Oct 16th - Small molecule transport
water movement is directed by ion movement
water has a higher membrane permeability than many other molecules, but it is not adequate for rapid movement
water direction is controlled by relative concentration of solutes
aquaporins are water channels that accelerate water flow across membrane
more aquaporins in cells that need higher water exchange
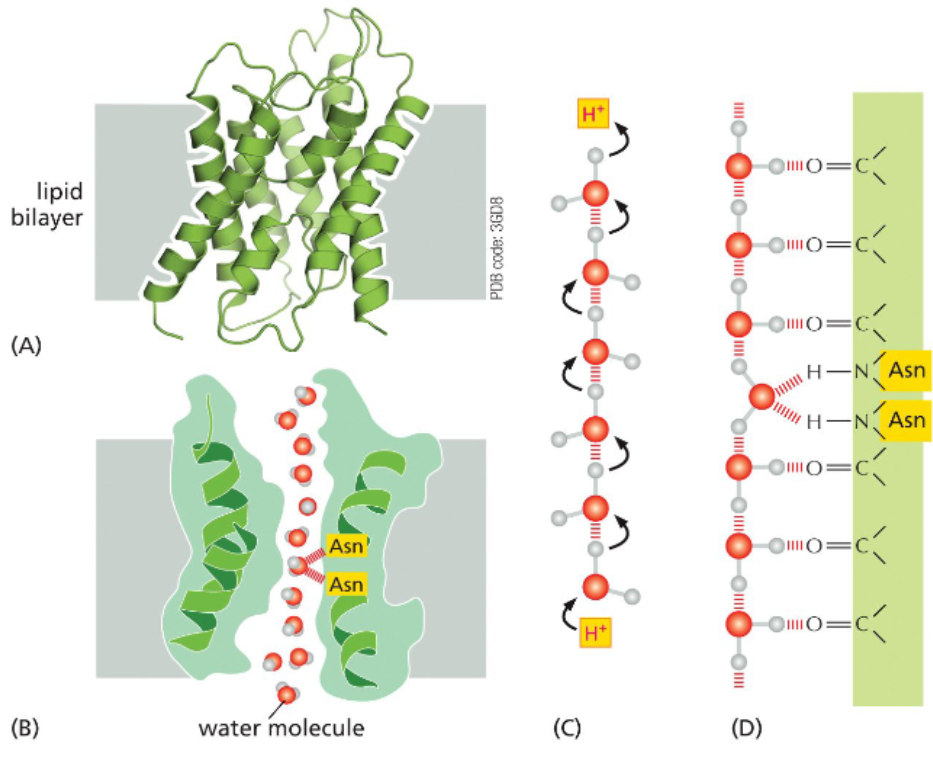
aquaporins move water molecules single-file across lipid bilayer
each pore of an aquaporin is composed of seven transmembrane helices that assemble around a hydrophilic channel
the channel is too narrow for hydrated ions but wide enough for water molecules to pass through single-file
a line of water molecules can sequentially pass protons between them so without a mechanism to prevent it, aquaporins could serve as proton channels
a pair of asparagine’s present in the channel transiently hydrogen bond with single water molecules as they pass by — this occupies the molecule’s electrons and prevents proton shuttling
