Histology
Muscle Fiber and Myofibril
Muscle fibers are long, cylindrical cells that make up muscles.
Inside each fiber are many myofibrils, which are tiny thread-like structures.
Myofibrils contain the proteins needed for muscle contraction.
Sarcomere and Proteins
The sarcomere is the smallest unit of a muscle that contracts.
It has regions called Z-lines, M-line, and I-band.
Z line is at the middle of Actin filaments
I band is only Actin, no myosin
A band, is all myosin, including actin
H zone is only Myosin
M line is in the middle of myosin where it is thicker
Major proteins involved are actin, myosin, titin, and others that help in contraction and stabilization.
During contraction, the sarcomere shortens, with actin and myosin sliding past each other.
Recognizing TEMs in Muscle Groups
Transmission Electron Microscopy (TEM) images show detailed muscle structures.
Different muscle groups (skeletal, cardiac, smooth) have unique TEM features.
For example, skeletal muscle has clear striations, while smooth muscle does not.
Motor End Plate TEM Components
The motor end plate is where nerve signals meet muscle (whole thing called neuromuscular junction[NMJ]).
TEM shows synaptic vesicles, junctional folds, and acetylcholine receptors.
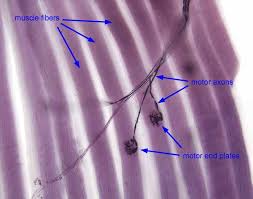
Excitation-Contraction (EC) Coupling
EC coupling links nerve signals to muscle contraction.
In skeletal muscle, it involves T-tubules and the sarcoplasmic reticulum.
Cardiac muscle has diads, and smooth muscle has different calcium handling.
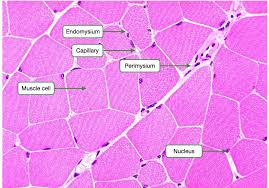
Connective Tissue Layers
Epimysium surrounds the entire muscle.
Perimysium surrounds fascicles (groups of fibers).
Endomysium surrounds individual muscle fibers.
Tendons connect muscle to bone, and bones have periosteum.
Series Elastic Component
It includes tendons and connective tissue that stretch during contraction.
It helps transmit force from muscle to bone.
Myocyte Folding at Myotendinous Junctions
Muscle cells fold at junctions to increase surface area.
This enhances force transfer to tendons.
Morphological Differences in Cardiomyocytes
Two types: atrial and ventricular.
Ventricular cells are larger with more organized sarcomeres.
Diads (dyads) are specialized structures in cardiac muscle for calcium release.
Understanding these structures helps explain how muscles contract and how they are built for their specific functions.
🧠 Epithelia – Study Summary
🧩 Definition & Key Trait
• Epithelia = continuous sheets of closely packed cells covering surfaces and lining cavities.
• Derived from all germ layers (ectoderm, mesoderm, endoderm).
• Functions: protection, secretion, absorption, transport, sensation, gas exchange, and lubrication.
• Polarity:
• Apical surface – faces lumen/exterior (microvilli, cilia, etc.)
• Lateral surface – has junctional complexes.
• Basal surface – rests on basement membrane (basal lamina + reticular lamina).
⸻
🔬 Basement Membrane
• Basal lamina (made by epithelial cells): contains laminin, type IV collagen, entactin, proteoglycans.
• Reticular lamina (made by connective tissue): type III collagen (reticular fibers).
• Functions: anchoring, selective barrier, scaffolding for regeneration.
⸻
🧱 Cell Junctions
• Tight (occluding) junctions – zonula occludens: seal cells together (claudins, occludins).
• Adherens junctions – zonula adherens: link actin filaments between cells (cadherins).
• Desmosomes – macula adherens: spot welds connecting intermediate filaments (keratin).
• Hemidesmosomes: anchor cells to basal lamina.
• Gap junctions: allow ion/small molecule exchange (connexons).
⸻
⚙ Apical Specializations
• Microvilli: increase surface area; actin core (seen in intestine, kidney).
