Excitation - contraction coupling in the heart
Cardiac muscle:
- Rectangular shaped, striated muscle cells (myocytes) connect with each other via gap junctions, which are large pores (or channels) spanning the membranes of two cells, allowing ions to pass between them.
- This allows action potentials to spread between cells, which are organised in branched networks.
- Gap junctions are found in structures known as intercalated discs, which hold the cells together via desmosomes The site at which a gap junction is found is known as a nexus
The cardaic muscle cell:
- Cardiac muscle cells are highly specialised to contract rapidly in response to electrical stimulation.
- The cell is packed with myofibrils that run the length of the cell.
- The plasma membrane enclosing the cell dips into the cell at regular intervals, forming invaginations that bring the membrane close to intracellular organelles. These invaginations are known as t-tubules.
- The sarcoplasmic reticulum is present as a network that wraps around the myofibrils and the t-tubules, bringing all 3 structures close together. This is an important feature for the following reasons
- the sarcoplasmic reticulum is the main intracellular store of Ca2+
- Excitation at the membrane triggers the store to release its Ca2+
- The released Ca2+ is the trigger for muscle contraction.
excitation contraction coupling
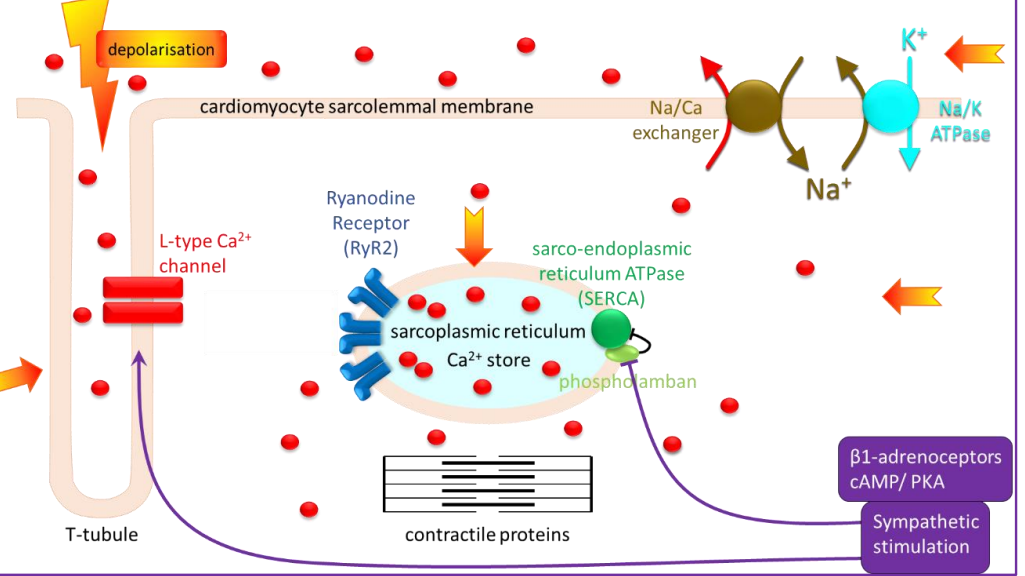
- the concentration of Ca ions in the cell cytoplasm is very low, but calcium is stored in organelles inside the cell at high concentrations.
- So in a cardiac myocyte, the Ca ion concentration is high outside the cell, low in the sarcoplasm and high in the sarcoplasmic reticulum. That is key to the mechanism of excitation-contraction coupling, which delivers Ca ions to the contractile proteins to trigger contraction.
- Calcium ions entering the cell across the membrane would therefore have to diffuse a long distance to reach the contractile proteins in the centre, which is not ideal for evoking a fast contraction. But the cells have evolved: At intervals along the cell, the membrane dips deep into the interior of the cell, forming invaginations known as t-tubules. Ion channels, including L-type Ca channels, are located in the t-tubular membrane, which brings them close to the sarcoplasmic reticulum located throughout the cell interior. The sarcoplasmic reticulum is, in turn, arranged around the contractile proteins. So, when the muscle cell fires an action potential, the Ca channels open and allow Ca ions to enter the cell.
- This Ca is not enough by itself to evoke contraction. Its main role is to bind to ryanodine receptors on the sarcoplasmic reticulum. These receptors are Ca-permeable ion channels that open in response to Ca binding.
- It is this Ca that binds to the contractile proteins, causing contraction. Ca levels in the cytoplasm must be rapidly restored to baseline between contractions. The main mechanism to remove Ca from the cytoplasm is the sarco-endoplasmic reticulum pump or SERCA. It pumps Ca against the concentration gradient back into the sarcoplasmic reticulum, allowing the cell to relax and replenishing the store until the next action potential arrives.
- Ca is also removed from the cell, mainly by the Na/Ca exchanger. This exchanger makes use of the large transmembrane gradient of Na that facilitates its entry and is set up by the Na/K ATPase or Na pump. It has at least one additional effect that also contributes. It inhibits the activity of a protein known as phospholamban. This protein normally has an inhibitory effect on SERCA. Sympathetic stimulation removes that inhibition and increases SERCA activity, resulting in a higher rate of Ca accumulation into the sarcoplasmic reticulum. This means that as the heart beats faster and stronger, it also relaxes faster between beats with a larger store of Ca available for the next beat. Both effects of sympathetic nerve stimulation are mediated by noradrenaline and adrenaline acting at beta 1 adrenoceptors, through the cAMP protein kinase A pathway.
Sliding filament theory of contraction:
- The contractile proteins actin and myosin interact to form crossbridges and generate force.
- The protein filament, tropomyosin, lies between actin and myosin filaments, blocking their interaction.
- When Ca2+ binds to the regulatory protein troponin, it pulls tropomyosin away from the myosin-binding sites on actin, allowing myosin to attach.
- The head of myosin is a molecular motor that uses ATP hydrolysis to pivot, bend and pull on actin to generate a power stroke.
- Myosin detaches from actin when a new ATP molecule binds, which is then hydrolysed for the next power stroke when actin is exposed.