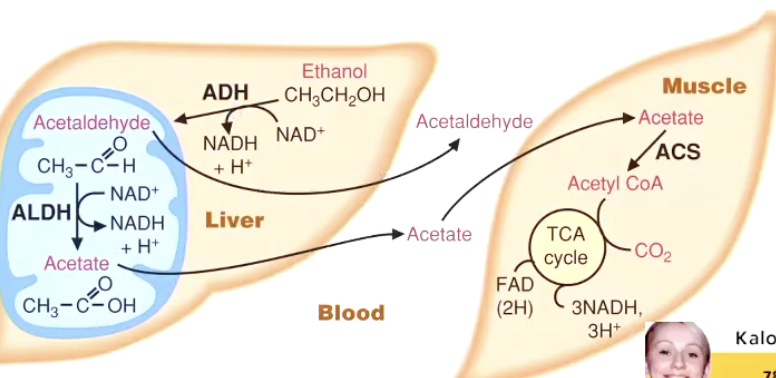
\text{Ethanol}+NAD^+ \xrightarrow{\text{Alcohol dehydrogenase}} \text{Acetaldehyde}+NADH+H^+
\text{Acetaldehyde}+NAD^+ \xrightarrow{\text{Acetaldehyde dehydrogenase}} \text{Acetate}+NADH+H^+
Acetate exits liver ➜ muscles ➜ reconverted to acetyl-CoA ➜ energy.
• Toxicity: Acetaldehyde = hangover molecule (inflammatory, nauseating).
• East Asian ALDH2 polymorphism ↓ enzyme activity ➜ flush/hangover at low intake.
• Disulfiram/coprin mushrooms inhibit ALDH ➜ used (harshly) to deter alcoholism; alcohol ingestion while inhibited → violent illness.
• Metabolic ripple effects:
• ↑ Cytosolic NADH drives \text{DHAP}\;\rightarrow\;\text{Glycerol-3-P} ➜ triacylglycerol synthesis ➜ fatty-liver.
• ↑ Mitochondrial NADH slows TCA cycle ➜ energy-storage bias.
In the Liver:
Ethanol → Acetaldehyde • Enzyme: Alcohol dehydrogenase (ADH) • Byproduct: NAD⁺ → NADH + H⁺
Acetaldehyde → Acetate • Enzyme: Aldehyde dehydrogenase (ALDH) • Byproduct: NAD⁺ → NADH + H⁺
Transport Phase:
Acetate is released into the bloodstream and taken up by muscle tissue
In the Muscle:
Acetate → Acetyl-CoA • Enzyme: Acetyl-CoA synthetase (ACS)
Acetyl-CoA enters the TCA cycle • Produces: CO₂, NADH, FADH₂
NADH and FADH₂ feed into oxidative phosphorylation • Produces: ATP (energy)
no ALDH from coprine (mushroom) = can’t detoxify ethanol/alcohol
5. Re-Synthesis & Storage of Triacylglycerol (TAG) in Adipose & Liver
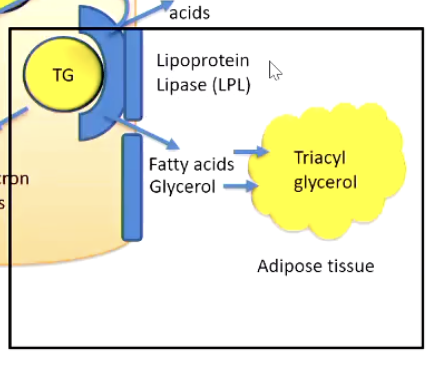
Steps
Fat arrives via chylomicrons (from diet) or VLDL (from liver).
Lipoprotein lipase at capillaries breaks down TAGs:
Releases fatty acids and glycerol into tissues.
Glycerol-3-phosphate (G3P) is formed:
In adipose: from DHAP (glycolysis).
In liver: from free glycerol.
TAG is rebuilt in three steps:
Add 2 fatty acyl-CoA → makes phosphatidic acid.
dephosphorylate/Remove phosphate → makes diacylglycerol (DAG).