• Cilia: motile, 9+2 microtubule arrangement (respiratory tract, oviduct).
• Stereocilia: long microvilli for absorption (epididymis) or mechanosensation (inner ear).
⸻
🧬 Epithelial Renewal
• High mitotic activity; renewal rates vary by location (rapid in intestines, slow in glands).
• Stem cells found in basal layers.
⸻
🧫 Classification by Layers & Shape
1. Simple (1 layer)
• Squamous: thin, flat → diffusion (endothelium, alveoli).
• Cuboidal: cube-shaped → secretion/absorption (kidney tubules, ducts).
• Columnar: tall → absorption, secretion (intestine, gallbladder).
2. Stratified (≥2 layers)
• Squamous keratinized: skin (epidermis).
• Squamous nonkeratinized: mouth, esophagus, vagina.
• Cuboidal/Columnar: larger ducts, conjunctiva.
3. Pseudostratified: looks layered but all cells touch basement membrane (respiratory epithelium).
4. Transitional (urothelium): stretches (bladder, ureter).
⸻
🩸 Specialized Types & Examples
• Endothelium: lines blood vessels (simple squamous).
• Mesothelium: lines body cavities (pleura, peritoneum).
• Ependyma: lines brain ventricles.
• Respiratory epithelium: pseudostratified ciliated columnar w/ goblet cells.
• Urothelium: dome-shaped surface cells; binucleated.
⸻
🧩 Quick Quiz
Q1: What two layers make up the basement membrane?
A1: Basal lamina and reticular lamina.
Q2: What are the main junctional complexes (from apical to basal)?
A2: Tight junction → Adherens junction → Desmosome → Gap junction.
Q3: What type of epithelium lines the urinary bladder?
A3: Transitional (urothelium).
Q4: Where would you find pseudostratified columnar ciliated epithelium?
A4: Respiratory tract (trachea, bronchi).
Q5: What protein forms the structural core of microvilli?
A5: Actin.
Q6: Which epithelial type is keratinized and what’s its function?
A6: Stratified squamous keratinized – protection (skin).
Q7: What’s the role of hemidesmosomes?
A7: Anchor epithelial cells to the basal lamina.
Q8: Which surface modification uses a 9+2 microtubule structure?
A8: Cilia.
🧠 Connective Tissue – Study Summary
🧩 Definition & Origin
• Connective tissue (CT) supports, connects, and binds other tissues.
• Derived primarily from mesenchyme (embryonic mesoderm).
• Composed of cells + extracellular matrix (ECM) (fibers + ground substance).
• ECM defines CT type and function.
⸻
🧫 Components of Connective Tissue
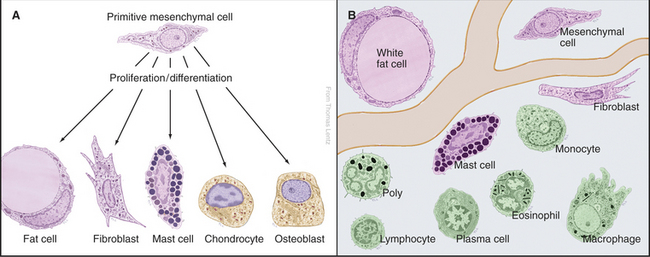
1. Cells
Resident cells (long-term):
• Fibroblasts: produce fibers (collagen, elastic, reticular) and ground substance.
• Adipocytes: store fat; secrete hormones like leptin.
• Macrophages: phagocytic defense and cleanup.
• Mast cells: release histamine/heparin (inflammation).
• Plasma cells: secrete antibodies (from B-lymphocytes).
Transient cells (move in/out): lymphocytes, eosinophils, neutrophils, monocytes.
⸻
2. Fibers
• Collagen fibers: strong, flexible; most abundant protein in body.
• Type I – skin, tendon, bone
• Type II – cartilage
• Type III – reticular fibers
• Type IV – basement membrane
• Reticular fibers: thin Type III collagen; form delicate meshwork (liver, spleen, lymph nodes).
