BIOC*2580 - 6 Study Notes on Enzyme Cofactors, Coenzymes, and Metabolism
Introduction to Metabolism and Enzyme Cofactors
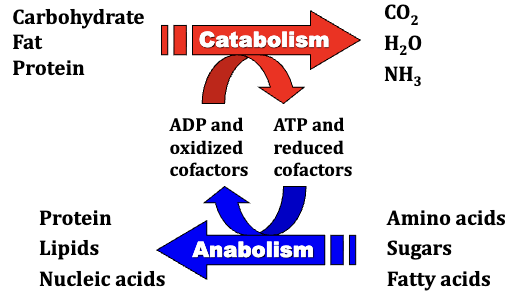
Metabolism involves all chemical reactions in cells, which can be categorized into two types: catabolism and anabolism.
Catabolism refers to the breakdown of complex, energy-rich biomolecules, releasing energy through exergonic reactions.
Anabolism involves building larger molecules from smaller units and requires energy input, typically from ATP and reduced cofactors.
Catabolic Processes
Catabolic pathways CONVERGE
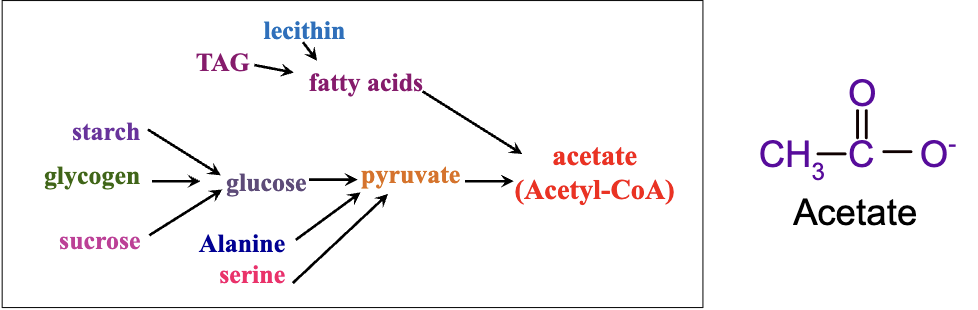
sugars, fats, and amino acids are converted into a single centrally-important metabolite, a carrier of two-carbon (acetate) units called acetyl coenzyme A (acetyl CoA).
Catabolic reactions break down carbohydrates, lipids, and proteins into simpler molecules such as carbon dioxide and water.
Exergonic reactions release energy used to convert ADP to ATP.
Catabolic processes involve oxidation of these biomolecules, requiring oxidized cofactors which are converted to reduced forms.
Oxidation: Loss of electrons or an increase in oxidation state.
Cofactors: Necessary components that assist enzymes in catalytic functions, particularly during oxidation processes.
Anabolic Processes
Anabolic pathways DIVERGE
Anabolic reactions construct larger biomolecules, such as polysaccharides and proteins, from smaller precursors like amino acids and monosaccharides.
Require energy input from ATP and reduced cofactors since these processes are reduction reactions.
Reduction: Gain of electrons or decrease in oxidation state.
Pathway Convergence and Divergence
Convergence in Catabolism: Various biomolecules (lipids, carbohydrates, amino acids) break down to a central metabolite, acetyl-CoA.
Acetyl-CoA is a vital molecule for cellular metabolism, analogous to roads converging into a highway leading to a city (Toronto analogy).
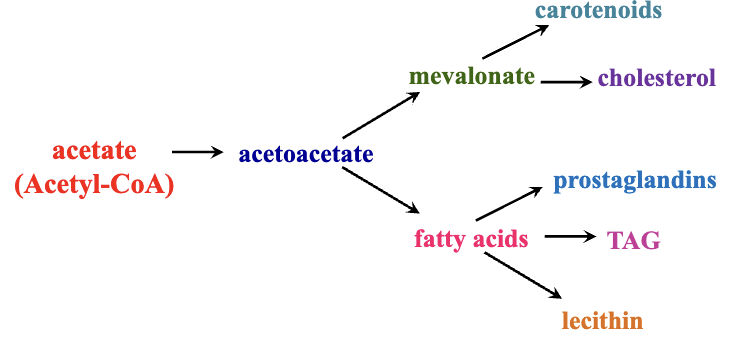
Divergence in Anabolism: Acetyl-CoA serves as a building block for synthesizing fatty acids, steroids, and other complex molecules.
