Chapter 20
The Dark Reactions and the Pentose Phosphate Pathway
light reactions = transform light energy into ATP and biosynthetic reducing power, NADPH
dark reactions = use the ATP and NADPH produced by the light reactions to reduce carbon atoms from CO2 to a more reduced state as a hexose
called the Calvin–Benson cycle
the pentose phosphate pathway = pathway that oxidizes glucose to generate NADPH
mirror image of the Calvin–Benson cycle in many ways
Section 20.3 The Pentose Phosphate Pathway Generates NADPH and Synthesizes Pentoses
pentose phosphate pathway = pathway generating a crucial source of NADPH for use in reductive biosynthesis and for protection against oxidative stress
occurs in the cytoplasm of all organisms
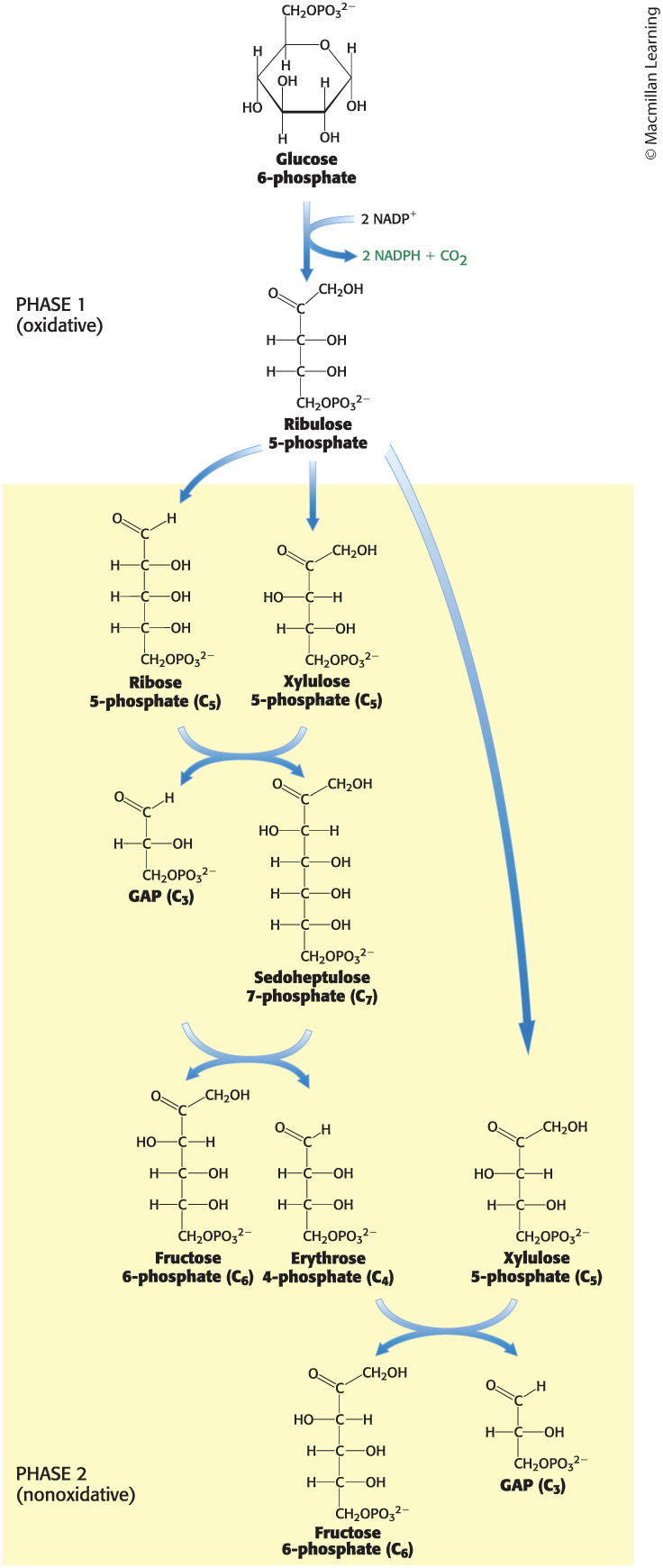
The Pentose Phosphate Pathway Consists of Two Phases
The first phase is the oxidative generation of NADPH.
occurs when glucose 6-phosphate is oxidized to ribulose 5-phosphate
Glucose 6-phosphate + 2 NADP+ + H2O → ribulose 5-phosphate + 2 NADPH + 2 H+ + CO2
The second phase is the nonoxidative interconversion of three-, four-, five-, six-, and seven-carbon sugars.
Interconversions rely on the same reactions leading to regeneration of ribulose 1,5-bisphosphate in the Calvin–Benson cycle.