• Elastic fibers: stretch and recoil (made of elastin + fibrillin; found in arteries, lungs, skin).
⸻
3. Ground Substance
• Amorphous gel-like material between fibers/cells.
• Contains glycosaminoglycans (GAGs), proteoglycans, glycoproteins.
• Function: diffusion medium for nutrients and waste; resists compression.
⸻
🧩 Classification of Connective Tissue
-Embryonic CT
• Mesenchyme: precursor to all CT types.
• Mucous CT: gelatinous (Wharton’s jelly in umbilical cord).
-Connective Tissue Proper
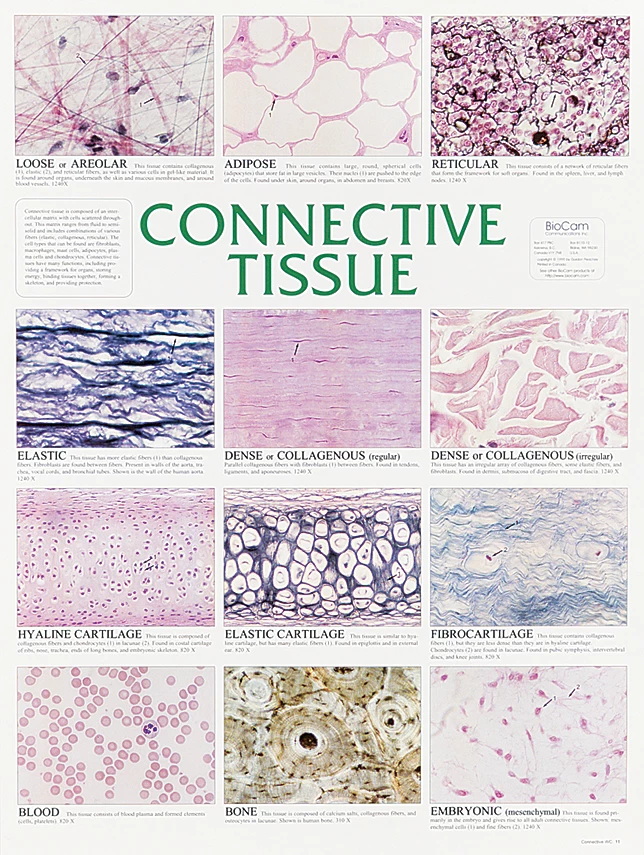
Loose (areolar) CT:
• More cells, fewer fibers; flexible; under epithelia, surrounds vessels/nerves.
• Functions: diffusion, cushioning, support.
Dense CT:
• More fibers, fewer cells.
• Dense irregular: fibers in many directions (dermis, organ capsules).
• Dense regular: parallel fibers (tendons, ligaments).
Specialized CT
• Adipose tissue: white (energy storage, insulation) and brown (heat production).
• Reticular tissue: supportive meshwork for lymphoid organs.
• Elastic tissue: found in large arteries and certain ligaments.
⸻
🩸 Supporting Connective Tissues
• Cartilage: avascular; cells = chondrocytes in lacunae.
• Types:
• Hyaline: most common (trachea, ends of long bones).
• Elastic: elastic fibers (ear, epiglottis).
• Fibrocartilage: collagen bundles (intervertebral discs).
• Bone: mineralized ECM; osteocytes in lacunae.
• Blood: fluid CT with plasma matrix and cellular components.
⸻
🧬 Clinical Connections
• Scurvy: Vitamin C deficiency → defective collagen synthesis.
• Ehlers–Danlos syndrome: abnormal collagen → hyperelastic skin, joint laxity.
• Marfan syndrome: fibrillin defect → weak elastic fibers (aortic aneurysm risk).
⸻
🧩 Quick Quiz
Q1: What are the three main components of CT?
A1: Cells, fibers, and ground substance.
Q2: Which CT fiber provides strength and flexibility?
A2: Collagen fibers.
Q3: What is the main fiber type in reticular tissue?
A3: Type III collagen.
Q4: What CT type forms tendons and ligaments?
A4: Dense regular CT.
