🌡chem FULL UNIT 4 NOTES (sem 2)
4.1 - States of Matter and Phase Changes
Things the LTC had on it:
* diffrence between heat and temp
* solids liquids and gaes
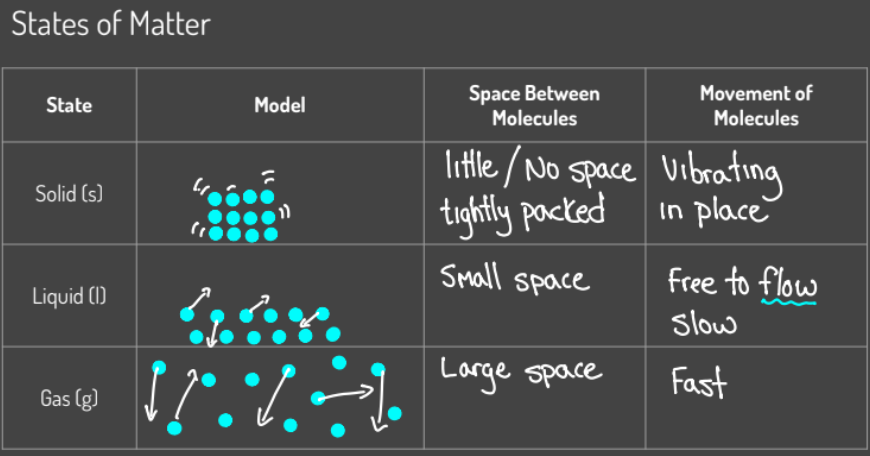
* know the diagrams for each of solids liquids and gases (the particle movement and if the movement is fast or not.
Heat and Temp
Heat: Heat is the thermal energy transferred between a hot substance to a cool one.
Temperature: Temperature measures the average kinetic energy of particles in a substance.
Solids, Liquids and Gases
gas to a solid: desposition
solid to a gas: sublimation
solid to a liquid: melting
liquid to solid: freezing
liquid to gas: vaporization
gas to liquid: condensation
Types of substances that you need heat energy
Vaporization, Melting and Sublimation need heat energy
Freezing, Despotion, and Condensation don’t need heat energy
Intermolecular forces
they change when going from a liquid, gas to a solid
NOTE TO SELF: look back on notes on paper about intermolecular forces.
4.2 Heating and Cooling Curves
Practice doing the calculations…in cooling curves the delta T and delta H become negative
4.21 Heating Curve of Water
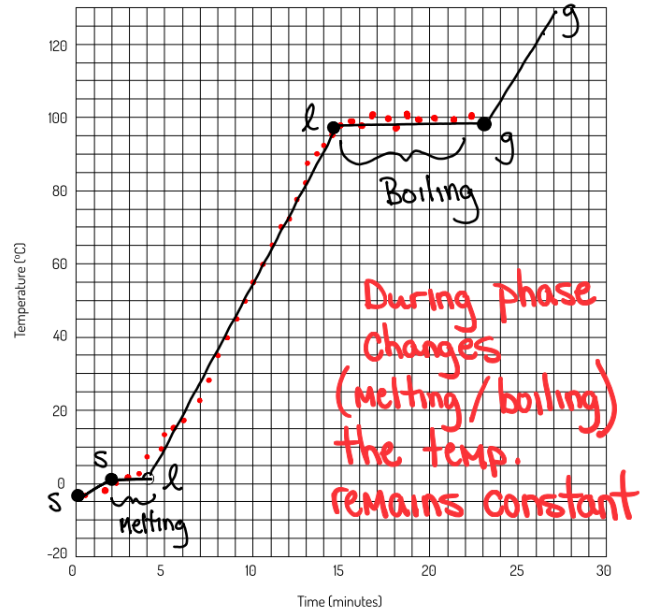
water boils at 100 deg C
water either melts or freezes at 0 degrees C
when going from a liquid to a gas the particles move faster
4.22
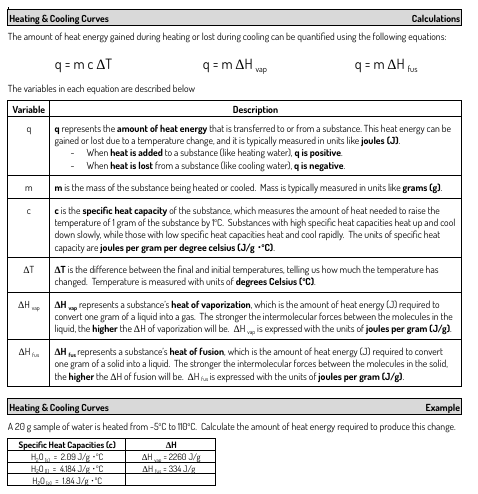
NOTE: see in-class notes for calculations…the cooling curve practice is on the back