Pathways Requiring NADPH
TABLE 20.2 Pathways requiring NADPH

The Pentose Phosphate Pathway
Glucose 6-phosphate
| NADP+
| NADPH + CO2
V
Ribulose 5-phosphate
| | |
| | |
V V V
HHH
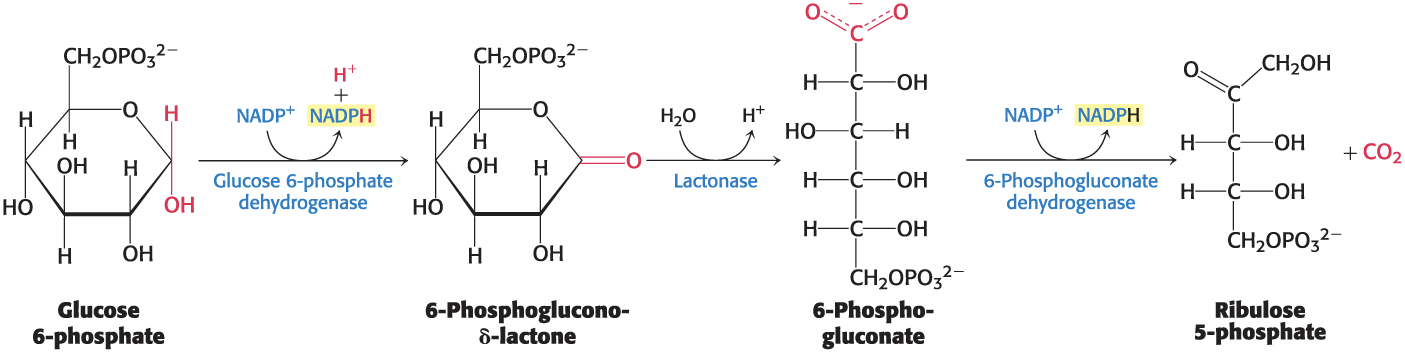
Two Molecules of NADPH Are Generated in the Conversion of Glucose 6-Phosphate into Ribulose 5-Phosphate
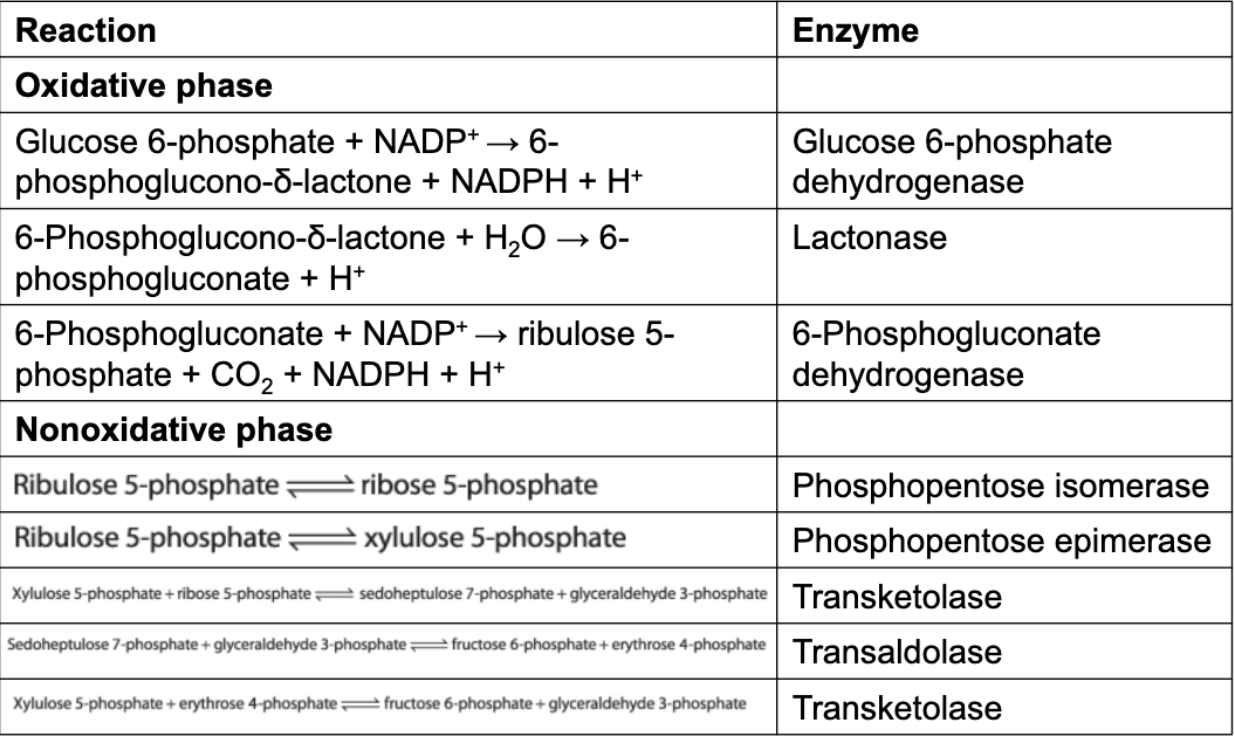
glucose 6-phosphate dehydrogenase = initiates the oxidative phase of the pentose phosphate pathway with the conversion of glucose 6-phosphate into 6-phosphoglucono-δ-lactone, yielding NADPH and H+
highly specific for NADP+
lactonase = hydrolyzes 6-phosphoglucono-δ-lactone to 6-phosphogluconate
6-phosphogluconate dehydrogenase = oxidatively decarboxylates 6-phosphogluconate to ribulose phosphate, yielding CO2 and NADPH
NADP+ is the electron acceptor
Oxidative Phase of the Pentose Phosphate Pathway Yields Two Molecules of NADPH
The Pentose Phosphate Pathway and Glycolysis Are Linked by Transketolase and Transaldolase
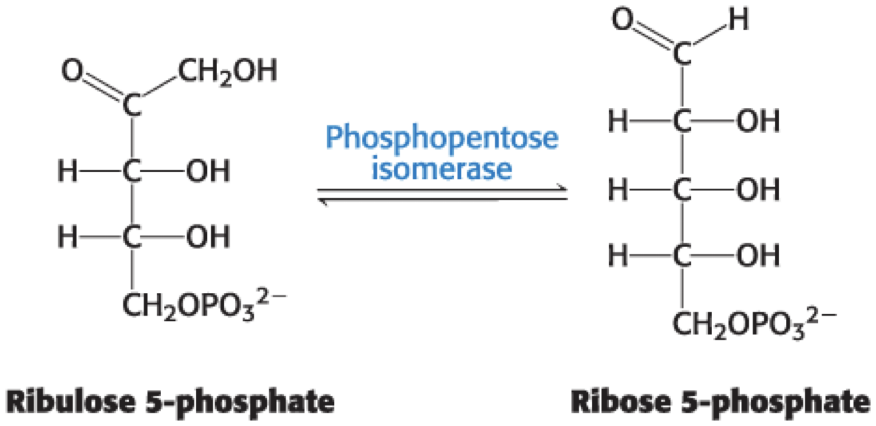
Phosphopentose isomerase isomerizes ribulose 5-phosphate into ribose 5-phosphate.
