S1.2 The nuclear atom Notes
S1.2.1 atomic structure
Components of the Atom and Nuclear Symbols
The Components of the Atom
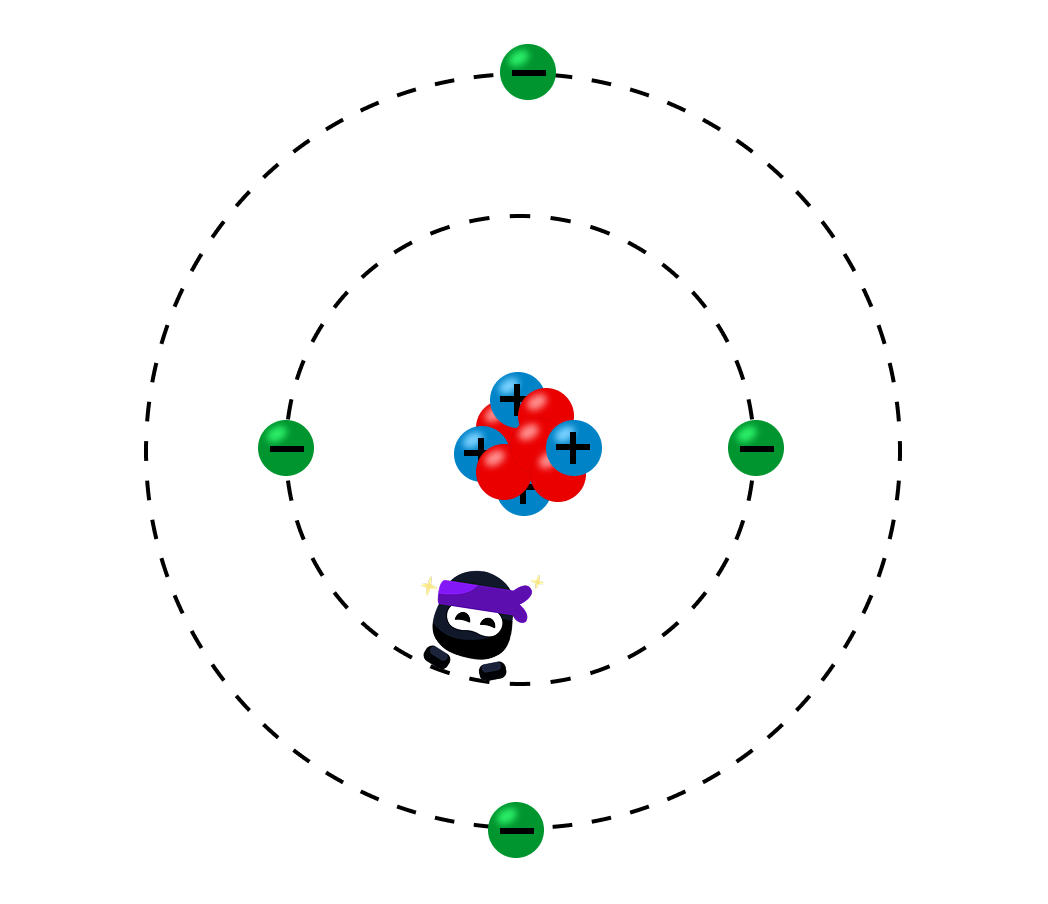
Atoms are the fundamental building blocks of matter, but they are not indivisible.
They consist of three types of subatomic particles: protons, neutrons, and electrons.
Each of these particles has distinct properties and plays a specific role in the atom.
The Nucleus: Protons and Neutrons
At the heart of the atom lies thenucleus.
Definition
NucleusA nucleus is a dense, positively charged core that contains protons and neutrons, collectively called nucleons.
Protons:
These are positively charged particles with a relative charge of and a relative mass of .
The number of protons in the nucleus determines the atomic number (Z), which defines the element.
Neutrons:
These particles are neutral, with no charge, and have a relative mass of .
Neutrons help to stabilize the nucleus by offsetting the repulsion between protons.
The number of neutrons can vary between atoms of the same element, leading to isotopes.
Note
Protons and neutrons are approximately 1,836 times more massive than electrons, meaning nearly all the mass of an atom is concentrated in its nucleus.
2.The Energy Levels
Surrounding the nucleus is a vast region of space occupied by electrons, which are negatively charged particles with a relative charge of .
Electrons have a negligible mass compared to protons and neutrons.
Electrons are arranged in regions of space called orbitals, which define the probability of finding an electron in a certain area.
In a neutral atom, the number of electrons equals the number of protons, balancing the positive and negative charges.
Relative Masses and Charges of Subatomic Particles
To understand the behavior of atoms, it's important to know the relative masses and charges of their subatomic particles.
These are summarized in the table below:
Particle | Relative Mass | Relative Charge | Location |
|---|---|---|---|
Proton | 1 | +1 | Nucleus |
Neutron | 1 | 0 | Nucleus |
Electron | Negligible | -1 | Outside the nucleus |
Tip
The mass of an electron is so small compared to protons and neutrons that it is often considered negligible in calculations.
Nuclear Symbols: Decoding the Atom's Identity
Chemists use nuclear symbols to summarize the structure of an atom or ion.