Q5: Which CT type is most abundant under epithelia?
A5: Loose (areolar) CT.
Q6: What vitamin deficiency leads to weak collagen?
A6: Vitamin C (Scurvy).
Q7: What are the main structural molecules in ground substance?
A7: GAGs, proteoglycans, and glycoproteins.
Q8: What defect is linked to Marfan syndrome?
A8: Fibrillin (affects elastic fibers).
Q9: What is the primary function of collagen in connective tissue?
A9: Provides strength and structural support to tissues.
🦴 Bone – Study Summary
🧩 Overview
• Bone is a specialized connective tissue with a calcified extracellular matrix.
• Functions: support, protection, movement (levers for muscles), mineral storage (Ca²⁺, phosphate), and blood cell production (in marrow).
• Highly vascularized and dynamic — constantly remodeled.
Cells of bones:
Cells of bones:
• Osteoprogenitor cells:
Origin: Mesenchymal stem cells.
Function: Undifferentiated stem cells that can divide and differentiate into osteoblasts.
• Osteoblasts:
Origin: Differentiate from osteoprogenitor cells.
Function: Bone-forming cells; synthesize and secrete organic components of the bone matrix (osteoid), which then becomes mineralized. They are found on the surface of bone tissue.
• Osteocytes:
Origin: Mature osteoblasts that become trapped within the calcified matrix they secreted.
Function: Maintain the bone matrix, sense mechanical stress, and communicate with other bone cells to regulate bone remodeling. They reside in lacunae within the bone.
• Osteoclasts:
Origin: Derived from hematopoietic stem cells (monocyte-macrophage lineage).
Function: Bone-resorbing cells; break down bone tissue by secreting acids and enzymes, helping in bone remodeling and calcium homeostasis. They are large, multinucleated cells.
🧱 Bone Matrix
• Organic component (35%) → Type I collagen + ground substance → flexibility.
• Inorganic component (65%) → hydroxyapatite crystals (Ca₁₀(PO₄)₆(OH)₂) → hardness.
• Matrix arranged in lamellae (layers) in compact bone.
⸻
🦷 Types of Bone Tissue
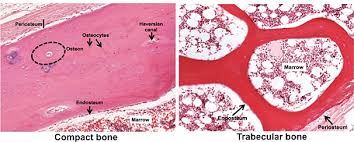
1. Compact (Cortical) Bone:
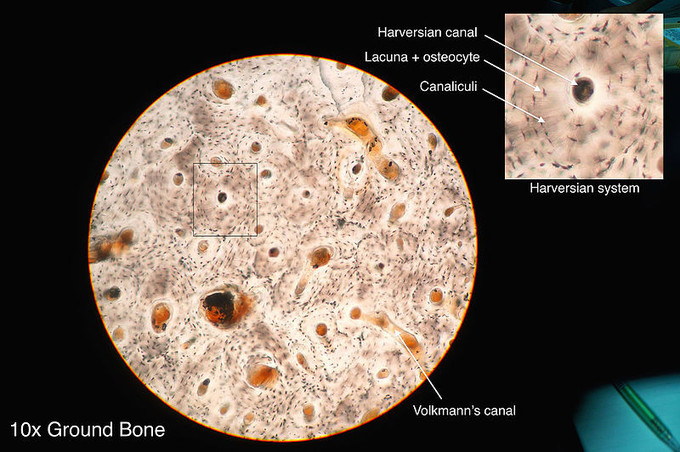
• Dense outer shell; composed of osteons (Haversian systems).
• Central (Haversian) canal = blood vessel + nerve; concentric lamellae around it.
• Volkmann’s canals connect Haversian canals transversely.
2. Spongy (trabecular) Bone:
• Meshwork of trabeculae with marrow spaces; no osteons; lighter but strong.
• Found in epiphyses and flat bones.
⸻
🧩 Periosteum & Endosteum
• Periosteum: outer fibrous layer (dense CT) + inner osteogenic layer (osteoprogenitors).