The Pentose Phosphate Pathway and Glycolysis Are Reversibly Linked
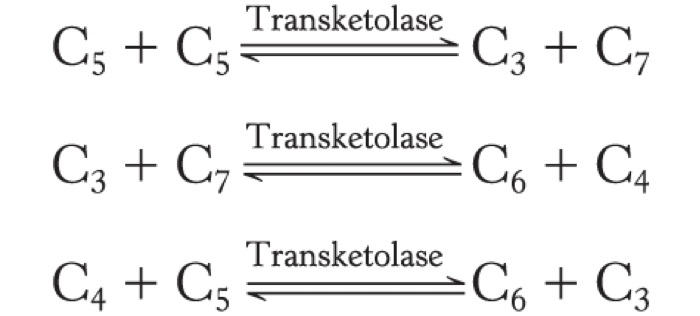
Transketolase and transaldolase catalyze three successive reactions:
 2nd one is actually a transaldolase
2nd one is actually a transaldolase
hdifferent mechanisms
The net result of these reactions is the conversion of three pentoses into two hexoses and one triose:
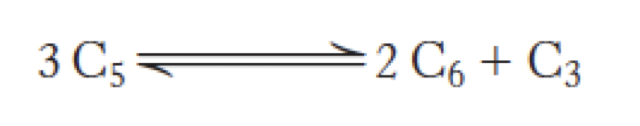
The Formation of Glyceraldehyde 3-Phosphate and Sedoheptulose 7-Phosphate
Transketolase catalyzes the formation of glyceraldehyde 3-phosphate and sedoheptulose 7-phosphate from two pentoses.
first of three linking reactions
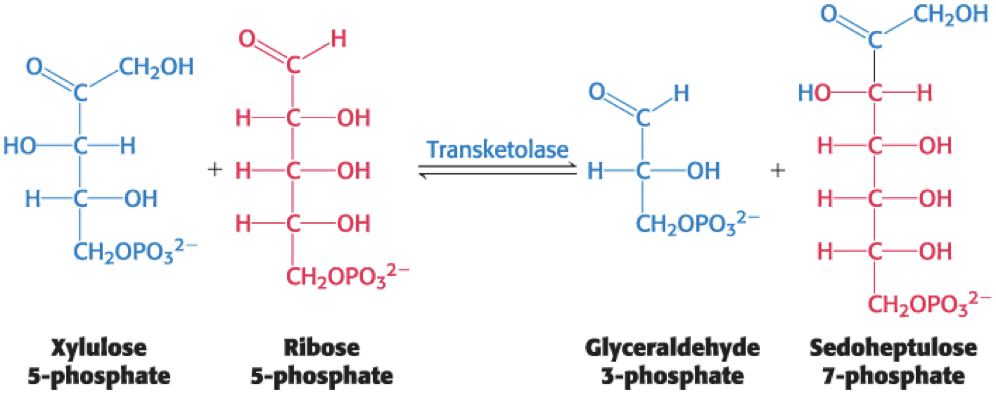
The Phosphopentose Epimerase Reaction
The transketolase substrate is a ketose with its hydroxyl group at C-3 having the configuration of xylulose rather than ribulose.
phosphopentose epimerase = catalyzes the conversion of xylulose 5-phosphate into its epimer ribulose 5-phosphate
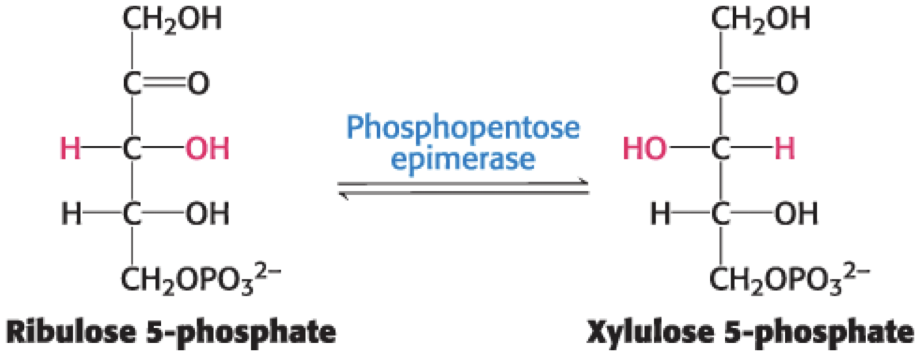
The Formation of Fructose 6-Phosphate and Erythrose 4-Phosphate
Transaldolase catalyzes the formation of a six-carbon and a four-carbon sugar from a three-carbon and a seven-carbon sugar.
second of three linking reactions
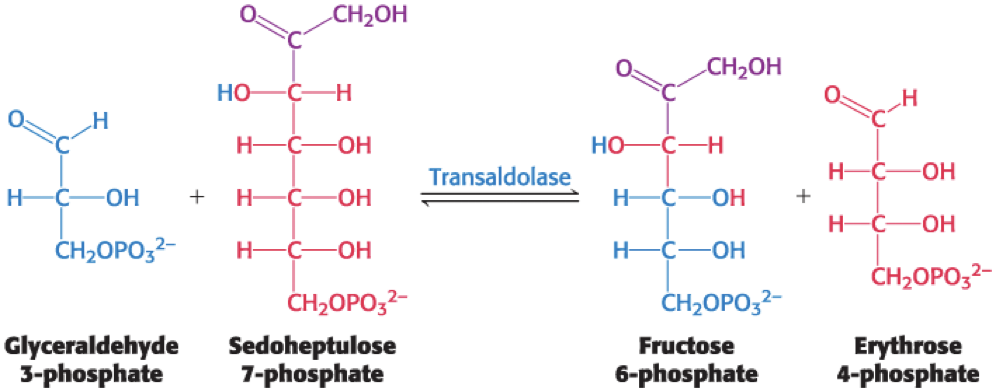
The Formation of Fructose 6-Phosphate and Glyceraldehyde 3-Phosphate
Transketolase catalyzes the formation of a six-carbon and a three-carbon sugar from a four-carbon and a five-carbon sugar.