ion channels fluctuate between open and closed states
the state of an ion channel, as open or closed, is based on the conformation of the protein
some channels rapidly alternate between open and closed, others may spend more time than open
other channels can be biased towards their open state by different signals
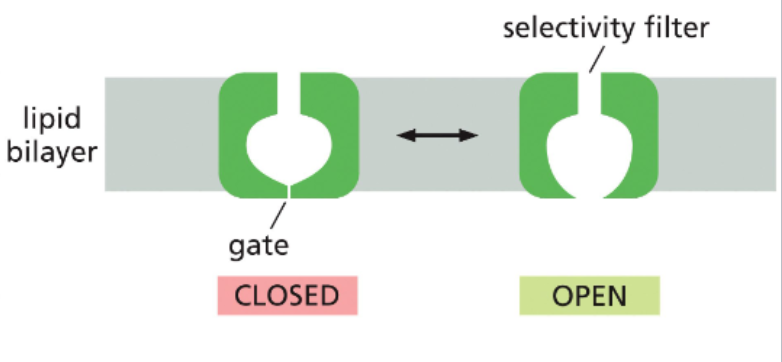
ion channels can be gated by different factors
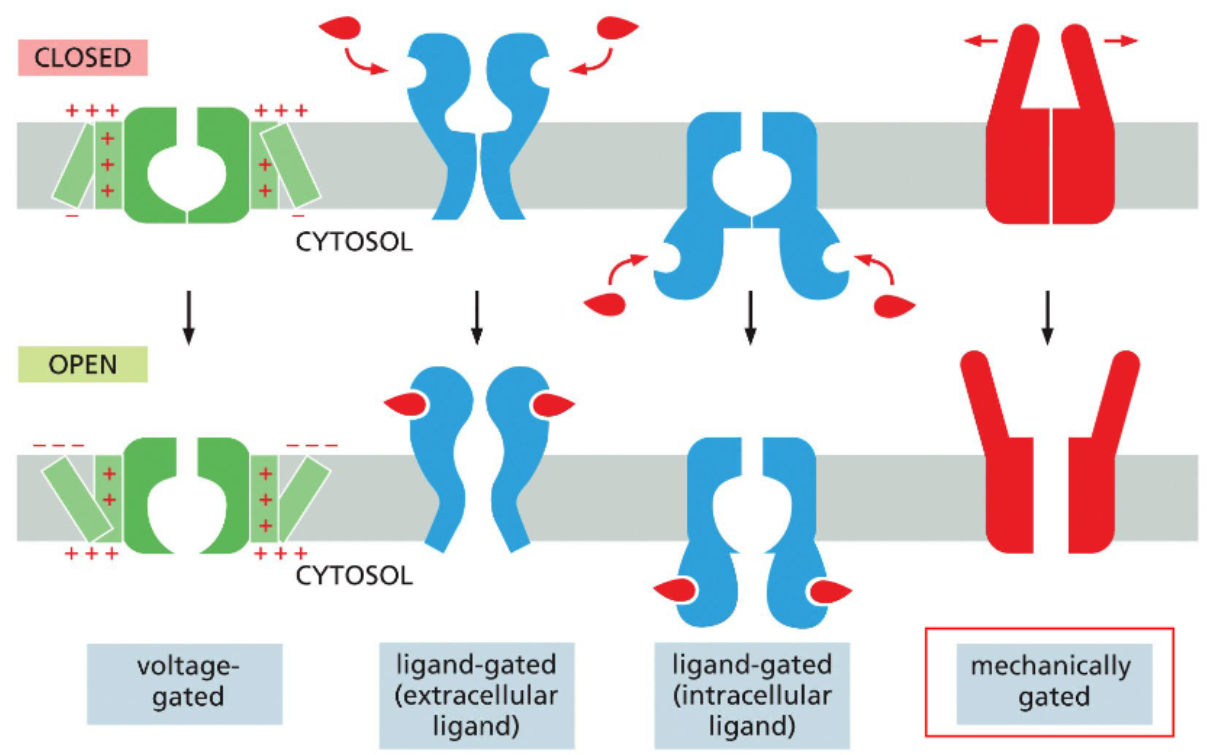
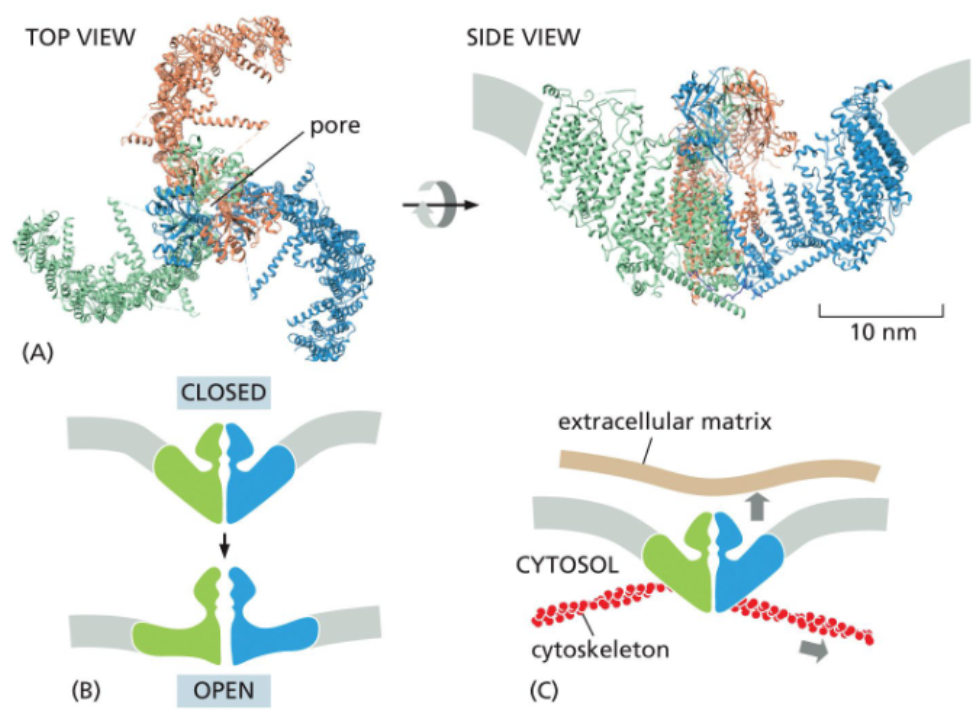
some mechanically-gated channels open via membrane deformation
a mechanism of mechanically-gated channels involves three large domains that spread into the plasma membrane and deform it into a dome
forces on the membrane flatten the dome and pull the channel open
forces can be experience from both outside and inside the cell
mechanically-gated channels are the basis of the senses of the touch & hearing