• Endosteum: thin osteogenic layer lining inner bone surfaces and trabeculae. (looks to me like its between bone and marrow, by looking at picture above)
• Both important for growth and repair.
⸻
⚙ Bone Formation (Ossification)
1. Intramembranous Ossification:
• Directly from mesenchyme → flat bones of skull, mandible, clavicle.
• Steps: mesenchymal condensation → osteoblast differentiation → osteoid secretion → mineralization.
2. Endochondral Ossification:
• Uses cartilage model → long bones.
• Steps: cartilage model → calcification → vascular invasion → primary ossification center (diaphysis) → secondary centers (epiphyses).
• Epiphyseal plate = growth region (until fusion after puberty).
⸻
🔄 Bone Remodeling
• Ongoing balance between osteoblasts (formation) and osteoclasts (resorption).
• Controlled by:
• PTH (parathyroid hormone): ↑ osteoclast activity → ↑ blood Ca²⁺.
• Calcitonin: inhibits osteoclasts → ↓ blood Ca²⁺.
• Estrogen/testosterone: maintain bone density.
• Vitamin D: promotes Ca²⁺ absorption and mineralization.
⸻
🩸 Histological Features
• Osteon: concentric lamellae around Haversian canal.
• Lacunae: contain osteocytes.
• Canaliculi: tiny channels connecting lacunae for nutrient exchange.
• Interstitial lamellae: remnants of old osteons.
• Cement line: boundary of osteon.
⸻
⚕ Clinical Notes
• Osteoporosis: decreased bone mass → fractures (↑ osteoclast activity).
• Rickets / Osteomalacia: vitamin D deficiency → poor mineralization.
• Paget’s disease: excessive remodeling → weak, disorganized bone.
⸻
🧩 Quick Quiz
Q1: What is the organic part of bone matrix mainly composed of?
A1: Type I collagen and ground substance.
Q2: What are the main inorganic crystals in bone?
A2: Hydroxyapatite.
Q3: What cells resorb bone?
A3: Osteoclasts.
Q4: Where are osteocytes found?
A4: In lacunae connected by canaliculi.
Q5: What type of ossification forms long bones?
A5: Endochondral ossification.
Q6: Which hormone increases blood calcium by stimulating osteoclasts?
A6: Parathyroid hormone (PTH).
Q7: What lines the inner surfaces of bone?
A7: Endosteum.
Q8: What connects adjacent Haversian canals?
A8: Volkmann’s canals.
🧠 Muscle Tissue - Study Summary
• Main function: contraction (movement & shape change)
• Origin: mesoderm (except some facial, eye, and aortic muscles from neural crest)
• Three major types:
• Skeletal – voluntary, striated
• Cardiac – involuntary, striated
• Smooth – involuntary, non-striated
Connective Tissue Layers
Epimysium surrounds the entire muscle.
Perimysium surrounds fascicles (groups of fibers).
Endomysium surrounds individual muscle fibers.
Tendons connect muscle to bone, and bones have periosteum.
💪 Skeletal Muscle
• Structure: long, cylindrical, multinucleated cells with peripheral nuclei
• Development: myoblasts → fuse into myotubes → myofibers once innervated
• Fiber types:
• Red (Type I): slow-twitch, oxidative
• White (Type II): fast-twitch, glycolytic
• Intermediate: mixed metabolism
• Sarcomere structure:
• Z-line to Z-line
• I-band (actin), A-band (myosin), H-zone (myosin only), M-line (center)
• Accessory proteins:
• Titin: anchors myosin to Z-line
• α-Actinin: anchors actin to Z-line
• Nebulin: stabilizes actin
• Myomesin/C-protein: M-line stabilization
• Triad: T-tubule + 2 terminal cisternae of SR (for excitation–contraction coupling)
• Myoneural Junction: 1 motor neuron per fiber (acetylcholine release)
• Myotendinous junction: collagen of tendon interdigitates with fiber folds → tension transfer
⸻
❤ Cardiac Muscle
• Origin: splanchnopleure (visceral mesoderm)
• Structure: branched fibers, central nucleus, striated, connected by intercalated discs
• Intercalated discs contain:
• Fascia adherens
• Desmosomes (macula adherens)
• Gap junctions
• Dyads (not triads) at Z-line (T-tubule + 1 SR cisterna)
• 99% contractile cells, 1% conduction system
• Myoendocrine cells (ANP): regulate blood pressure & fluid balance
• Purkinje fibers: part of conducting system
⸻
🌀 Smooth Muscle
• Location: GI, respiratory, urinary, reproductive tracts, blood vessels, skin, etc.