third of three linking reactions
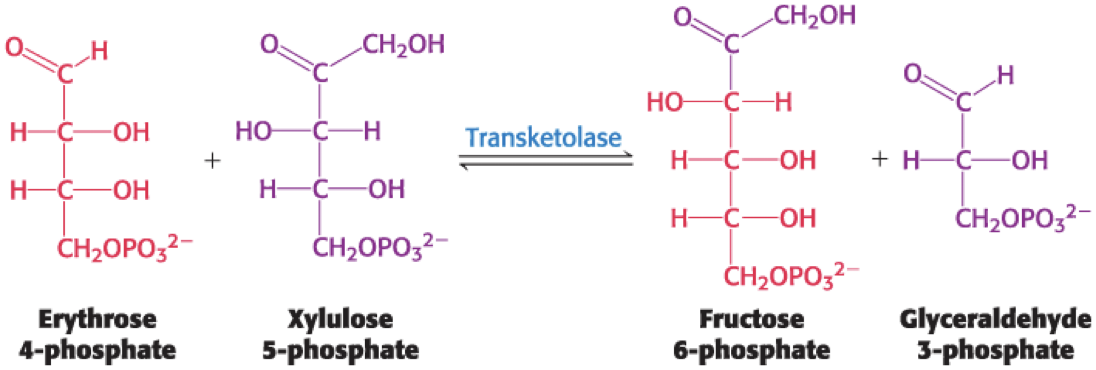
The Sum of the Transketolase, Epimerase, and Transaldolase Reactions
The sum of the reactions catalyzed by the transketolase, phosphopentose epimerase, and transaldolase is
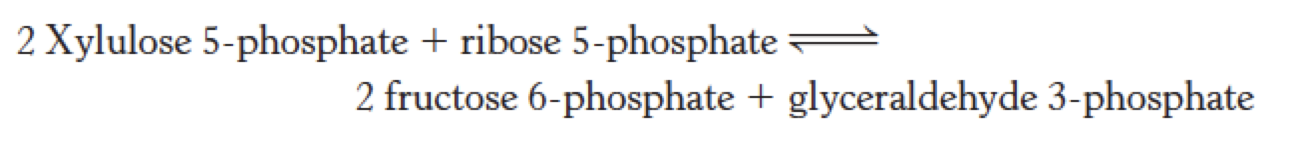 Xylulose 5-phosphate is formed by ribulose
Xylulose 5-phosphate is formed by ribulose
Xylulose 5-phosphate can be formed from ribulose 5-phosphate by phosphopentose isomerase and phosphopentose epimerase, so the net reaction starting from ribose 5-phosphate is
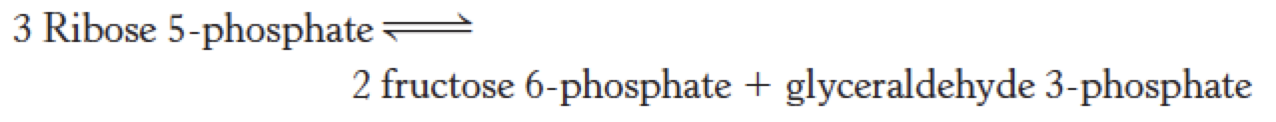
Pentose Phosphate Pathway
TABLE 20.3 Pentose phosphate pathway
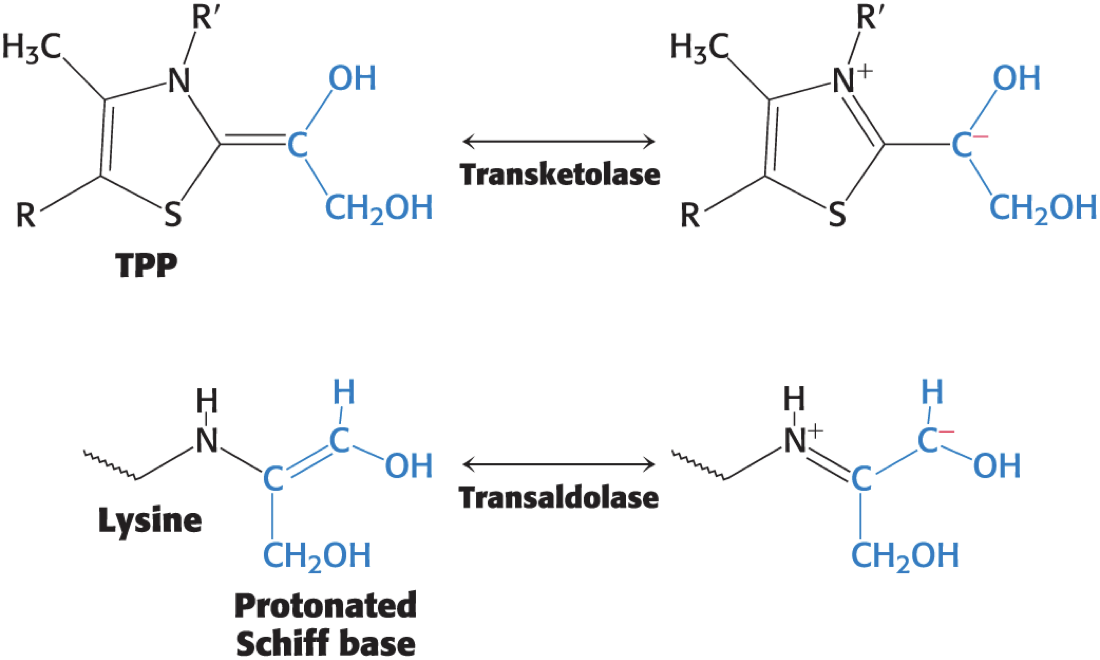
Transketolase and Transaldolase Stabilize Carbanionic Intermediates by Different Mechanisms
Transketolase transfers a two-carbon unit and contains a tightly bound thiamine pyrophosphate (TPP) as its prosthetic group.
Transaldolase transfers a three-carbon unit and does not contain a prosthetic group.
Each of these carbon units is transiently attached to the enzyme in the course of the reaction.
