Gas Laws
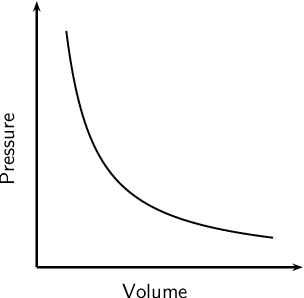
Boyle’s Law
the volume of a given mass of gas varies inversely with the pressure when the temperature is kept constant
We use P1 and V1 to stand for the initial pressure and initial volume of a gas. After a change has been made, P2 and V2 stand for the final pressure and volume. The mathematical relationship of Boyle's Law becomes:
P 1 × V 1 = P 2 × V 2
2 changing variables - pressure + volume
constant variable - temperature
inverse relationship
real life example: syringe with marshmallow inside
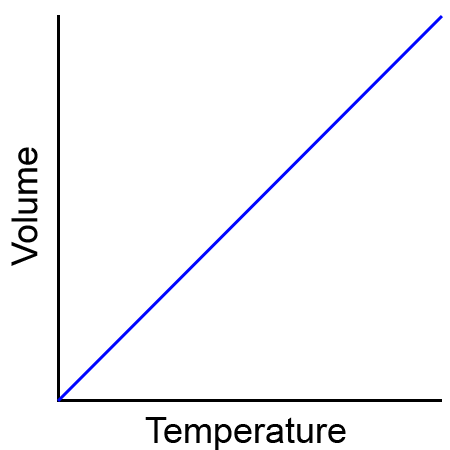
 Charles’s Law
Charles’s Law
the volume of a given mass of gas varies directly with the temperature of the gas when the pressure is kept constant
We will use V1 and T1 to stand for the initial volume and temperature of a gas, while V2 and T2 stand for the final volume and temperature. The mathematical relationship of Charles's Law becomes:

2 changing variables - temperature + volume
constant variable - pressure
direct relationship
real life example: tire cold in the winter (temperature decreases) ➡ pressure decreases
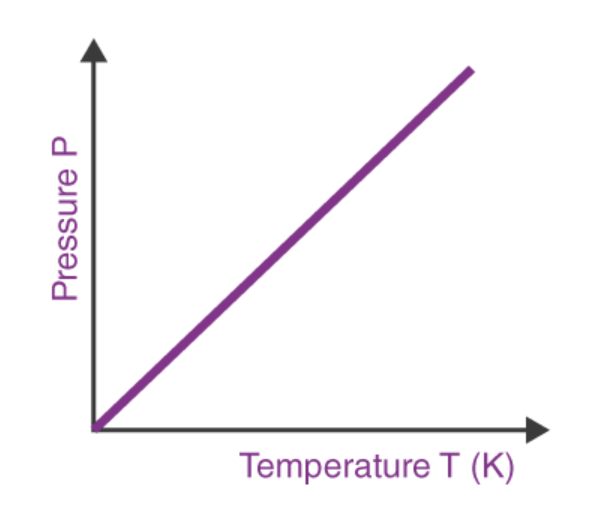
Gay-Lussac’s Law
the temperature of gas varies directly with the pressure when the volume is kept constant
We will use V1 , P1, and T1 to stand for the initial volume, pressure, and temperature of a gas, while V2, P2, and T2 stand for the final volume, pressure, and temperature. The mathematical relationship of Charles's Law becomes:
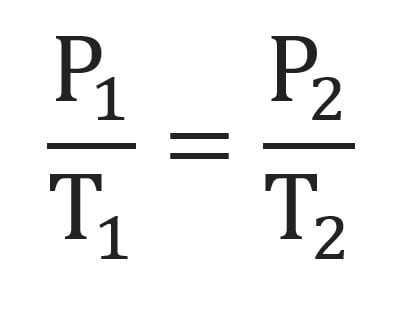
2 changing variables - pressure + temperature
constant variable - volume
direct relationship
real life example: rice cooker gets heated up (temperature increases), and pressure increases inside of the cooker; this causes the whistle to blow