pH
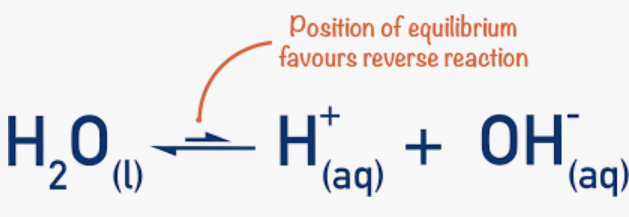
At 298K:
Kw= [H^+][OH^-]=1.00×10^-14 mol²dm^-6
[H^+]² = 1.00×10^-14
[H^+] = 1.00×10^-7
pH = - log(b10)[H^+]
given to 2dp
Strong acids
HA(aq) —> H^+ + A^- (aq)
strong acids fully dissociate
thus [H^+] = [HA]
[H^+] = 10^-pH
conc of acid is thus [H^+]/# of H+ ions