• Structure: fusiform cells with central nuclei, dense bodies (α-actinin-rich), no striations
• Filaments: actin:myosin ratio ≈ 12–14:1 (skeletal = 6:1)
• No troponin or T-tubules
• Excitation–Contraction coupling:
• Ca²⁺ binds calmodulin → activates myosin light-chain kinase → phosphorylates myosin → contraction
• Triggered by voltage (ACh, norepinephrine) or hormones (angiotensin, vasopressin)
• Types:
• Visceral (unitary): many gap junctions → synchronized contraction
• Multiunit: few gap junctions, individually innervated (e.g. iris, large arteries)
• Synthetic function: produces collagen, elastin, and laminin (important in vessel repair and atherosclerosis)
• Variants: myoepithelial cells (in glands) and pericytes (around capillaries)
Excitation-Contraction Coupling Summary
Skeletal muscle is voluntary, it gets its Ca²+ from SR via the t-tubule, its key proteins are triponin and tropomyosin and its structure is triad.
Cardiac muscle is involuntary, it gets its Ca²+ from SR and extracellular, its key protein is troponin and its structure is dyad.
Smooth muscle is involuntary. It gets its Ca²+ from Extracellular and SR. Its key proteins are calmodulin and MLCK and its structure is caveolae.
Quick quiz
Q1: What are the three main types of muscle tissue?
A1: Skeletal (voluntary, striated), Cardiac (involuntary, striated), and Smooth (involuntary, non-striated).
Q2: What embryonic layer gives rise to most muscle tissue?
A2: Mesoderm (except some from neural crest).
Q3: Which connective tissue layer surrounds individual muscle fibers?
A3: Endomysium.
Q4: What connective tissue layer surrounds fascicles (bundles of fibers)?
A4: Perimysium.
Q5: What are the structural and functional units of striated muscle?
A5: Sarcomeres (Z-line to Z-line)
Q6: What are the major proteins of the sarcomere?
A6: Actin (thin), Myosin (thick), Titin, Nebulin, α-Actinin, Myomesin, C-protein.
Q7: What is a triad, and where is it found?
A7: One T-tubule + two terminal cisternae of the sarcoplasmic reticulum; found in skeletal muscle at the A–I junction.
Q8: What neurotransmitter is released at the neuromuscular junction?
A8: Acetylcholine (ACh).
Q9: What type of muscle has peripheral nuclei and voluntary control?
A9: Skeletal muscle.
Q10: What type of muscle has intercalated discs?
A10: Cardiac muscle.
Q11: What are the three components of intercalated discs?
A11: Fascia adherens, Desmosomes (macula adherens), and Gap junctions.
Q12: What structure in cardiac muscle replaces the triad found in skeletal muscle?
A12: Dyad (T-tubule + one SR cisterna).
Q13: What type of specialized cardiac cells produce atrial natriuretic peptide (ANP)?
A13: Myoendocrine cells.
Q14: What are Purkinje fibers?
A14: Modified cardiac muscle fibers that conduct electrical impulses (part of the conduction system).
Q15: What are dense bodies in smooth muscle, and what do they do?
A15: α-actinin–rich structures that anchor actin filaments (analogous to Z-lines).
Q16: How is contraction initiated in smooth muscle?
A16: Ca²⁺ binds to calmodulin → activates myosin light-chain kinase (MLCK) → phosphorylates myosin → contraction.
Q17: What are the two main types of smooth muscle?