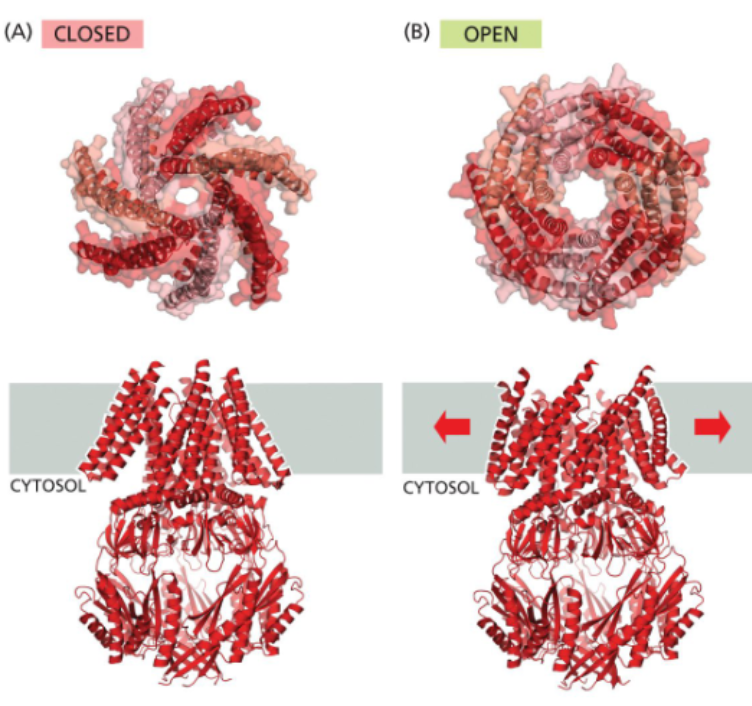
bacteria also express mechanically gated channels
small and large conductance mechanosensitive channel are bacterial mechanically-gated channels that allow passage of a variety of solutes
when internal pressure builds in a bacterium, the cell swells, stretching the membrane and pulling the Msc open
the non-selective Mscs allow solutes to leave the cell, restoring osmotic balance and preventing the cell from bursting
unequal ion distribution establishes membrane potential
when there is an imbalance of positive and negative charges on either side of a membrane, the excess cations and anions will be attracted to each other at the membrane — the rest of the fluid is electrically neutral
the small number of ions concentrated at the membrane means very few ions must move to alter membrane potential
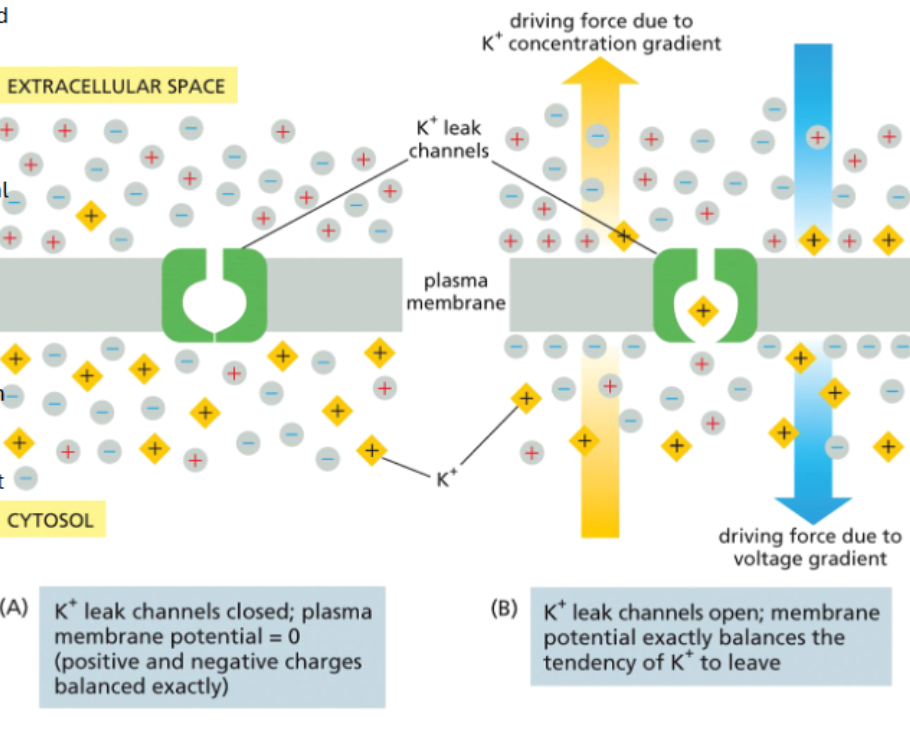
potassium leak channels are a major driver of membrane resting potential
leak channels are ion channels that open and close randomly
