24/4 Energy Production and Electron Transport Chain
Recap and Introduction
The lecture recaps the course's progress, focusing on energy production and transfer to sustain life.
The primary macronutrients discussed are carbohydrates and fats, including digestion, energy extraction via oxidation, and resynthesis.
The production of carbon dioxide (CO2) during the TCA cycle is noted, linking it to respiration.
The next three lectures will explain the origin of water produced during respiration.
Differences between fats (hydrophobic) and carbohydrates (hydrophilic) are highlighted.
Protein structure, enzyme function (allosteric regulation, phosphorylation), membrane proteins, and transporters have been covered.
Future lectures will cover proteins (by Stefan) and delve deeper into ATP production and the electron transport chain.
ATP Production Overview
The lecture will focus on ATP production, the electron transport chain, and water generation.
Topics include substrate-level phosphorylation, the electron transport chain, ATP structure, its role in energy provision, and production challenges.
The need for a more efficient system (oxidative phosphorylation) to sustain life is emphasized.
Electron-carrying molecules and their role in capturing and transferring electrons will be discussed.
Understanding Energy (Watt)
Energy is commonly measured in watts (rate of energy production/consumption).
In the context of food, energy is measured in calories (American term) or kilojoules.
Watts relate to a rate (with a time component), while kilojoules indicate the total energy in a food source.
A typical light bulb consumes about 1,700 watts, similar to the energy required for a person at rest.
Professional cyclists may require up to 400 watts or more during races.
Example calculation: A two-hour race at 400 watts requires .
Average daily energy intake: 8,500 to 10,000 kilojoules, depending on body weight.
Conversion: 1 kilocalorie (calorie) = 4.18 kilojoules.
ATP: The Energy Currency
Energy is stored and utilised in the form of adenosine triphosphate (ATP).
ATP consists of an adenine base, a sugar group (ribose), and three phosphate groups (has charge repulsion between phosphates).
ATP is inherently unstable due to charge repulsion among the phosphate groups.
It has a high phosphoryl transfer potential, meaning it readily transfers a phosphate group to other molecules, releasing energy.
This energy transfer drives changes, such as protein conformational changes, enabling them to perform functions.
Challenges in ATP Synthesis
ATP synthesis is energetically unfavorable, requiring .(even hi
Cells maintain a high ATP:ADP ratio (1000:1), and a near 1:1 ratio indicates cell death.
Glycolysis generates ATP through reactions catalyzed by phosphoglycerate kinase and pyruvate kinase.
These reactions use molecules like 1,3-bisphosphoglycerate and phosphoenolpyruvate, which have a higher energy potential (hence, they can efficiently donate phosphate groups to adp to form atp).
A large negative associated with these reactions drives ATP production.
delta g reactions naturally proceed without external energy input
energy rich molecs can efficiently transfer phosphate groups to adp to form atp
Substrate-Level Phosphorylation
Glycolysis exemplifies substrate-level phosphorylation.
The creatine kinase system in muscles maintains ATP levels.
Creatine phosphate, a high-energy compound, facilitates ATP production from ADP via creatine kinase.
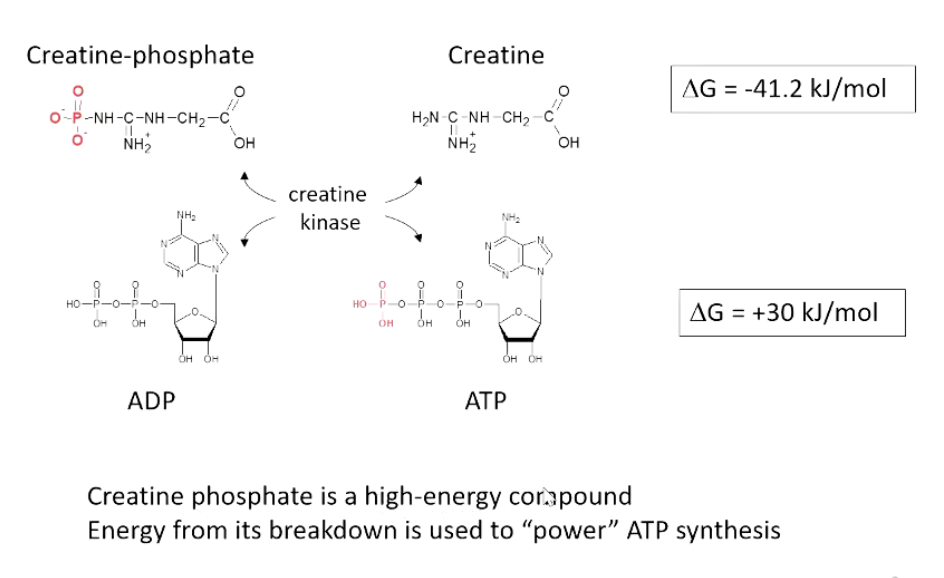
adp + creatine phosphate = atp +creatine
creatine phosphate to creatine = delta g -41.2
adp to atp = delta g +30
During intense activity, ATP is quickly depleted, followed by creatine phosphate.
Creatine phosphate can sustain energy needs for about a minute.
Creatine phosphate is replenished from ATP during rest, forming a cycle.
Limitations of Substrate-Level Phosphorylation (transfer phosphate group, inefficient)
Glycolysis (anaerobic metabolism, oxygen idependent) produces ATP and lactate.
For sustained ATP production, aerobic metabolism (oxidative phosphorylation) is required.
Inefficiency: Substrate-level phosphorylation is inefficient because it relies on precursor molecules.
It requires high-energy phosphate donors, which must be synthesized.
