Orders of reaction
Orders of reaction
how concentration of a reactant affects rate
Half life
the half life of a reaction is the time taken from concentration to decrease by a half
the second half life is the time taken from the end of the first half life
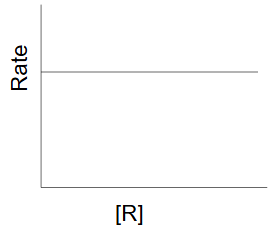
Zero order reaction
as the concentration of the reactant decreases the rate of reaction remains constant and the reactant decreases at a constant value
rate = k
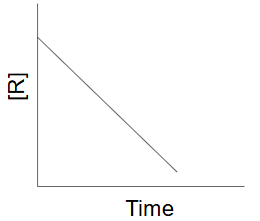
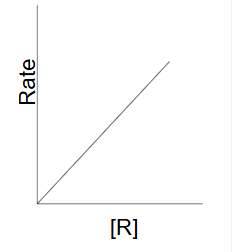
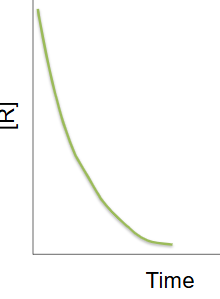
First order reaction
as the concentration of the reactant decreases the rate of change of [R] decreases
rate = k[R]
half lives are identical
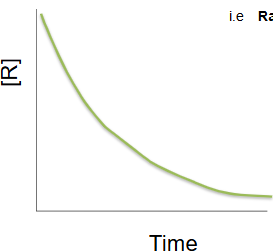
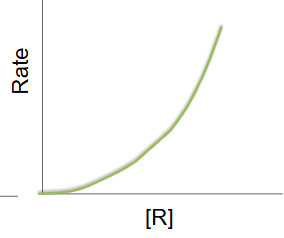
Second order reaction
as the concentration of the reactant decreases the rate of reaction changes rapidly
rate = k[R]²
Rate equation
for A + B + C —> products
rate is directly proportional to [A] which is directly proportional to [B] which is directly proportional to [C]
rate = k[A]^0 x [B]^1 x [C]²
k = rate constant
where the order is determined experimentally from reactant rate/concentration graph