Chemical Reactions
Chemical Reactions
Chemical reactions are processes in which substances (reactants) undergo a transformation to form new substances (products). These reactions are fundamental to both chemistry and various fields such as biology and environmental science.
Identifying Reactants and Products
In any chemical reaction, the substances that undergo the change are called reactants, and the substances formed as a result of the reaction are called products.
For example, in the combustion of methane:
Reactants: Methane (( CH_4 )) and Oxygen (( O_2 ))
Products: Carbon Dioxide (( CO_2 )) and Water (( H_2O ))
Equation: ( CH_4 + 2O_2 → CO_2 + 2H_2O )
Law of Conservation of Matter and Energy
The Law of Conservation of Matter states that matter cannot be created or destroyed in a chemical reaction; it can only change forms. Similarly, the Law of Conservation of Energy states that energy cannot be created or destroyed, only transformed. Chemical reactions uphold these laws as the total mass of reactants is always equal to the total mass of products, and the energy before and after a reaction remains constant.
Balancing Chemical Equations
A chemical equation must be balanced to obey the Law of Conservation of Matter. This means the number of each type of atom must be the same on both sides of the equation. For example, for the reaction of water:[ 2H_2 + O_2 → 2H_2O ]Here, there are 4 hydrogen atoms and 2 oxygen atoms on each side of the equation.
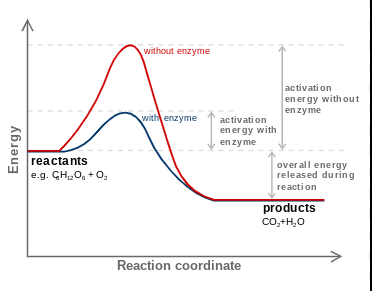
Reaction Coordinates
Reaction coordinates are graphical representations of the energy changes during a chemical reaction. They illustrate the energy contained in reactants and products:
The energy of reactants is higher in endergonic reactions, which require energy input, while in exergonic reactions, the energy of products is lower, releasing energy.
Organisms utilize enzymes (organic catalysts) to lower the activation energy required for reactions.In reaction coordinates, enzymes are represented by a decrease in the peak of the activation energy barrier, promoting the reaction efficiently without being consumed.
Exergonic vs. Endergonic Reactions
Exergonic Reactions: Release energy, usually in the form of heat.
Example: Cellular respiration (breaking down glucose)
Reaction Coordinate: Shows products with lower energy than reactants.
Endergonic Reactions: Absorb energy, requiring an input for process to occur.
Example: Photosynthesis (building glucose from CO2 and H2O)
Reaction Coordinate: Displays products with higher energy than reactants.
Conclusion
Chemical reactions are essential to understanding how substances interact and transform. Whether in nature, industry, or laboratories, these processes play a crucial role in our everyday lives.