Antibodies
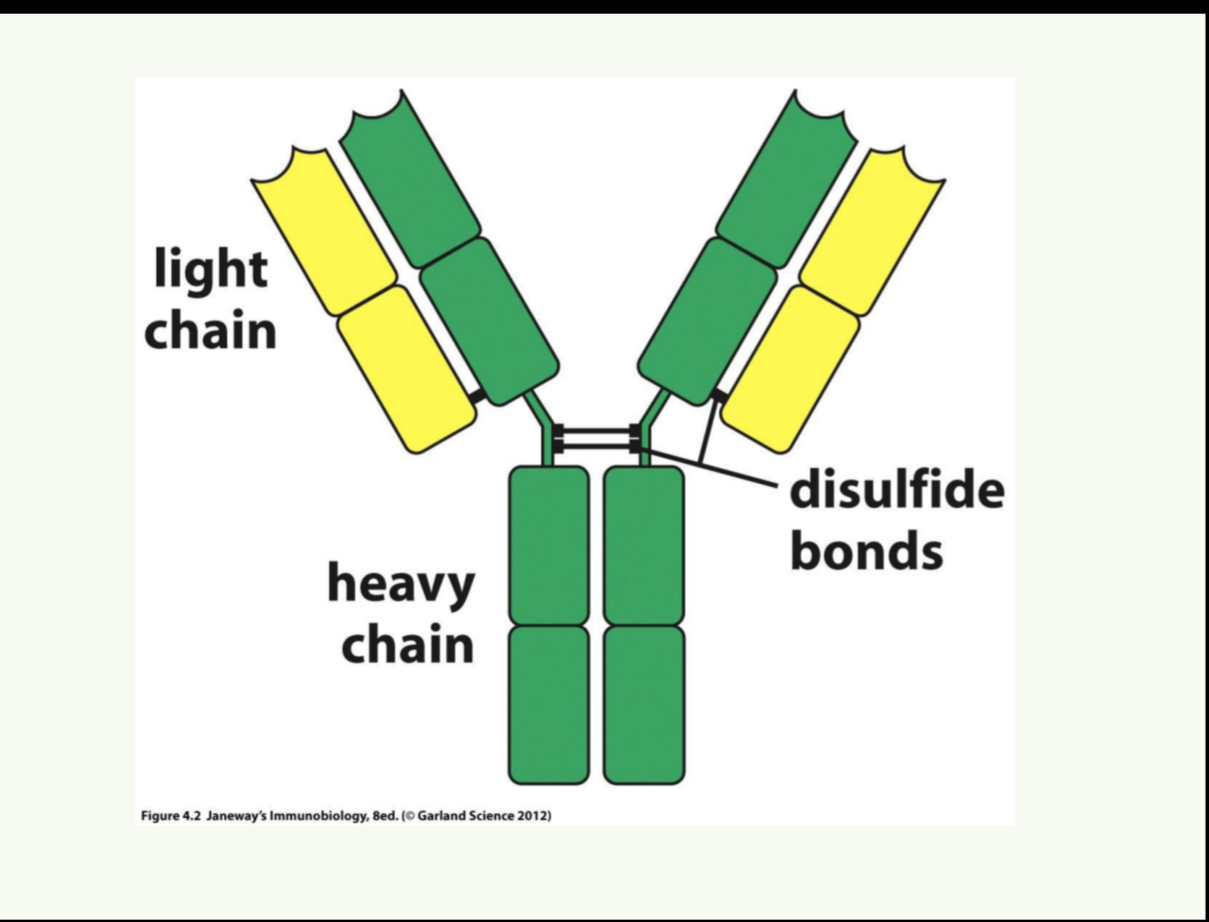
Structure of Antibodies
Antibodies also called (immunoglobulins) are glycoproteins produced by plasma cells. There are five major classes, each with distinct structure and function.
Each antibody molecule has:
2 identical heavy (H) chains
2 identical light (L) chains
These form a Y-shaped molecule connected by disulfide bonds.
Each chain has:
A variable region (V) – responsible for antigen binding.
A constant region (C) – determines antibody class and function.
The V region forms the antigen-binding site. One antibody has 2 identical sites.
📍 The Fc region is at the base of the Y – it controls immune functions (e.g. binding to receptors, complement activation).
📍 The Fab regions are the arms of the Y – they bind antigens.
📍 The Hinge region (in some classes) gives flexibility to the antibody arms.
Classes of Antibodies (Isotypes)
Class Structure Function
IgG Monomer Most abundant in blood. Crosses placenta
IgA Monomer/dimer Found in secretions (tears, saliva, breast milk). Protects mucosal surfaces.
IgM Pentamer First antibody made. Good at agglutination and complement activation.
IgE Monomer Involved in allergies. Binds mast cells and triggers histamine release.
IgD Monomer Found on naïve B cells. Role in B cell activation.