Add 3rd fatty acyl-CoA → forms triacylglycerol (TAG).
Outcome:
Adipose tissue: TAG stored in lipid droplets.
Liver: TAG packed into VLDL → sent to bloodstream → delivered to other tissues.
6. De Novo Fatty Acid Synthesis (Lipogenesis)
• purpose: converts excess dietry carbs/protein → FA
• Location & Compartmentalisation:
• Cytosol of hepatocytes (liver cell) (minor in adipose, lactating mammary glands).
• Spatial separation from β-oxidation (mitochondrial) avoids futile cycling.
• Overview of 4 key steps:
acetyl-coA into cytoplasm with citrate
generate NADPH in cytoplasm
Acetyl-coA turn into Malonyl-CoA
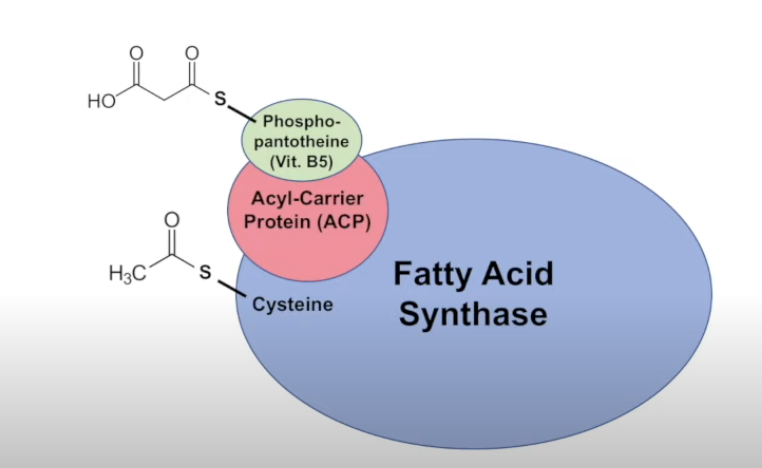
FA synthase (protein complex)
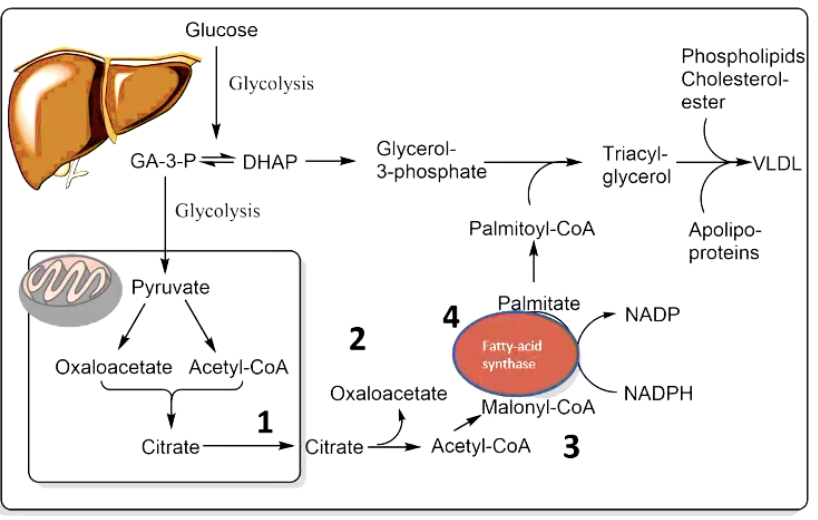
1st step: getting Acetyl-CoA into the cytosol
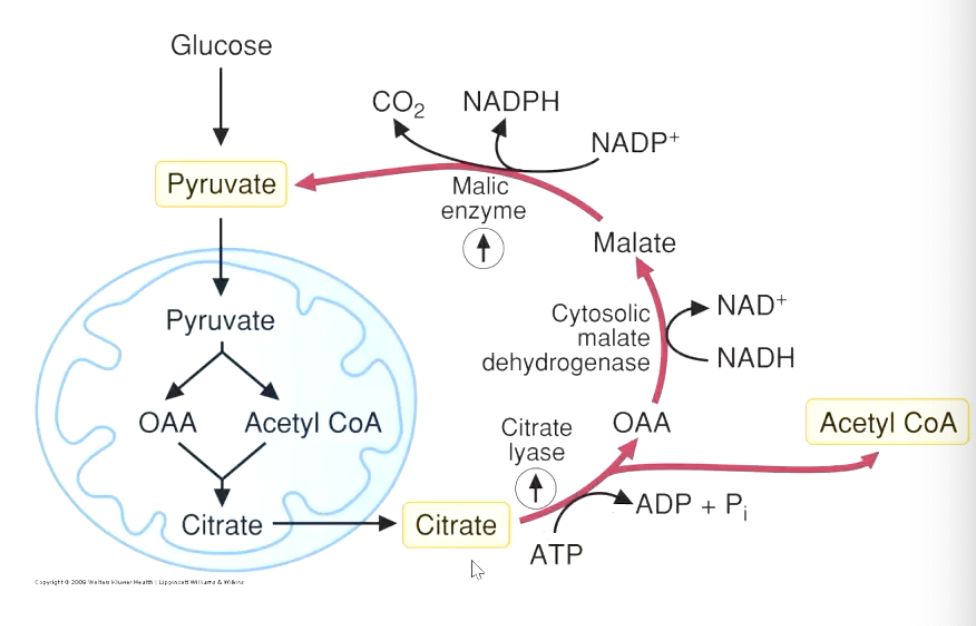
citrate is exported out of the mitochondria and into the cytosol
can be converted back into oxalacetate OAA (requires ATP) (citrate lyase byproduct)
Acetyl-CoA accumulates as a result
OAA converts into malate (enzyme Cytosolic malate dehydrogenase, byproduct: NAD+)
malate converts back to pyruvate (NADPH and CO2 byproduct from NADP+)
2nd Step: NADPH in the cytosol