A17: Visceral (unitary) – many gap junctions, synchronized contraction;
Multiunit – few gap junctions, independently controlled (e.g. iris).
Q18: What are caveolae, and which muscle type has them?
A18: Invaginations of the membrane functioning like T-tubules; found in smooth muscle.
Q19: What is the ratio of actin to myosin in smooth muscle?
A19: Approximately 12–14:1 (compared to 6:1 in skeletal muscle).
Q20: What is the function of the myotendinous junction?
A20: Increases surface area for force transfer between muscle fibers and tendons.
Q21: Which muscle types can regenerate?
A21: Skeletal muscle (limited, via satellite cells) and smooth muscle (readily); cardiac muscle has almost no regenerative ability.
🧠 Neurohistology Summary
1. Organization of the Nervous System
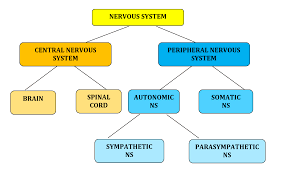
• CNS: Brain and spinal cord.
• PNS: 43 paired nerves (cranial + spinal).
• Afferent (sensory) → to CNS.
• Efferent (motor) → away from CNS.
• Somatic: Skeletal muscle.
• Autonomic: Cardiac, smooth muscle, glands.
• Sympathetic: Thoracolumbar.
• Parasympathetic: Craniosacral.
⸻
2. Neurons
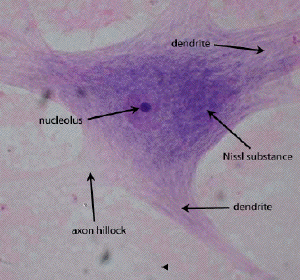
• Functional units of the nervous system.
• Contain cell body (soma), dendrites, axon.
• Nissl bodies: Basophilic clumps of RER + ribosomes (protein synthesis).
• Axonal transport:
• Anterograde (kinesin) → away from soma.
• Retrograde (dynein) → toward soma.
• Slow: 0.2–4 mm/day.
• Fast: 20–400 mm/day.
Neuron classifications:
• A & C fibers: By conduction velocity (physiologists).
• I–IV fibers: By diameter (anatomists).
• Large diameter = faster conduction.
⸻
3. Myelination
• CNS: Oligodendrocytes (can myelinate multiple axons).
• PNS: Schwann cells (one segment per axon).
• Mesaxon: Membrane wrap start point.
• Major dense line: Cytoplasmic surfaces.
• Interperiod line: Extracellular surfaces.
• Nodes of Ranvier: Gaps allowing saltatory conduction.
• Incisures of Schmidt-Lanterman: Schwann cell cytoplasmic channels for metabolite exchange.
Glial Cells - Location and Function
Glial cells, also known as neuroglia, are non-neuronal cells in the central nervous system (CNS) and the peripheral nervous system (PNS) that do not produce electrical impulses. They maintain homeostasis, form myelin, and provide support and protection for neurons.
Central Nervous System (CNS) Glia
Astrocytes
Location: Abundant throughout the CNS, especially in the gray matter.
Function:
Provide structural support to neurons.
Regulate the extra-cellular environment by controlling ion concentrations and neurotransmitter uptake (e.g., glutamate).
Form the blood-brain barrier by interacting with endothelial cells.
Guide neuronal migration during development and aid in synapse formation and function.
Respond to injury by forming glial scars.
Oligodendrocytes
Location: Found in the white and gray matter of the CNS.
Function:
Produce myelin sheaths for axons within the CNS, insulating them and increasing the speed of nerve impulse conduction.
A single oligodendrocyte can myelinate multiple axons.
Microglia
Location: Distributed throughout the CNS.
Function:
Resident immune cells of the CNS; act as phagocytes to remove cellular debris, pathogens, and damaged neurons.
Monitor the CNS environment for injury and inflammation.
Ependymal Cells
Location: Line the ventricles of the brain and the central canal of the spinal cord.
Function:
Form a permeable barrier between the cerebrospinal fluid (CSF) and the brain/spinal cord tissue.
