soils 3a
the water molecule
oxygen wants to gain 2 electrons
two hydrogen atoms want to lose one electron each
h2o
two H share electrons w O
everyone is happy
charge is balanced
covalent bond
oxygen holds the electrons closer to its nucleus than the H
causes the bend in the molecule
results in “dipole moment”
zones within the molecule that are more negatively charged or more positively charged than the rest of the molecule
water molecules act like magnets
opposite charges attract
+ attracts -
- attracts +
results in polarity
importance of polarity
explains why water molecules are attracted to charged ions and to colloidal surfaces
positively charged ions are attracted to oxygen side of water (neg charged side)
negatively charged ions are attracted to hydrogen side of water (pos charged side)
ionic compounds have greater attraction for water molecules than each other
makes water the “universal solvent”
nonpolar compounds
have an equal charge balance throughout the compound
no dipole moments
nonpolar substances can only be dissolved in nonpolar solvents
that is why grease (organic compound) does not dissolve in water without soap
hydrogen bonds
hydrogen atom from one water molecule is attracted to the oxygen atom of another
forms a low energy bond between the two molecules
hydrogen bond
h attracted to other o, vice versa
cohesion and adhesion
cohesion
attraction of water molecules for each other
“cohorts”
adhesions
attraction of water molecules to solid surfaces
“adhere”
surface tension
liquid air interface surface tension results from the greater attraction of water molecules to each other than for the air
results in an inward force at the surface
water molecules act as if there was a stretched elastic membrane covering the surface
capillary action
due to two forces
attraction of water for the solid (adhesion)
surface tension of the water (due strongly to cohesion)
in glass tubes
water molecules are attracted to the glass (adhesion)
simultaneously cohesive forces hold water molecules together and create surface tension
causes a curved surface
lower pressure below meniscus
allows higher pressure in free tube to push water up
height of rise in capillary tube is inversely proportional to tube inside radius, r
with water at 20 degrees C, height, h can be found with:
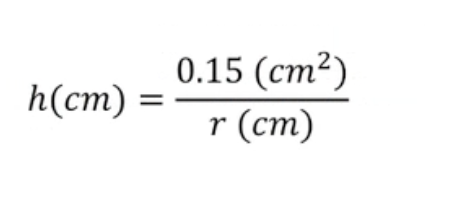
capillary action in soils
capillary forces are at work in most soils
rate of movement and rise in height are less than you’d expect based on pore size alone
pores aren’t straight smooth tubes like glass
some pores are filled with air
could be entrapped air
slows down movement of water and decreases rise in height.
the pore size distribution that controls capillary rise
sands have abundance of medium to large sized pores
permits rapid capillary rise
limits ultimate height of rise
clays have abundance of small sized pores
takes a long time for water to rise up the small pores
ultimately rises to a height greater than in sands