NADPH can also come from the pentose-phosphate pathway
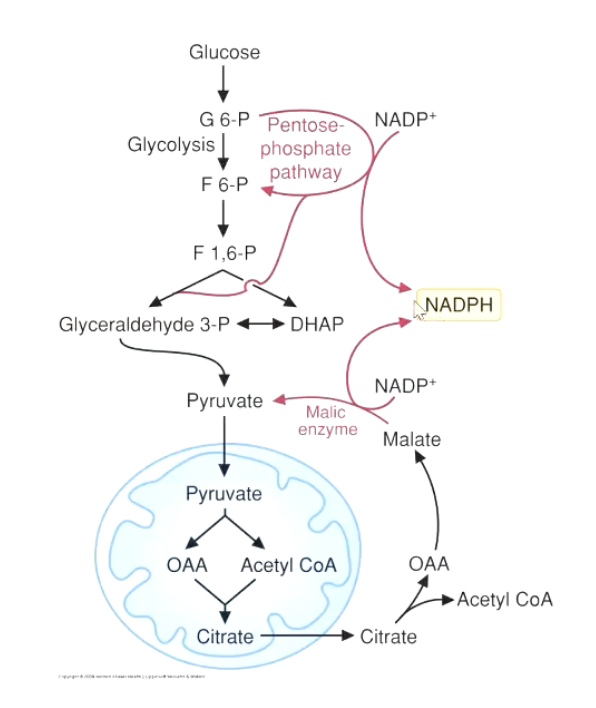
NADPH generated from Malate→pyruvate step
3rd Step: Generation of Malonyl-CoA from Acetyl-CoA
acetyl-CoA generated from citrate→OAA step
biotin (vitamin) required to coordinate CO2
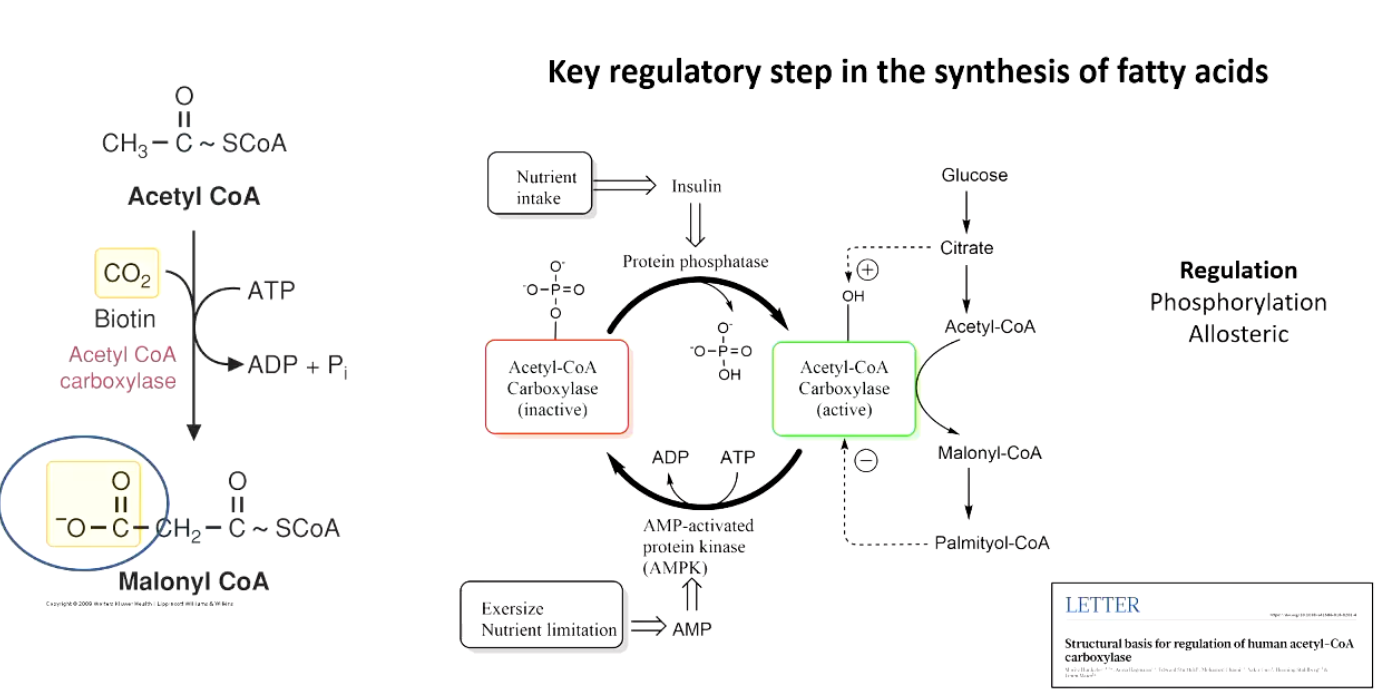
(still 3rd step) Regulation of ACC (Committed Step)
• Phosphorylation-state control (phosphorylation allosteric regulation):
• Insulin ➜ activates protein phosphatase ➜ dephosphorylated ACC = ON.
• AMP (energy stress) ➜ AMP-activated protein kinase (AMPK) phosphorylates ACC = OFF.