A nuclear symbol is written in the form: where:
= Chemical symbol of the element (e.g., for gold).
= Atomic number (number of protons).
= Mass number (total number of protons and neutrons).
Example
Gold
For gold, the nuclear symbol is:
This tells us:
: Gold has 79 protons.
: Gold has a total of 197 nucleons.
Number of neutrons = .
Self Review
Can you determine the number of protons, neutrons, and electrons in ?
Atoms vs. Ions: What Happens When Electrons Change?
While atoms are neutral, they can gain or lose electrons to form ions.
Definition
IonAn ion is an atom or group of atoms that has gained or lost electrons, resulting in a net electrical charge.
Thisdoes not affectthe number ofprotonsorneutronsbut changes the overall charge of the species.
Cations: Formed when an atom loses electrons, resulting in a positive charge.
Anions: Formed when an atom gains electrons, resulting in a negative charge.
Example
Magnesium ion () has 12 protons but only 10 electrons.
Example
An oxide ion () has 8 protons and 10 electrons.
Example question
Deduce the nuclear symbol for an ion with:
24 protons
21 electrons
28 neutrons
Show answer
Example
Magnesium forms a ion by losing two electrons.
Its nuclear symbol is .
This indicates 12 protons, 12 neutrons (), and 10 electrons.
Warning
Students often confuse the mass number () with the atomic number ().
Remember: includes both protons and neutrons, while is just the number of protons.
s1.2.2 isotopes
Isotopes and Their Properties
You're comparing coins of the same currency.
Some are slightly heavier than others because they contain more metal, yet they belong to the same denomination and have the same face value.
This is similar to how isotopes work in chemistry.
What Are Isotopes?
Atoms are defined by their number of protons, known as the atomic number (Z).
However, atoms of the same element can have different numbers of neutrons, which changes their mass number (A).
These different versions of the same element are called isotopes.
Definition of Isotopes
Definition
IsotopesIsotopes are atoms of the same element that have the same number of protons but different numbers of neutrons.
This meansisotopesshare:
The same atomic number (Z), since the number of protons determines the element.
Different mass numbers (A), because the mass number is the sum of protons and neutrons.
Example
Chlorine isotopes
Chlorine-35 () has 17 protons and 18 neutrons.
Chlorine-37 () has 17 protons and 20 neutrons.
Note
The chemical properties of isotopes are nearly identical because chemical behavior depends on the number of electrons, which remains the same for all isotopes of an element.
Relative Atomic Mass and Isotopic Abundance
Take a look at the periodic table in the data booklet.
You'll notice that atomic masses of elements are rarely whole numbers.
For instance, chlorine's relative atomic mass () is 35.45, even though its isotopes have whole-number mass numbers (35 and 37).
You can ask, why is this?
Definition
Relative atomic massThe relative atomic mass of an element is a weighted average of the masses of its isotopes, based on their natural abundances, compared to 1/12th the mass of a carbon-12 atom. Since it is a ratio, has no units.
Definition
Relative abundanceRelative abundance refers to the percentage of a specific isotope of an element present in a naturally occurring sample.
It indicates how common each isotope is compared to the total amount of the element, which is used to calculate the element's average atomic mass.
Calculating Relative Atomic Mass
To calculate therelative atomic mass(), use the formula:
Example
Relative atomic mass of chlorine
Chlorine has two isotopes:
Chlorine-35 () with 75.8% abundance.
Chlorine-37 () with 24.2% abundance.
Calculate the relative atomic mass:
Step-by-step:
Multiply each isotope's mass by its abundance:
Add these values together:
Divide by 100 to find the average:
Thus, the relative atomic mass of chlorine is approximately 35.45.
Tip
Before starting your calculation, double-check that the percentage abundances add up to 100% to avoid errors.
Determining Percent Abundances
To determine percent abundance from the relative atomic mass () and mass numbers of isotopes, use the weighted average formula: where and are the mass numbers of the isotopes, and .
If the percent abundance of one isotope is unknown, let and solve using:
Rearrange the equation to solve for , then multiply by 100 to express the abundance as a percentage.
Note
This method can be extended for elements with more than two isotopes.
Physical Properties of Isotopes
While isotopes of the same element have nearly identical chemical properties, their physical properties can differ.
These differences arise because isotopes have different masses, which can impact properties like density, melting point, and boiling point.
The slight difference in mass allows these isotopes to be separated using methods like gas centrifugation.
Example
Hydrogen Isotopes:
Protium (): The most common isotope, with 1 proton and no neutrons.
Deuterium (): A heavier isotope, with 1 proton and 1 neutron.
Tritium (): A radioactive isotope, with 1 proton and 2 neutrons.
Example
Uranium Isotopes:
Uranium-235 (): Used in nuclear reactors and weapons.
Uranium-238 (): More abundant but less useful for nuclear reactions.
Warning
Don't confuse physical property differences of isotopes with their chemicalproperties.
Isotopes react chemically in the same way because they have the same number of electrons.
Self Review
What are the key differences between isotopes of the same element?
Why do isotopes have the same chemical properties but different physical properties?
Calculate the relative atomic mass of an element with the following isotopes:
Isotope A: mass = 10, abundance = 20%
Isotope B: mass = 11, abundance = 80%.