It yields one mole of ATP per mole of phosphate donor, which is insufficient for cellular needs.
Cells use a complex, efficient system (oxidative phosphorylation) for large-scale ATP production.
phospho creatine (PCr) and glycolysis are both severely limited/dependent on the availability of PCr and NAD+ respectively
Oxidative Phosphorylation
Topics include high-energy intermediates, their transport, the electron transport chain, proton transport, and the proton-motive force.
Metabolites are funneled to the mitochondria: Carbohydrates yield pyruvate, fatty acids enter via the carnitine shuttle, and both are metabolized to acetyl CoA.
Oxidation produces electron carriers NADH and FADH2.
Oxidation involves electron loss, stored in NADH and FADH2; reduction involves electron gain.
These carriers donate electrons to molecules with a greater affinity, forming an electron transport chain.
The final electron acceptor is oxygen, which is reduced to water.
Electron Transfer: Electrons from NADH and FADH₂ (produced in glycolysis and the citric acid cycle) travel through the ETC.
Proton Gradient Formation: As electrons move, the ETC pumps protons (H⁺) across the mitochondrial membrane, creating a gradient.
ATP Synthesis: The enzyme ATP synthase uses this gradient to drive the formation of ATP from ADP and inorganic phosphate.
Oxygen’s Role: Oxygen acts as the final electron acceptor, combining with electrons and protons to form water.
Electron Carriers: NADH, FADH2, Ubiquinone, and Heme Groups
NAD - nicotinamide adenine dinucleotide, NADH - reduced form
NADH and FADH2 (flavin adenine dinucleotide) store and transfer electrons generated during oxidation reactions.
Structure and function of NAD+ and NADH are described; NADH carries two electrons in the form of a hydride ion.
NADP+ / NADPH is associated with synthesis reactions, such as fatty acid synthesis, where NADPH provides electrons.
FAD (flavin adenine dinucleotide) also accepts two electrons and a proton.
Ubiquinone (coenzyme Q) is a mobile electron carrier within the membrane due to its hydrophobic isoprenoid tail.
It accepts electrons in two steps, forming an unpaired electron intermediate before becoming ubiquinol.
Porphyrin electron acceptors (heme groups) carry oxygen in blood and are bound to proteins as prosthetic groups.
Iron at the center of heme groups accepts and transfers electrons, switching between ferric (Fe3+) and ferrous (Fe2+) states.
Iron-sulfur clusters within proteins coordinate iron via cysteine residues, facilitating electron transfer.
These molecules, generated in glycolysis and the citric acid cycle, contain high-energy electrons.
NADH donates electrons to Complex I of the electron transport chain (ETC), while FADH₂ feeds into Complex II
Electrons move sequentially through Complex I → III → IV, passing through intermediate carriers like ubiquinone (CoQ) and cytochrome c.
Each transfer releases energy, which is used to pump protons (H⁺) across the mitochondrial membrane, creating a gradient.

Complex I of the Electron Transport Chain
Complex I (NADH dehydrogenase) is a membrane protein that transfers electrons from NADH to ubiquinone.
NADH is oxidised, and ubiquinone is reduced to ubiquinol.
NADH produced during the TCA cycle and beta-oxidation delivers electrons to Complex I.
Electrons are transferred from NADH to flavin mononucleotide (FMN), a prosthetic group within the protein.
Electrons then pass through a series of iron-sulfur clusters within the protein.
Ubiquinone, located within the protein, accepts electrons from the iron-sulfur clusters and is reduced to ubiquinol.
Electron transfer causes conformational changes in the protein, resulting in the movement of protons from the matrix to the intermembrane space. (The intermembrane space now has high H⁺ concentration, while the matrix has low H⁺ concentration.)
Chemiosmosis and ATP Synthesis
Oxidation of NADH by Complex I results in the transport of four protons across the membrane.
This proton movement generates a charge difference and a pH difference across the membrane.
The next lecture will cover the remaining complexes of the electron transport chain and how they contribute to proton movement and ATP synthesis.
Oxidation of metabolites (glucose, fatty acids, amino acids) leads to the production of high-energy electrons and CO2.
Electron acceptors facilitate electron movement via the electron transport chain, ultimately driving ATP synthesis.
Based on the lecture recap, prioritize these key areas for your exam:
Energy Production and Transfer: Focus on how energy is produced and transferred to sustain life, particularly:
Digestion, energy extraction (oxidation), and resynthesis of carbohydrates and fats.
The significance of production during the TCA cycle and its link to respiration.
Differences between hydrophobic fats and hydrophilic carbohydrates.
ATP Production: Understand the processes involved in ATP production and its challenges:
ATP structure, its role in energy provision, and the challenges in its production.
Substrate-level phosphorylation (e.g., glycolysis and the creatine kinase system).
Limitations of substrate-level phosphorylation and the need for oxidative phosphorylation.
Oxidative Phosphorylation: Concentrate on ATP synthesis through this process, including:
The electron transport chain, proton transport, and the proton-motive force.
The roles of NADH and FADH2 as electron carriers.
The final electron acceptor being oxygen, which is reduced to water.
Electron Carriers: Understand the function and types of electron carriers:
NADH and FADH2: How they store and transfer electrons.
Ubiquinone (coenzyme Q): Its role as a mobile electron carrier.
Heme groups: How they carry oxygen and facilitate electron transfer via iron.
Complex I of the Electron Transport Chain: Study the specifics of Complex I:
Its function in transferring electrons from NADH to ubiquinone.
The roles of flavin mononucleotide (FMN) and iron-sulfur clusters.