Metabolic Pathways
Defined as a series of enzyme-catalyzed reactions transforming a substrate (e.g., A) into another molecule (e.g., E) through steps (A to B to C to E).
Each step in a pathway constitutes a metabolite, an intermediate product.
Identifying Changes: Understanding structures can help identify the nature of chemical changes (oxidation, reduction, functional group modification) happening at each step.
Irreversibility: Most metabolic pathways are unidirectional due to thermodynamically favorable steps that drive the process.
Example: Glycolysis (glucose to pyruvate) differs from gluconeogenesis (synthesis of glucose), sharing some steps but not reversing the pathway.
Regulation: Pathways are not continuously active; they are regulated based on cellular energy needs.
Studying Metabolic Pathways
Challenges exist due to the complexity and number of pathways. Methods include:
Using Metabolic Inhibitors: Inhibitors halt specific enzyme activities, raising metabolite concentrations for study.
Biochemical Genetics: Genetic diseases (e.g., L-captonuria) caused by enzyme mutations can shed light on metabolic pathways by revealing accumulated intermediates.
Radioactively Labeled Substrates ³H ^14C ³²P ³^5S: Utilization of radioactive isotopes allows tracing the conversion and identification of metabolites in complex mixtures.
If a compound is synthesized with a 14C carbon atom in it, then the compound, and any products derived from the 14C carbon atom, will be radioactive and is easily traceable
Redox Reactions in Metabolism
Overview of oxidation-reduction (redox) reactions, highlighting electron transfer between oxidized and reduced forms of molecules.
Oxidizing Agent: Gains electrons and is reduced.
Reducing Agent: Loses electrons and is oxidized.

Standard reduction potentials (E^0’) are used to determine the directionality of redox reactions, with higher potentials signifying a greater tendency to gain electrons.


Enzyme Cofactors and Coenzymes
Cofactors: Assist enzymes by either being inorganic (e.g., metals like iron) or organic molecules (coenzymes).
Inorganic ions: Fe2+, Mg2+, Mn2+, Zn2+, Cu2+
Coenzymes: Organic carriers of functional groups; play transient roles in various enzymatic reactions. (derivatives of adenosine)
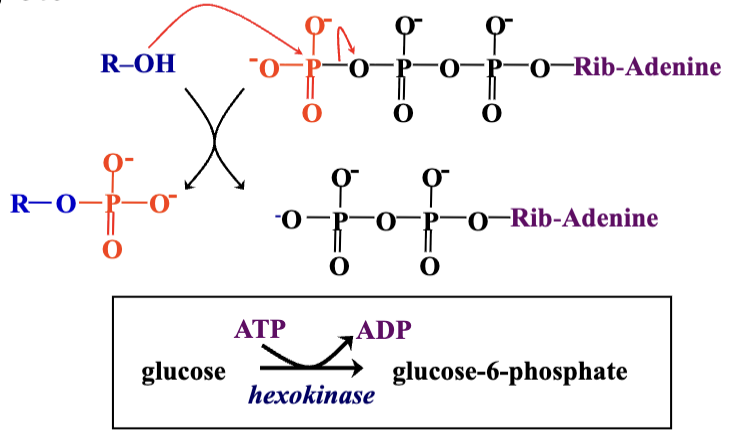
ATP Functions: Acts as an energy currency and a phosphate group donor in phosphorylation reactions, catalyzed by kinases (e.g., hexokinase for glucose phosphorylation).
Key Coenzymes
Coenzyme A (CoA) (CoA/CoASH): Acts as an acetyl group carrier, yielding thioesters. Derived from Vitamin B5 and involved in various metabolic reactions.