characteristic of double displacement reactions
The Transketolase Reaction Takes Place on Thiamin Pyrophosphate
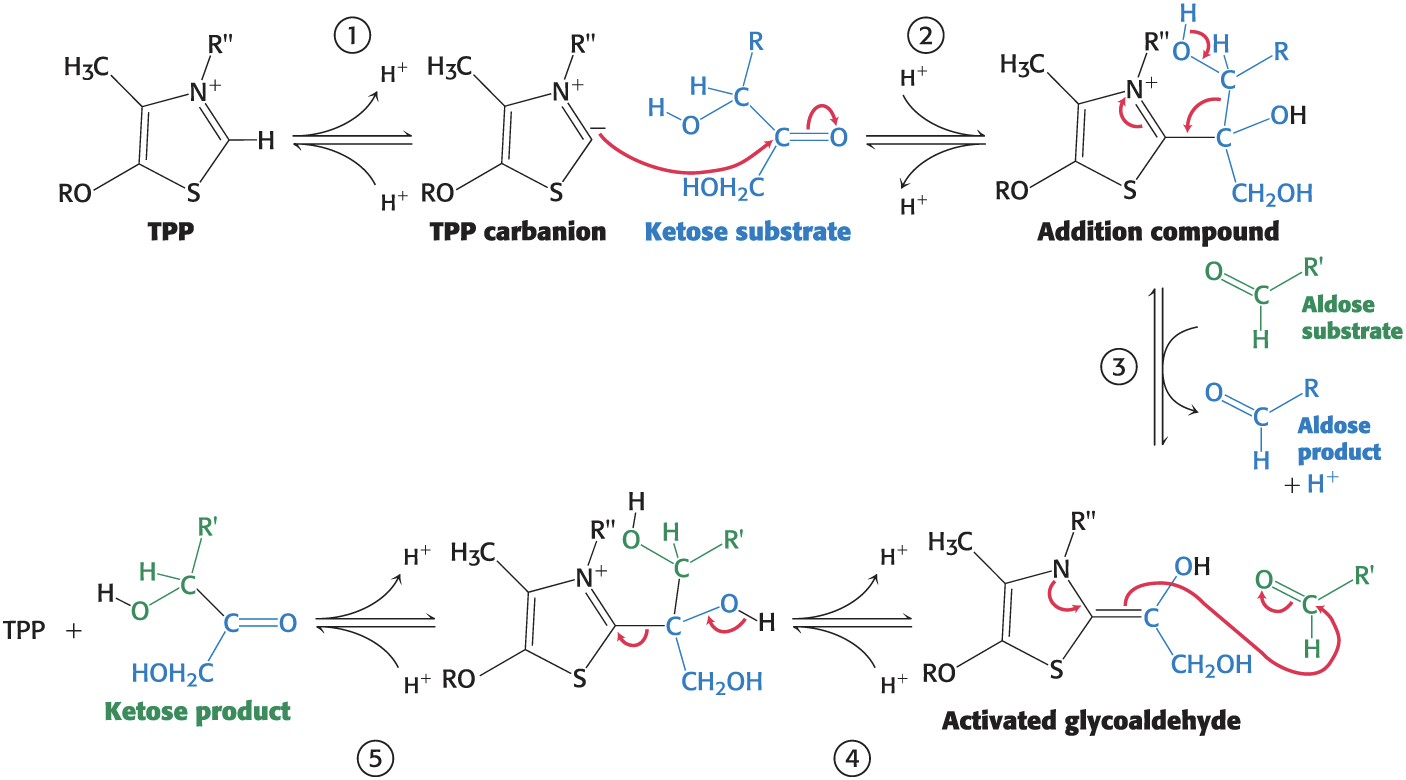
Transketolase Reaction
Step 1: The C-2 carbon of bound TTP ionizes to give a carbanion.
Step 2: The carbanion of TPP attacks the carbonyl group of the ketose substrate.
Step 3: Cleavage of a carbon–carbon bond frees the aldose product, yielding an activated glycolaldehyde joined to TPP.
Step 4: The carbonyl group of an aldose acceptor condenses with the activated glycolaldehyde, forming a new ketose.
Step 5: The ketose is released from the enzyme, freeing TPP for the next reaction cycle.
The Transaldolase Reaction Involves a Schiff Base Intermediate
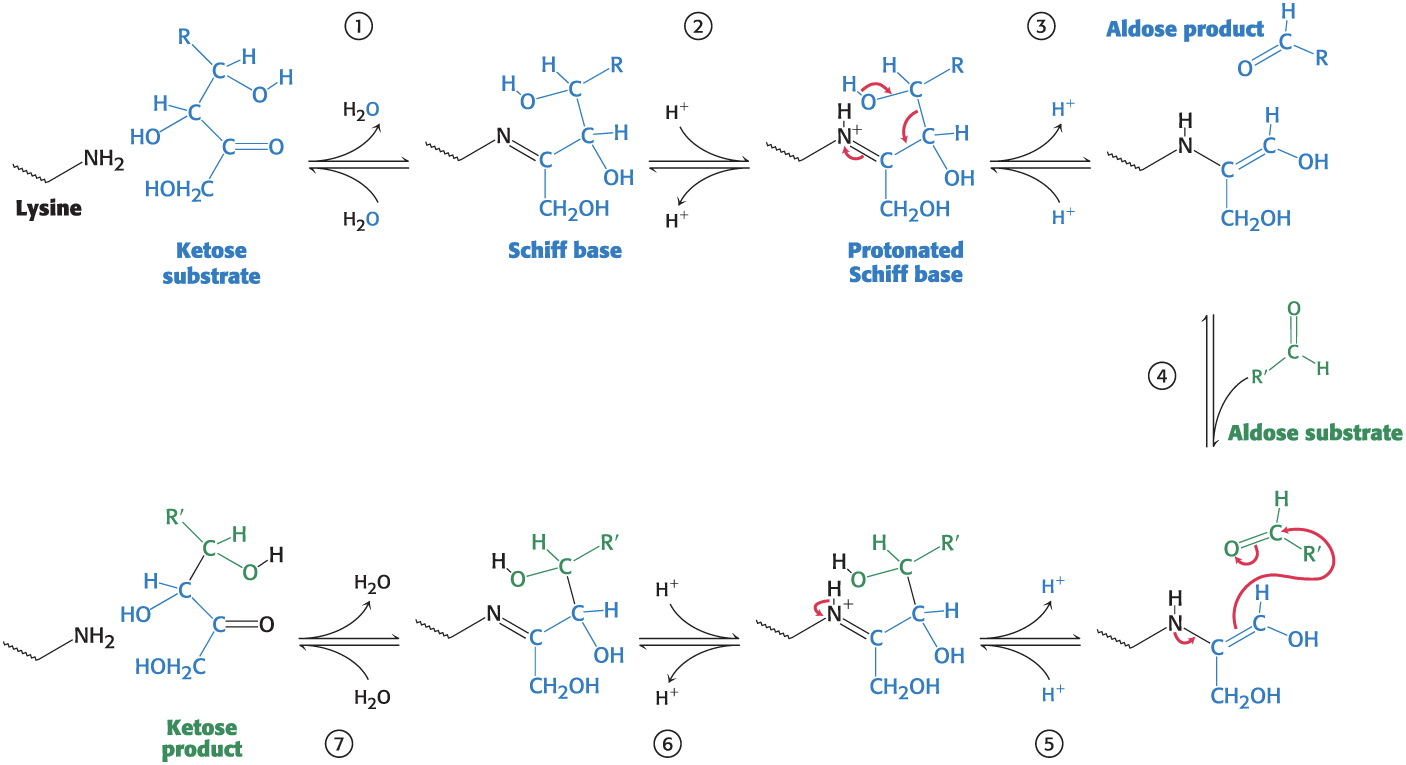
Transaldolase Reaction
Step 1: A Schiff base forms between a Lys residue intransaldolase and the ketose substrate.