Na+ leak channels operate at around 5% the activity of K+ leak channels
because these two ions, K+ has a stronger influence on resting membrane potential
if all charges are balanced but there’s more K+ inside the cell
if K+ leak channels are added, K+ will leave
the exiting positively charged K+ makes the inner leaflet more negative
as the membrane potential becomes more negative, it becomes more difficult for K+ to leave — this creates a K+ driven resting potential
K+ leak channels and Na-K pumps that move Na+ out and K+ in maintain a resting potential of around -70mV
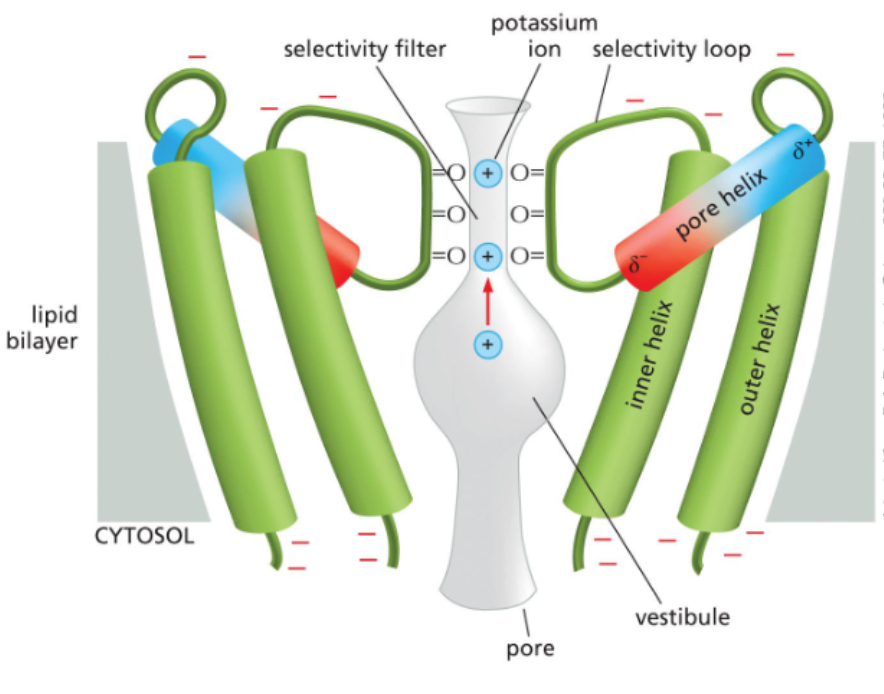
ion channels have water- based selectivity filters
most ion channels are selective
a Na channel won’t allow a K+ ion to cross and vice versa
ions enter a channel at a wide opening, the vestibule
the selectivity filter is a narrow region that interacts with ions based on their size
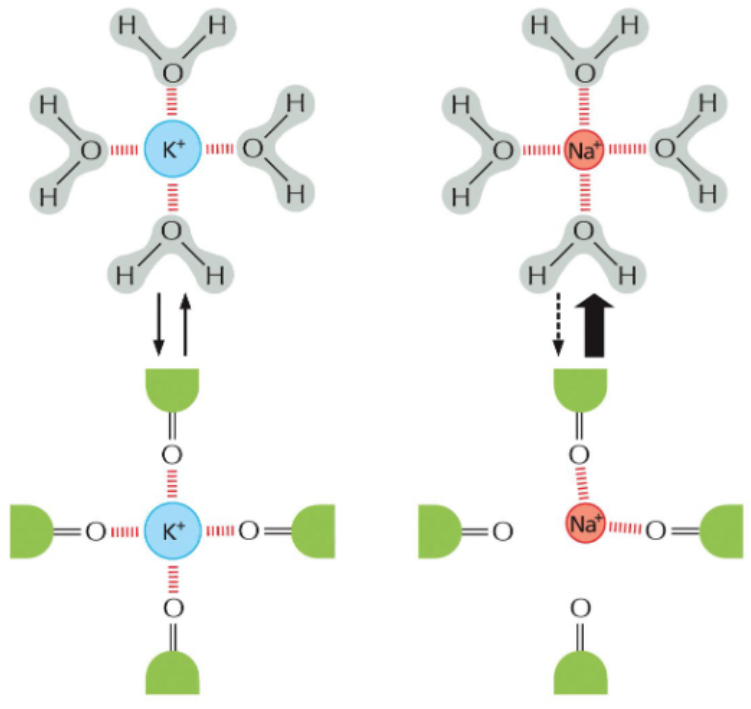