Produce and circulate CSF via their cilia.
Peripheral Nervous System (PNS) Glia
Schwann Cells
Location: Surround axons in the PNS.
Function:
Form myelin sheaths around axons in the PNS.
Unlike oligodendrocytes, each Schwann cell typically myelinates only one segment of a single axon.
Aid in regeneration of damaged PNS axons.
Satellite Cells
Location: Surround neuron cell bodies (soma) in peripheral ganglia (e.g., dorsal root ganglia, autonomic ganglia).
Function:
Provide structural and metabolic support to neurons in ganglia.
Regulate the external chemical environment around neuronal cell bodies.
5. Synapses
• Types: Axodendritic, Axosomatic, Axoaxonic (can have varicosities).
• Can be chemical (via neurotransmitters) or electrical (gap junctions).
⸻
6. CNS Structures
• Gray matter: Neuron cell bodies.
• White matter: Myelinated axons.
• Cerebellum: Distinct Purkinje cells, large dendritic trees.
• Ependyma: Ciliated lining of ventricles + central canal.
• Choroid plexus: Modified ependymal folds with capillaries → produces CSF.
• Arachnoid villi: Drain CSF into venous sinuses (one-way valve).
⸻
7. PNS Structures
Connective Tissue Layers:
• Endoneurium – around individual axons.
• Perineurium – around fascicles (blood-nerve barrier).
• Epineurium – outer dense CT around entire nerve.
Ganglia:
• Dorsal root (sensory): Pseudounipolar neurons, central nucleus.
• Sympathetic: Multipolar neurons, eccentric nucleus, mixed fibers.
• Satellite cells: Small glial cells around neuron cell bodies.
⸻
8. Nerve Injury & Regeneration
• Wallerian (anterograde) degeneration: Axon distal to injury degenerates.
• Chromatolysis: Soma swelling, Nissl dispersal, nucleus moves peripherally.
• Regeneration: Schwann cells proliferate → form regeneration tube → axonal sprouts regrow (~1–3 mm/day) if external lamina intact.
⸻
9. Origins
• Neurons & glia (except microglia): Neuroectoderm.
• Microglia: Mesoderm (monocyte lineage).
• Schwann & satellite cells: Neural crest.
⸻
10. Lipofuscin
• Age pigment (residual bodies) in older neurons.
⸻
🧩 Quick Q&A (for flashcards)
Q1: What are the two main divisions of the nervous system?
A1: CNS (brain, spinal cord) and PNS (nerves, ganglia).
Q2: Which cells form the blood-brain barrier?
A2: Astrocytes (via end-feet) and capillary endothelial tight junctions.
Q3: What are Nissl bodies composed of?
A3: Rough endoplasmic reticulum and ribosomes.
Q4: What proteins mediate axonal transport?
A4: Kinesin (anterograde) and dynein (retrograde).
Q5: What is the function of the Nodes of Ranvier?
A5: Allow saltatory conduction to speed impulse transmission.
Q6: What’s the difference between Schwann cells and oligodendrocytes?
A6: Schwann = PNS, one segment; Oligodendrocyte = CNS, multiple axons.
Q7: What are the three connective tissue layers around nerves?
A7: Endoneurium, Perineurium, Epineurium.
Q8: What is Wallerian degeneration?
A8: Degeneration of an axon distal to a site of injury.
Q9: What happens during chromatolysis?
A9: Soma swells, Nissl disperses, nucleus shifts to edge.
Q10: What produces cerebrospinal fluid (CSF)?
A10: Ependymal cells of the choroid plexus.
Q11: Where is lipofuscin found?
A11: In older neurons as residual pigment granules.
Q12: What is the function of arachnoid villi?
A12: Drain CSF into venous circulation via one-way valves.
Q13: Which glial cells are phagocytic?
A13: Microglia.
Q14: What types of neurons are in dorsal root ganglia?
A14: Pseudounipolar sensory neurons.
Q15: What type of neurons are in sympathetic ganglia?
A15: Multipolar efferent neurons.