Step 2: Protonation of the Schiff base occurs, and the bond between C-3 and C-4 is split.
Step 3: Deprotonation leads to the release of the aldose product, leaving a three-carbon fragment attached to Lys.
Step 4: A suitable aldose binds.
Step 5: Protonation allows the formation of new carbon–carbon bond.
Step 6: Deprotonation occurs.
Step 7: hydrolysis of the Schiff base releases the ketose
For Transketolase and Transaldolase, a Carbanion Intermediate Is Stabilized by Resonance
Section 20.4 The Metabolism of Glucose 6-Phosphate by the Pentose Phosphate Pathway Is Coordinated with Glycolysis
Glucose 6-phosphate is metabolized by both the glycolytic pathway and the pentose phosphate pathway.
The fate of glucose 6-phosphate is controlled by the cytoplasmic concentration of NADP+
The Rate of the Oxidative Phase of the Pentose Phosphate Pathway Is Controlled by the Level of NADP+
The first reaction of the pentose phosphate pathway is the dehydrogenation of glucose 6-phosphate by glucose 6-phosphate dehydrogenase.
This is the rate-limiting step of the pathway.
The reaction is essentially irreversible.
Low levels of NADP+ limit dehydrogenation of glucose 6-phosphate because it is needed as the electron acceptor.
ensures NADPH is not generated unless needed for reductive biosyntheses or protection against oxidative stress
The Flow of Glucose 6-Phosphate Depends on the Need for NADPH, Ribose 5-Phosphate, and ATP
The pentose phosphate pathway can operate in four distinct modes:
much more ribose 5-phosphate than NADPH is required
the needs for NADPH and ribose 5-phosphate are balanced
much more NADPH than ribose 5-phosphate is required
both NADPH and ATP are required
The Pentose Phosphate Pathway Can Operate in Four Modes
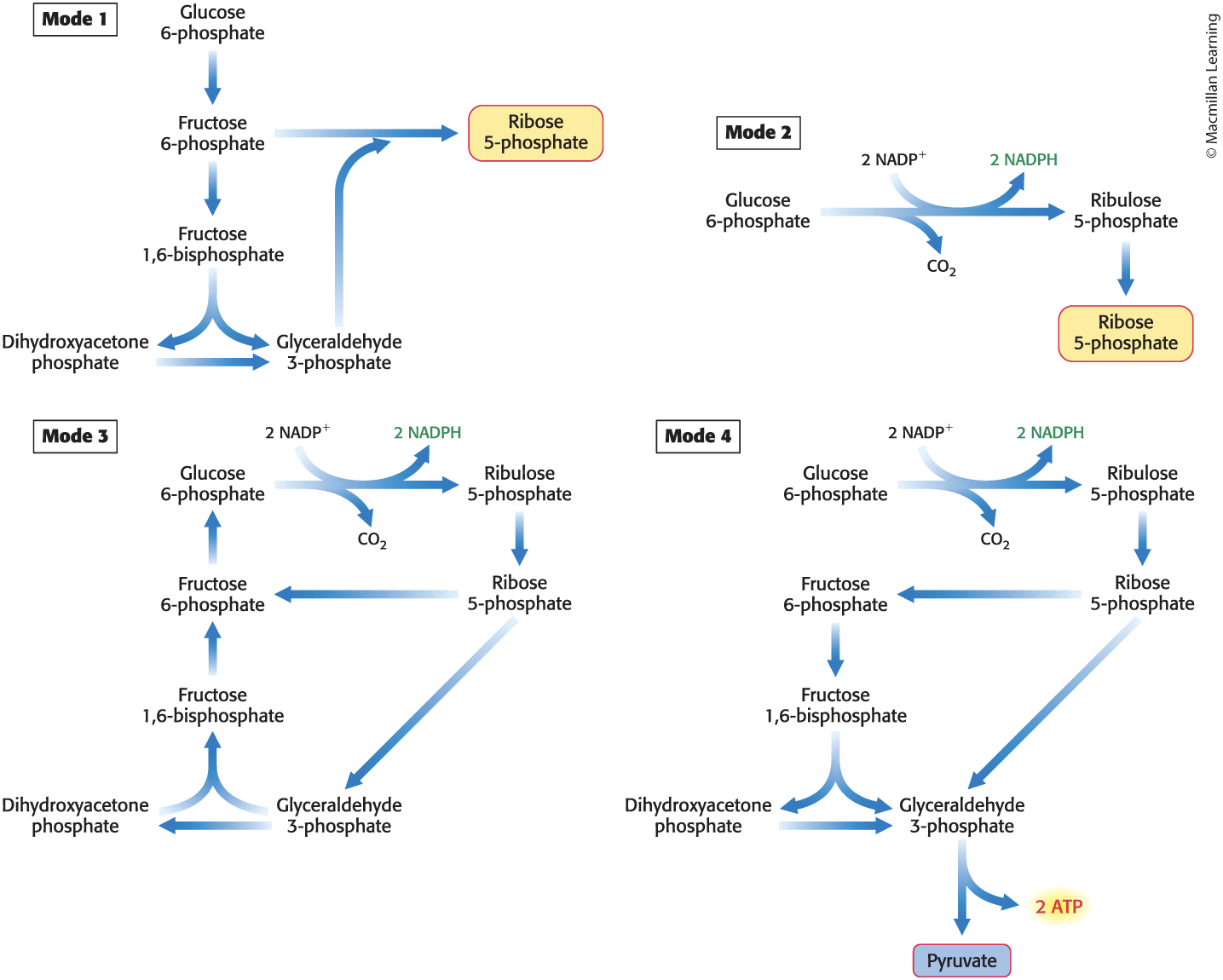
Mode 1—Much More Ribose 5-Phosphate Than NADPH Is Required
occurs, for example, when rapidly dividing cells need ribose 5-phosphate for the synthesis of nucleotide precursors of DNA
Glucose 6-phosphate is converted into fructose 6-phosphate (F-6P) and glyceraldehyde 3-phosphate (GAP) by the glycolytic pathway.