the selectivity filter is based on position of polar amino acids in the channel vestibule
the selectivity filters are larger than a naked ion, but smaller than an ion with a hydration shell
the radius of K+ ions is large enough to interact with four carbonyls in the selectivity filter
the smaller radius of Na+ ions can only interact with two carbonyls in the selectivity filter — hydration shell removal is less removal and hydrated Na+ is too big to pass through the channel
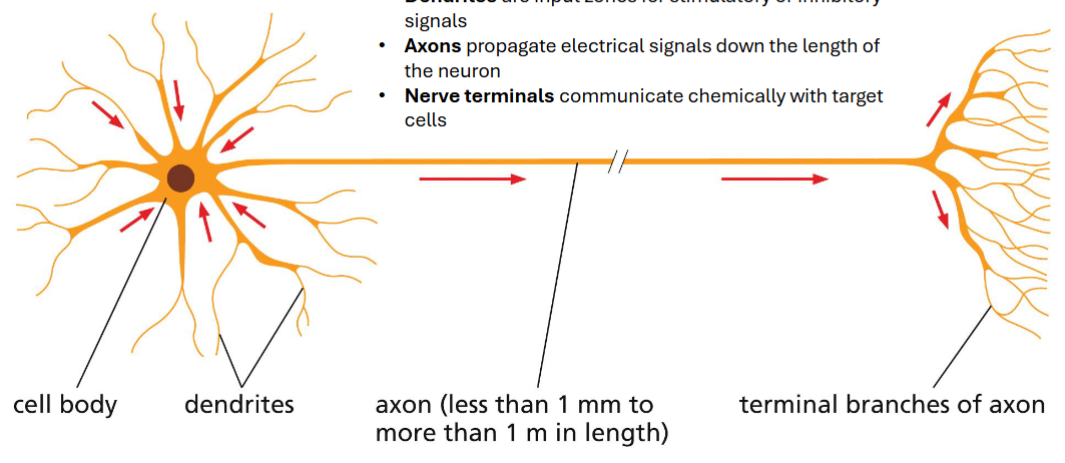
neurons operate through transient localized alterations in membrane potential
the cell body contains the nucleus and most organelles
dendrites are input zones for stimulatory or inhibitory signals
axons propagate electrical signals down the length of the neuron
nerve terminals communicate chemically with target cells
voltage-gated channels are controlled by membrane potential
voltage-gated channels have membrane potential sensing helices that are enriched in positively charged amino acids within a polar space that is open to the cytosol
