Section IX - exam 3
Bold face terms
Maxwell’s equations: man showed in 1865 that electrical phenomena were related to magnetic phenomena. These equations describe how electric fields and magnetic fields interact and propagate through space.
Magnetic monopoles: hypothetical particles that are proposed to exist as isolated magnetic charges, differing from the dipole structure of conventional magnets, where each magnet has both a north and a south pole.
Self propagate: the ability of a wave to maintain its shape while traveling at a constant speed in a medium, indicating that a disturbance in the field can travel without the need for any external influence. Maxwell’s last 2 laws allow electromagnetic waves to do this.
Speed of light: referred to as ‘c’ since visible light is a form of electromagnetic radiation.
it is impossible for anything with mass, even an electron, to be accelerated to the speed of light.
photon: a quantum of electromagnetic radiation, specifically a particle of light that carries energy and has no mass, allowing it to travel at the speed of light in a vacuum. Planck described a packet of electromagnetic radiation as this in the early 1900s, this term has been used ever since to describe E/M radiation.
Photoelectric effect: if a photon hits a certain type of metal, it can knock an electron off of an atom in the metal and produce an electric current.
Alpha particle: have positive charge and were later determined to be He nuclei. as such, He nuclei are referred to as this.
Beta particles: have negative charge and were later found to be the simple electron.
Gamma particles: have no charge and were later realized to be high energy photons. these particles were renamed this and represent the highest energy form of E/M radiation.
E/M spectrum: (and spectra in general) is defined to be the intensity, or flux, of an E/M wave as a function of wavelength, frequency, or energy. also called the continuum.
Gamma rays - E/M spectrum: highest energy, highest frequency, and shortest wavelengths that are less than 0.01 nm. These types of photons are given off by nuclei during high-energy nuclear reactions and by extremely hot gas (T > 3.0 × 108 K — over 300 million degrees Kelvin!).
X - rays - E/M spectrum: 0.01 nm < λ < 10 nm. These photons are given off by low-energy nuclear reactions and very hot gas (3.0× 105 K < T < 3.0 × 108 K).
Ultraviolet (UV) - E/M spectrum: 10 nm < λ < 400 nm. These photons are given off by electron transitions in atoms, typically involving the ground state and by hot gas (7000 K < T < 3.0 × 105 K).
Visible (visual) - E/M spectrum: 400 nm < λ < 700 nm. These photons are given off by electron transitions in atoms and warm gas (4000 K < T < 7000 K).
Infrared (IR) - E/M spectrum: 700 nm < λ < 1 mm. These photons are given off by electron transitions in atoms (between excited levels) and molecules. Cool gas (3 K < T < 4000 K) can also thermally emit these photons.
Microwaves - E/M spectrum: 1 mm < λ < 10 cm. These photons are given off by electronic devices or from cold gas (0.03 K < T < 3 K).
Radio waves - E/M spectrum: λ > 10 cm. These photons have the lowest energy, lowest frequency, and longest wavelengths and can be created by electrical circuits and from extremely cold gas (T < 0.03 K). The longest radio waves (λ > 1 km) are sometimes just referred to as long waves.
Kirchhoff’s law #1: A luminous opaque object (solid, liquid, or gas) emits light at all wavelengths (E/M spectrum), thus producing a continuous spectrum — a complete rainbow of colors without any spectral lines.
Kirchhoff’s law #2: A rarefied luminous gas emits light whose spectrum shows an emission-line spectrum — a series of bright spectral lines against a dark background.
Kirchhoff’s law #3: If white light (i.e., a continuum) from a luminous source passes through a lower-density gas, an absorptionline spectrum appears — a series a dark spectral lines among the colors of the continuous spectrum.
ideal or perfect radiator:
a) A hypothetical body that completely absorbs every kind of E/M radiation that falls on it.
b) This absorption continues until an equilibrium temperature is reached.
c) At that point, all incoming radiation is immediately reradiated away as soon as it is absorbed (note that this is not the same as being reflected).
d) referred to as a blackbody.
energy flux: F (defined as the energy emitted per unit area each second and related to the intensity) that is radiated away which is proportional to the 4th power of temperature
Luminosity: the flux integrated over all of the surface of the object. For a spherical object, the surface area is 4π R2 , where R is the radius of the spherical blackbody, so the luminosity is L = 4π R^2F = 4π σ R^2 T^4
Wien’s displacement law: the peak wavelength of radiation emitted by a blackbody is inversely proportional to
its temperature.
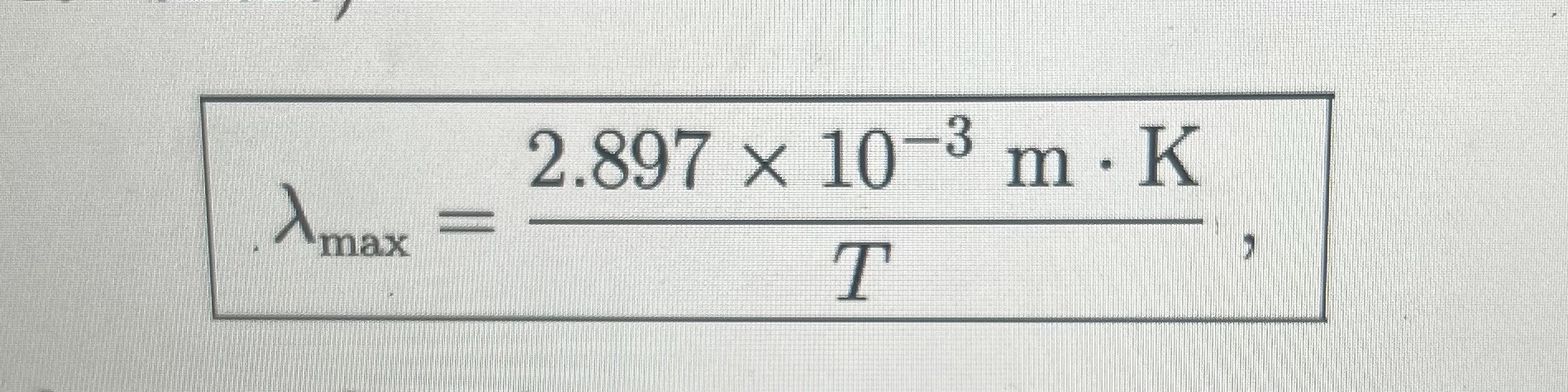
Planck curves: describe the distribution of electromagnetic radiation emitted by a blackbody at a given temperature, illustrating how intensity varies with wavelength and temperature.
Spectral lines: Discovered Jospeh von Fraunhofer in 1814, are the distinct dark or bright lines seen in a spectrum, corresponding to specific wavelengths absorbed or emitted by elements, providing crucial information about the chemical composition and physical conditions of stars and other celestial bodies.
Spectral analysis: Discovered Gustav Kirchhoff and robert Bunsen in the mid-19th century, this method involves breaking light into its component colors to analyze the spectra of various substances, allowing scientists to identify elemental compositions and gain insights into the properties of stars and other astronomical phenomena.
Spectrograph or spectroscope: A device that breaks white light into its component colors, hence displaying spectral lines if they are present.
Quantum mechanics: Electrons can only orbit nuclei in allowed orbits or states. This is the basis of the branch of modern physics called