Transaldolase and transketolase convert two molecules of F-6P and one molecule of GAP into three molecules of ribose 5-phosphate.
The stoichiometry of mode 1 is
5 Glucose 6-phosphate + ATP → 6 ribose 5-phosphate + ADP + 2 H+
Mode 2—The Needs of Ribose 5-Phosphate and NADPH Are Balanced
Glucose 6-phosphate is processed to one molecule of ribulose 5-phosphate while generating two molecules of NADPH.
Ribulose 5-phosphate is converted into ribose 5-phosphate.
The stoichiometry of mode 2 is
Glucose 6-phosphate + 2 NADP+ + H2O → ribose 5-phosphate + 2 NADPH + 2 H+ + CO2
Mode 3—Much More NADPH Than Ribose 5-Phosphate Is Required
occurs, for example, when adipose tissue requires a high level of NADPH for the synthesis of fatty acids
Glucose 6-phosphate is completely oxidized to CO2 through three groups of reactions:
The oxidative phase of the pentose phosphate pathway forms two molecules of NADPH and one molecule of ribulose 5-phosphate.
Ribulose 5-phosphate is converted into F-6P and GAP by transketolase and transaldolase.
Glucose 6-phosphate is resynthesized from fructose.
The Stoichiometry of Mode 3
The stoichiometries of the three sets of reactions are
6 Glucose 6-phosphate + 12 NADP+ + 6 H2O → 6 Ribose 5-phosphate + 12 NADPH + 12 H+ + 6 CO2
6 Ribose 5-phosphate → 4 Fructose 6-phosphate + 2 glyceraldehyde 3-phosphate
4 Fructose 6-phosphate + 2 glyceraldehyde 3-phosphate + H2O → 5-Glucose 6-phosphate + Pi
The stoichiometry of mode 3 is
Glucose 6-phosphate + 12 NADP++ 7 H2O → 6 CO2 + 12 NADPH +12 H+ + Pi
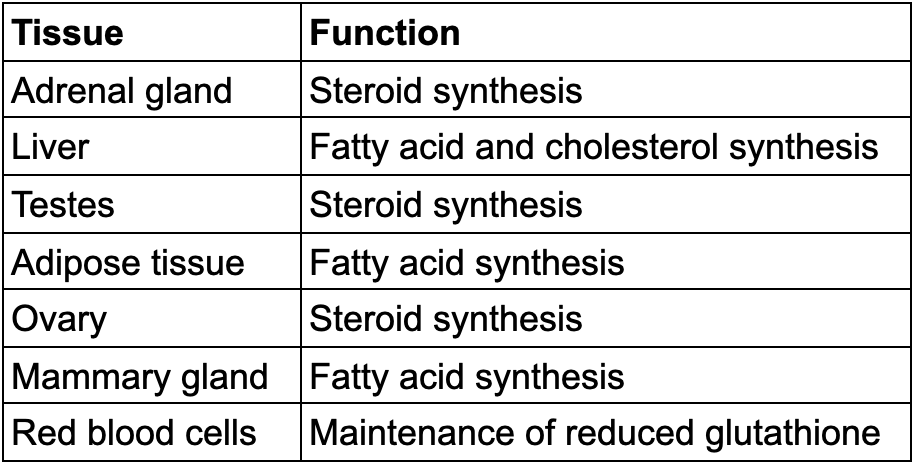
Tissues with Active Pentose Phosphate Pathways
TABLE 20.4 Tissues with active pentose phosphate pathways
Mode 4—Both Ribose 5-Phosphate and NADPH Are Required
Ribulose 5-phosphate formed from glucose 6-phosphate can be converted into pyruvate.
F-6P and GAP derived from ribose 5-phosphate enter the glycolytic pathway rather than reverting to glucose 6-phosphate.
Pyruvate formed can be oxidized to generate more ATP or used as a building block in a variety of biosynthesis.
The stoichiometry of mode 4 is
3 Glucose 6-phosphate + 6 NADP+ + 5 NAD+ + 5 Pi + 8 ADP → 5 pyruvate + 3 CO2 + 6 NADPH + 5 NADH + 8 ATP + 2 H2O + 8 H+
The Pentose Phosphate Pathway Is Required for Rapid Cell Growth
Rapidly dividing cells, such as cancer cells, require:
ribose 5-phosphate for nucleic acid synthesis.
NADPH for fatty acid and membrane lipid synthesis.
Rapidly dividing cells:
switch to aerobic glycolysis to meet ATP needs.
divert glucose 6-phosphate and glycolytic intermediates to the nonoxidative phase of the pentose phosphate pathway to generate NADPH and ribose 5-phosphate.
Pyruvate Kinase Isozyme, PKM
pyruvate kinase isozyme (PKM) = facilitates the diversion of glycolytic intermediates into the nonoxidative phase
PKM has a low catalytic activity.
Glycolytic intermediates accumulate and enter the pentose phosphate pathway.
The diversion of phosphorylated intermediates is further enabled by the inhibition of triose phosphate isomerase by phosphoenolpyruvate, the substrate of PKM.
The Calvin–Benson Cycle and the Pentose Phosphate Pathway Are Essentially Mirror Images of One Another
The Calvin–Benson cycle begins with the fixation of CO2 and proceeds to use NADPH in the synthesis of glucose.
The pentose phosphate pathway begins with the oxidation of a glucose-derived carbon atom to CO2, generating NADPH.
In photosynthetic organisms, many enzymes are common to the two pathways.