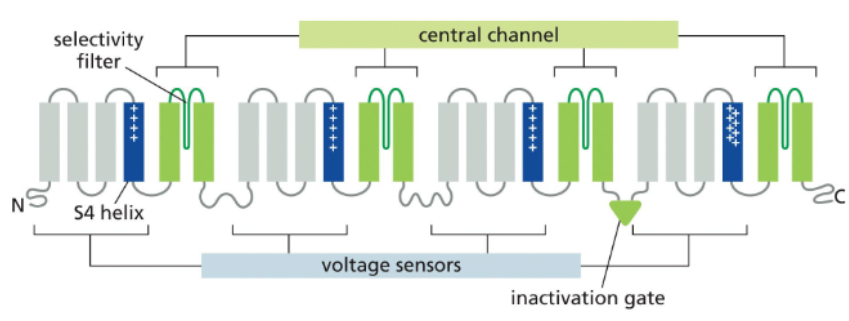
voltage-gated channels are controlled by membrane potential
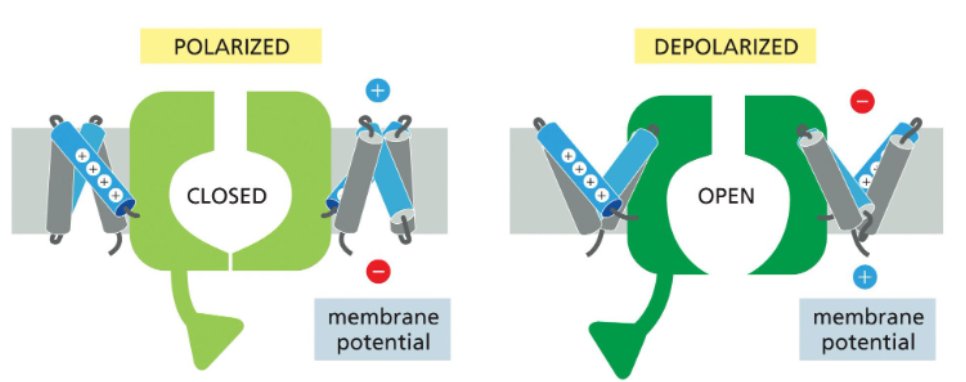
at resting potential, the inner leaflet is more negative the outer leaflet — the voltage-sensing helices face towards it
with depolarization, the outer leaflet becomes positive; this attracts the voltage-sensing helices which rotate towards it
rotation of the voltage-sensing helices pull the channel into an open conformation
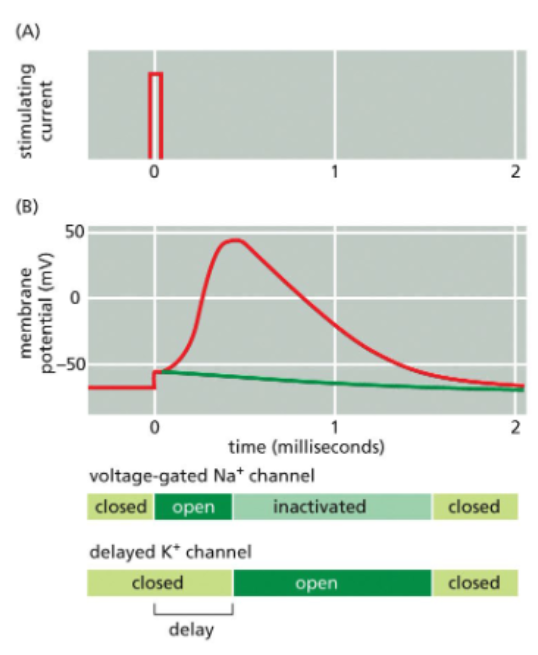
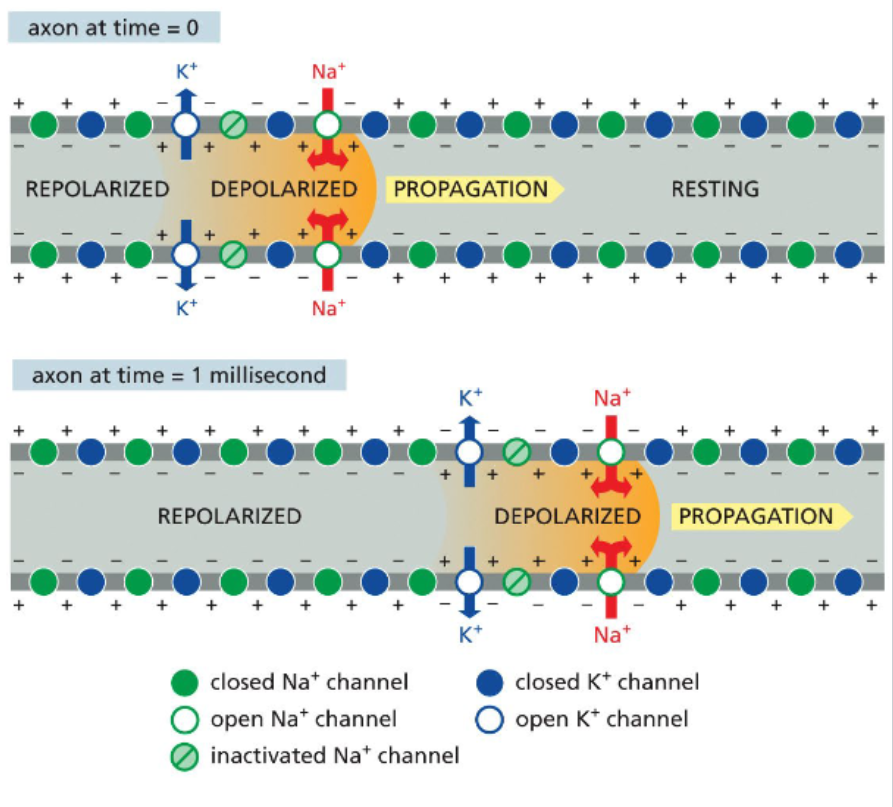
voltage-gated Na+ channels cycle between closed, open, and inactivated states
at sub-threshold membrane potentials, voltage-gated Na channels are closed
with sufficient depolarization, the channels opens
an influx of Na cations produces an action potential
at the peak of the action potential, a separate domain of the channel called the inactivation gate swings into the channel and blocks Na+ flow
