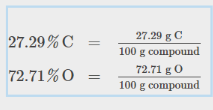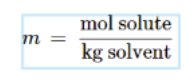The Mole
Mole = unit amount (ex. dozen, couple…)
Avogadro’s Number
molecules/atoms per mole
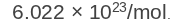
Molar Mass: mass in grams of 1 mole of __ substance (compound or element)
1 moles of any elements contain the same number of atoms; the masses of 1 mole of different elements are different
Molar Mass = Atomic/Formula mass in amu
Mass —> Moles and vice versa
same with compounds, divide by total molar mass

Number of Atoms
multiply mole number by avogadro’s number
same with compounds
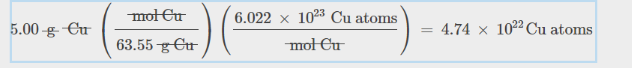
for number of Carbon atoms in this comopund
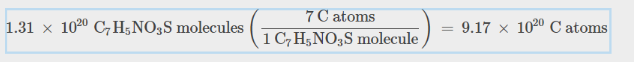
Standard Temperature and Pressure
273 Kelvins (standard Temp)
1 atm, 101.325 kPa (standard Pressure)
1 mole = 22.4 L (standard Molar Volume)
Ideal Gas Law
Gases described by this law exhibit ideal behavior; when given any 4 of the terms below the 5th will be able to be found
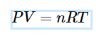
P = gas pressure
V = gas volume
T = temperature in Kelvins
R = ideal/universal gas constant
n = number of moles of gas
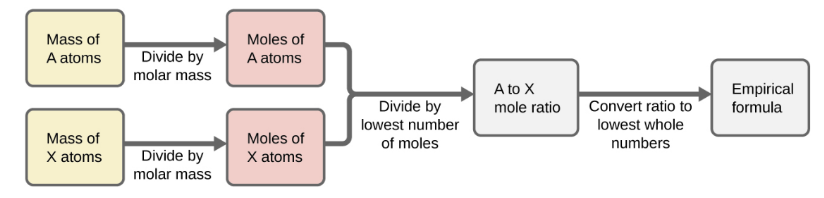
Empirical Formulas
molar amounts used to compute whole-number ratios to derive empirical formulas of substances
convert from grams to number of moles
Divide each subscript by the smaller subscript
Empirical formula (should end with whole number subscript, if not close multiple to get whole number)
Percent Compositions with Empirical Formulas
Convert percentages to out of 100 g
Convert to Moles, then find ratio
The result her eis: CO2
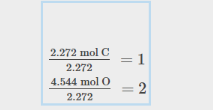
Derivation of Molecular Formulas
comparing molecular/molar mass to empirical formula mass for the number of formula units per molecule
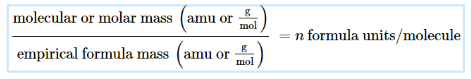
then applied to each subscript, determine molecular formula

Solutions
Concentration: relative amount of a given solution component
Solvent: the medium the other component(s) are dissolved in
Aqueous Solution: solution where water is the solvent
Solute: the component being dissolved (described with dilute and concentrated)
Molarity: number of moles of solute per liter of solution
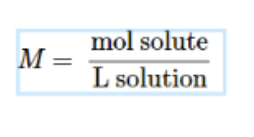
In a solute, calculate the number of moles (conversion) over number of Liters

Mass of NaCl given molarity and volume

Molality: ratio of number of moles of solute to the mass of solvent in kg