Section 20.5 Glucose 6-Phosphate Dehydrogenase Plays a Key Role in Protection Against Reactive Oxygen Species
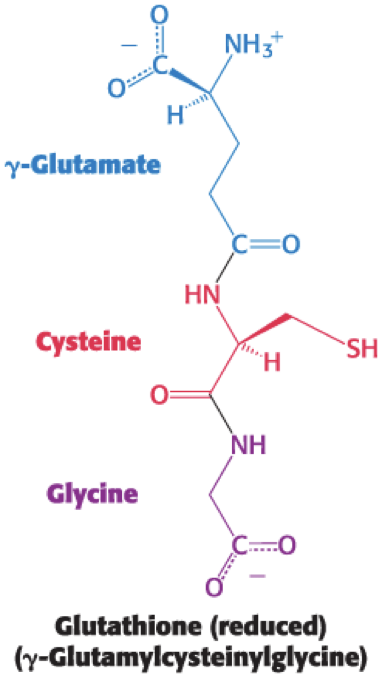
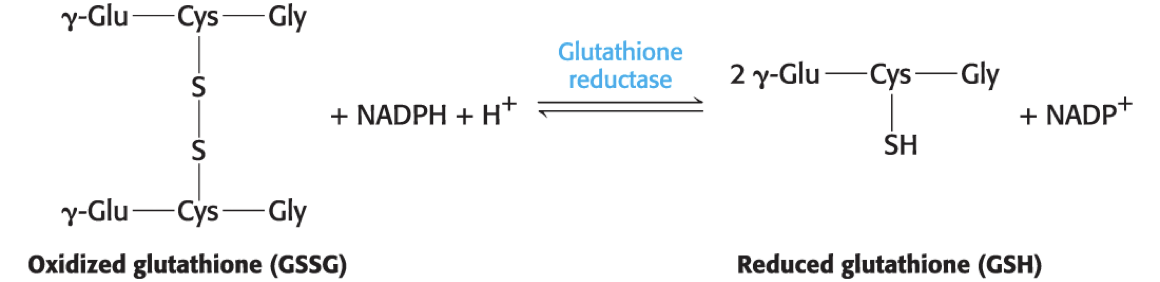
reduced glutathione (GSH) = a tripeptide with a free sulfhydryl group that combats oxidative stress by reducing ROS to harmless forms
Oxidized glutathione (GSSG) must be reduced to regenerate GSH.
Reducing power is supplied by the NADPH generated by glucose 6-phosphate dehydrogenase in the pentose phosphate pathway.
Glucose 6-Phosphate Dehydrogenase Deficiency Causes a Drug-Induced Hemolytic Anemia
pamaquine = the first synthetic antimalarial drug introduced in 1926
Some patients developed drug-induced hemolytic anemia due to a deficiency of glucose 6-phosphate dehydrogenase.
This deficiency results in a scarcity of NADPH in all cells.
most acute in red blood cells because they lack mitochondria
Primaquine
primaquine = an antimalarial closely related to pamaquine that is widely used in malaria-prone regions of the world
Drug-induced hemolytic anemia is seen in patients deficient in glucose 6-phosphate dehydrogenase.
Fava Beans Produce a Pyrimidine Glycoside
vicine = a pyrimidine glycoside of fava beans
People deficient in glucose 6-phosphate dehydrogenase suffer hemolysis from eating fava beans.
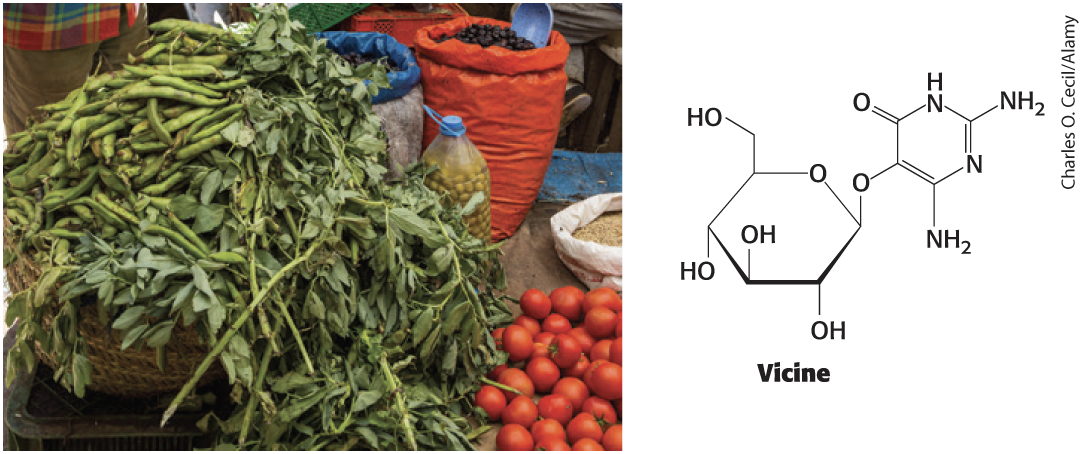
Pamaquine, Primaquine, and Vicine Generate Peroxides
peroxides = reactive oxygen species that can damage membranes and other biomolecules
Pamaquine, primaquine, and vicine are oxidative agents that generate peroxides.
Hemolysis Caused by Pamaquine, Primaquine, and Vicine
glutathione peroxidase = eliminates peroxides using glutathione as a reducing agent
In red blood cells, glutathione reductase uses NADPH to regenerate the reduced form of GSH.
Reduced Glutathione Maintains the Structure of Hemoglobin
Reduced form of glutathione serves as a sulfhydryl buffer that keeps the residues of hemoglobin in the reduced sulfhydryl form.
Without adequate levels of reduced glutathione, hemoglobin molecules cross-link with one another to form aggregates called Heinz bodies on cell membranes.
Membranes damaged by Heinz bodies and reactive oxygen species become deformed.
These cells are more likely to undergo lysis.
Heinz Bodies Are Made of Denatured Hemoglobin

A Deficiency of Glucose 6-Phosphate Dehydrogenase Confers an Evolutionary Advantage in Some Circumstances
glucose 6-phosphate dehydrogenase deficiency = characterized by a 10-fold reduction in enzymatic activity in red blood cells
more common among Americans of African heritage
protects against the deadliest form of malaria, which is prevalent in Africa's subtropical regions
Parasites causing malaria require NADPH for growth and infection induces oxidative stress in infected human cells.
Because the pentose phosphate pathway is compromised, the cells and parasite die from oxidative damage.