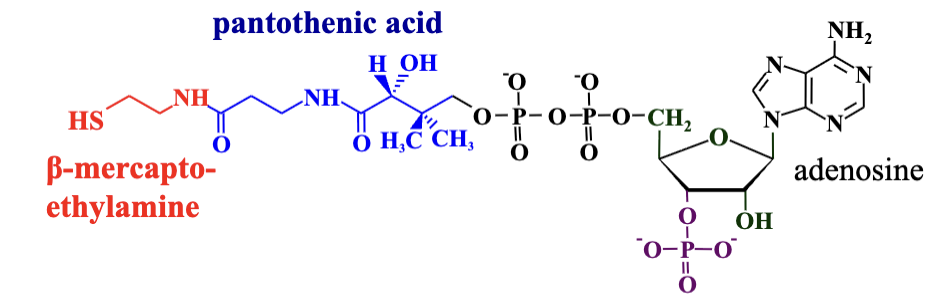
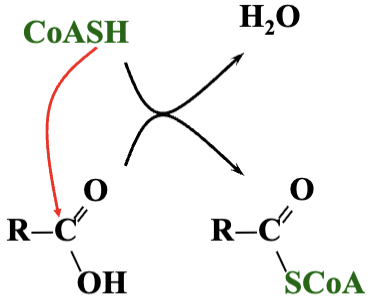
CoASH forms thioester derivatives with organic acids (R-COOH)
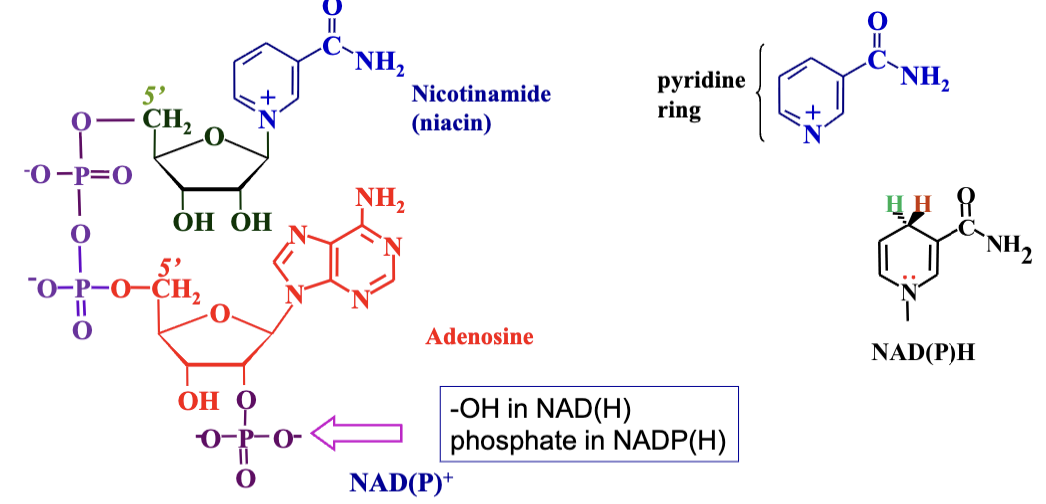
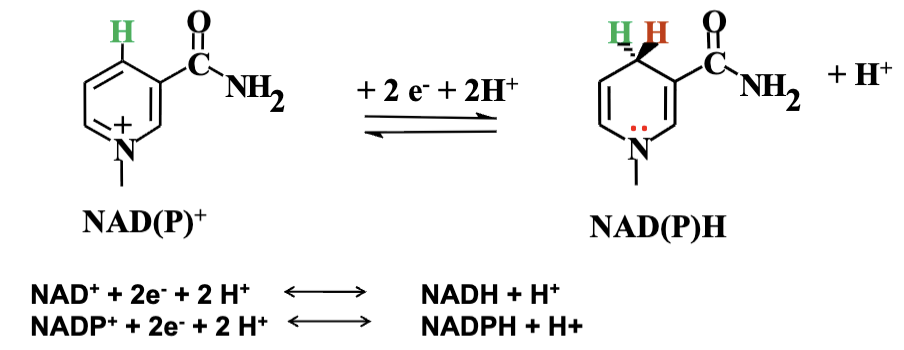
Universal Electron Carriers: Coenzymes like NAD+ and NADP+ play crucial roles in redox reactions. They are derived from niacin (B3), accept electrons, forming NADH and NADPH while facilitating the oxidation of substrates.
Flavin adenine dinucleotide (FAD) and Flavin mono nucleotide (FMN)
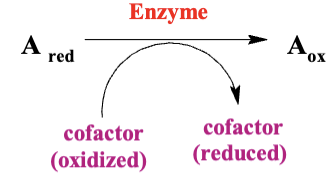
Many enzymes oxidize different molecules, but they only use a few common cofactors to accept the electrons. When these electrons are removed from the original molecules, they are transferred to these cofactors. This process reduces the cofactors and helps to save the energy that was released during the oxidation.
Redox Chemistry: They occur at the nicotinamide ring. Involves the removal of hydrogen atoms (1 electron and 1 proton) from the substrate, with NAD/NADP donation resulting in the formation of reduced forms (NADH/NADPH) and releasing protons.
Conclusion
Understanding enzyme cofactors and coenzymes is vital for grasping metabolic reactions and pathways successfully in biochemistry.