during the refractory period, the inactivation gate remains in place
after repolarization, the gate resets
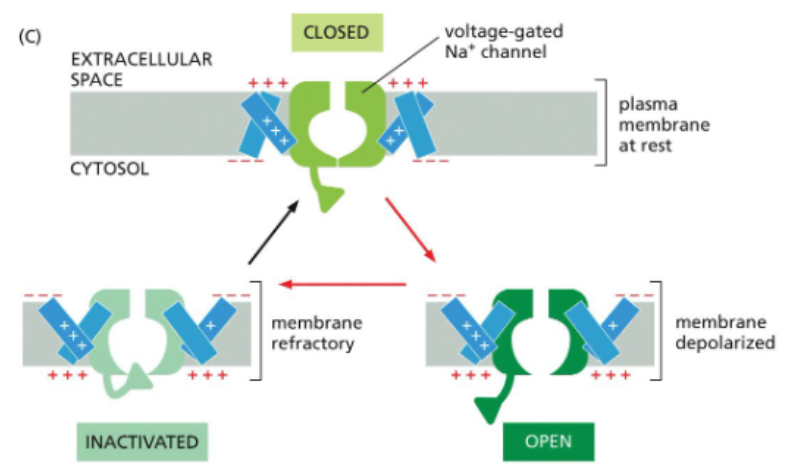
action potentials propagate in one direction down axons
the inactivation gate ensures that a voltage-gated Na channel that has just closed does not immediately reopen
by the time a voltage-gated Na+ channel resets, the action potential is to far away to influence it
this keeps the action potential moving in one direction
K+ channel opening restores the membrane potential
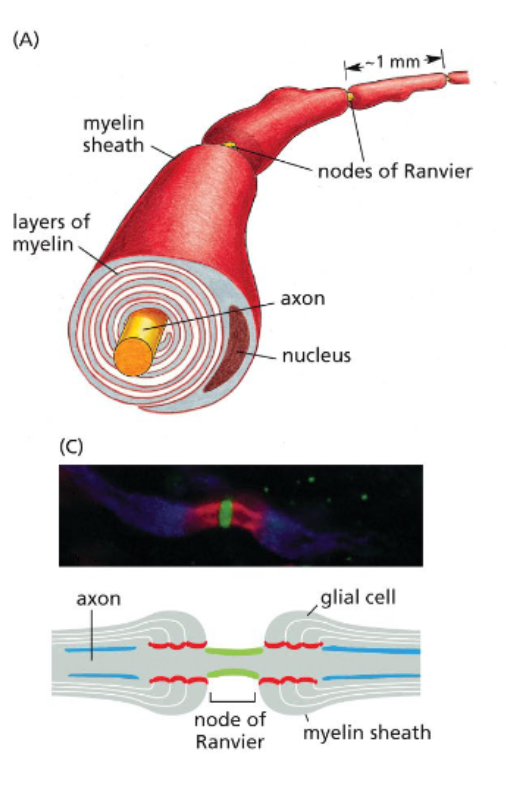
neurons are wrapped in electrically insulating myelin sheaths
most neurons only expose a small portion of their plasma membranes
myelin sheaths are wrapped plasma membranes provided by either Schwann cells or oligodendrocytes, surrounds axons and prevent ion exchanges
Nodes of Ranvier are small, non-myelinated regions where action potentials form