• Allosteric control:
• citrate activates ACC (turns it on), palmitoyl-coa inhibits ACC (turns it off)
insulin presence =ACC on = acetyl coa turn into malonyl coa
4th Step: FA synthase
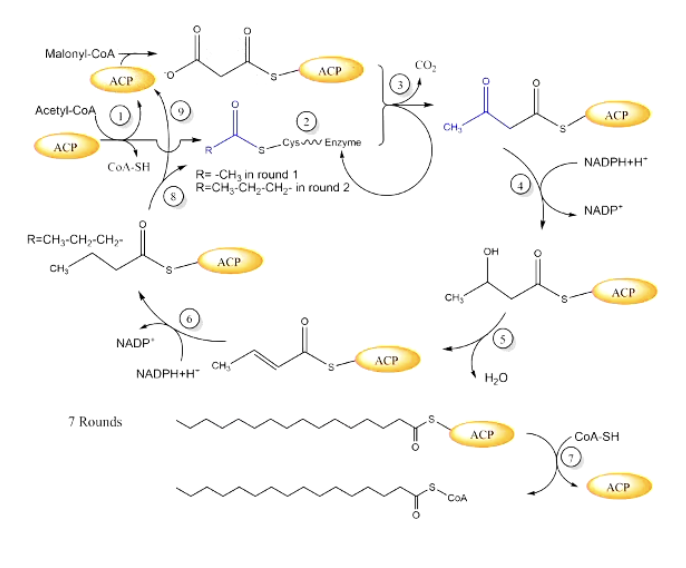
step-by-step: FA synthase cycle
loading the enzyme
acetyl coa binds to ACP (coa is removed)
acetyl then binds to cys, and malonyl coa binds to ACP
condensation rctn
malonyl coa +acetyl coa → acetoacetyl-ACP
releases CO2 and extends the chain by 2 C atoms
(adds more malonyl coa each round which releases more CO2)
reverse beta oxidation-like modifications (making the chain more saturated like a saturated FA)
reduction 1: use NADPH to reduce ketone
dehydration: remove water
reduction 2: uses NADPH again to form a fully saturated bond
chain transfer and restart
the elongated chain is transferred from ACP back to the cysteine site
new malonyl coa molec binds to ACP
repeat the cycle
this process repeats 7 times, each cycle adds 2 C
after 7 rounds = total 16 Carbons
final release
the 16C FA is released from ACP
it’s reactivated into palmitoyl coa, ready for storage
🧬 Overall Product:
Palmitate (C16:0) — the first fully synthesized fatty acid
Requires:
8 Acetyl-CoA, 7 ATP, 14 NADPH for one palmitate
7. Packaging & Distribution of Newly Synthesised Fat
• Palmitoyl-CoA + glycerol-3-P ➜ TAG ➜ assembled into very-low-density lipoproteins (VLDL).
• VLDL exported from liver, hydrolysed by endothelial lipoprotein lipase ➜ FA uptake by muscle (oxidation) or adipose (storage).
8. Integrated Regulation & Physiological Context
• Fed state (high insulin, high glucose):
• Glycolysis/TCA supply cytosolic citrate ➜ ACC ON ➜ FAS active ➜ TAG synthesis & storage.
• Fasting/exercise (high glucagon/epinephrine, ↑ AMP):
• PKA & AMPK phosphorylate ACC (OFF) ➜ malonyl-CoA ↓.
• Low malonyl-CoA lifts inhibition on carnitine acyl-transferase I ➜ β-oxidation ↑.
• Ethanol ingestion distorts this balance via NADH surplus, promoting lipogenesis and fatty-liver.
9. Key Equations & Numbers to Remember
• Activation of FA: ATP\;→\;AMP (cost = 2 ~Pi bonds).
• ACC reaction: attaches CO$2$ (biotin-dependent). • FA synthesis to palmitate consumes 14 NADPH & 7 ATP. • β-oxidation: each cycle → 1 FADH$2$ (≈1.5 ATP) + 1 NADH (≈2.5 ATP) + 1 acetyl-CoA (10 ATP via TCA/ETC under aerobic conditions).
10. Take-Home Summary
• Fatty acids must first be converted to acyl-CoA, carnitine-shuttled, then oxidised in mitochondria.
• Surplus acetyl-CoA in liver mitochondria is diverted to ketone bodies that can nourish the brain during prolonged fasting or ketogenic diets.
• Ethanol metabolism yields acetate for energy but generates toxic acetaldehyde and excess NADH, predisposing to fatty-liver.
• TAG storage in adipose involves re-esterification of imported fatty acids with glycerol-3-P.
• De novo lipogenesis occurs in the cytosol, relies on citrate export, NADPH supply, and is governed by the highly regulated enzyme ACC followed by the multi-enzymatic FAS.
• Hormonal (insulin vs. glucagon/epinephrine), energy (AMP/ATP), and allosteric cues (citrate, palmitoyl-CoA) tightly coordinate the switch between fat synthesis and oxidation, ensuring metabolic flexibility.
VLDL delivers TAG from liver (also produced from liver)
when fed state → distributes new fats
releases FA when hydrolysed by LPL in capillary beds
muscle: burns FA for energy
adipose tissue: re-esterfies FA into stored TAGs
LDL delivers cholesterol to tissues
for cellular growth and hormone synth
utilised when cells need cholesterol for:
memb synth
steroid hormone prod
bile acid synth
Key Concepts for Exam Study
To effectively study these notes for an exam, focus on the following key concepts:
Fatty Acid Mobilisation & Oxidation (β-Oxidation):
Fatty acids are "activated" to fatty acyl-CoA in the cytoplasm (\text{Fatty acid}+\text{CoA}+ATP \;
rightarrow\; \text{Fatty acyl-CoA}+AMP+PP*i).The carnitine shuttle ferries fatty acyl-CoA into the mitochondrial matrix.
Each β-oxidation cycle removes a two-carbon unit as acetyl-CoA and yields 1\,FADH*2 and 1\,NADH. These reduced co-enzymes feed into the electron transport chain (ETC) for ATP synthesis.
Acetyl-CoA feeds into the TCA cycle or other pathways like ketogenesis.
Ketogenesis (When Glucose is Low):
Occurs in liver mitochondria when the brain needs an alternative energy source (e.g., during prolonged fasting or a ketogenic diet), as the brain cannot directly use long-chain fatty acids.
Surplus acetyl-CoA is converted into ketone bodies: Acetoacetate, D-β-hydroxybutyrate, and Acetone.
Ketone bodies act as "transportable acetyl-CoA" for peripheral tissues, crossing the blood-brain barrier and being reconverted to acetyl-CoA for the TCA cycle.
Ethanol Metabolism:
Primarily in the liver, ethanol is converted to acetaldehyde (toxic, causes hangover) by alcohol dehydrogenase (\text{Ethanol}+NAD^+ \xrightarrow{\text{Alcohol dehydrogenase}} \text{Acetaldehyde}+NADH+H^+).
Acetaldehyde is then converted to acetate by acetaldehyde dehydrogenase (\text{Acetaldehyde}+NAD^+ \xrightarrow{\text{Acetaldehyde dehydrogenase}} \text{Acetate}+NADH+H^+), which can be used for energy in muscles.
Key metabolic consequence: Excess NADH promotes triacylglycerol synthesis (\text{DHAP}\;\rightarrow\;\text{Glycerol-3-P}) leading to fatty liver, and slows the TCA cycle.
Triacylglycerol (TAG) Re-Synthesis & Storage:
Dietary fatty acids released from chylomicrons/VLDL are re-esterified with glycerol-3-P to form TAG.
TAG is stored in adipose tissue (lipid droplets) or packaged into VLDL particles by the liver for distribution to peripheral tissues.
De Novo Fatty Acid Synthesis (Lipogenesis):
Occurs in the cytosol of hepatocytes (and other tissues) when there's an excess of dietary carbohydrate/protein.
Key steps:
Export acetyl-CoA from mitochondria as citrate via the citrate shuttle.
Generate NADPH in the cytosol (pentose phosphate pathway and malic enzyme).
Committed/Rate-Limiting Step: Acetyl-CoA Carboxylase (ACC) converts acetyl-CoA to malonyl-CoA (\text{Acetyl-CoA}+CO2+ATP \xrightarrow[biotin]{ACC} \text{Malonyl-CoA}+ADP+Pi).
Fatty Acid Synthase (FAS) complex elongates the chain to 16-carbon palmitate, using malonyl-CoA and NADPH.
Regulation of ACC:
Insulin activates (dephosphorylation).
AMP (energy stress) and AMPK inhibit (phosphorylation).
Citrate is an allosteric activator.
Palmitoyl-CoA (product) is a feedback inhibitor.
Integrated Regulation:
Fed State (High Insulin): Glycolysis/TCA supply citrate, ACC is ON, FAS active, leading to TAG synthesis and storage.
Fasting/Exercise (High Glucagon/Epinephrine, ↑ AMP): ACC is OFF (phosphorylated by PKA/AMPK), malonyl-CoA decreases, lifting inhibition on carnitine acyl-transferase I to promote β-oxidation.
Ethanol ingestion disrupts this balance, promoting lipogenesis due to excess NADH.