because action potentials form only at nodes of Ranvier, current appears to jump down the axon — saltatory conduction
ions can diffuse through the cytosol between nodes of Ranvier
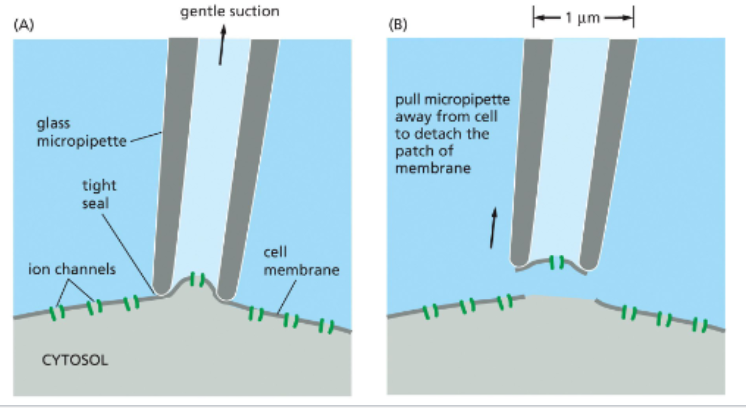
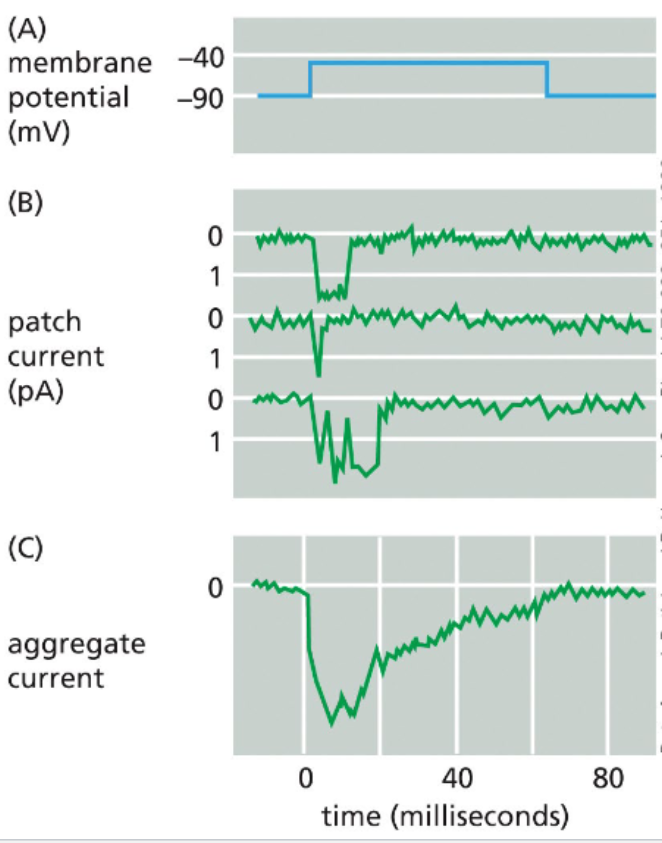
patch clamps measure current across isolated membrane regions
since a single cell can have many different ion channels on its surface, it is difficult to measure an individual ion channel’s function with whole cells
patch clamping uses a fine glass pipette and suction to detach a small patch of membrane and a single ion channel
channels can also be isolated on intact cell membranes
the patch is next immersed in an electrolyte bath
electrodes are placed in the bath & pipette — fluid inside the pipette is isolated from the fluid in the bath such that electrical current is only possible when the ion channel is open
treatments can be added to the bath or pipette and the effect on channel opening can be measured via detection of electrical current