Chapter 2: Atoms, Ions, and Compounds
2.1 Atomic Structures and Isotopes
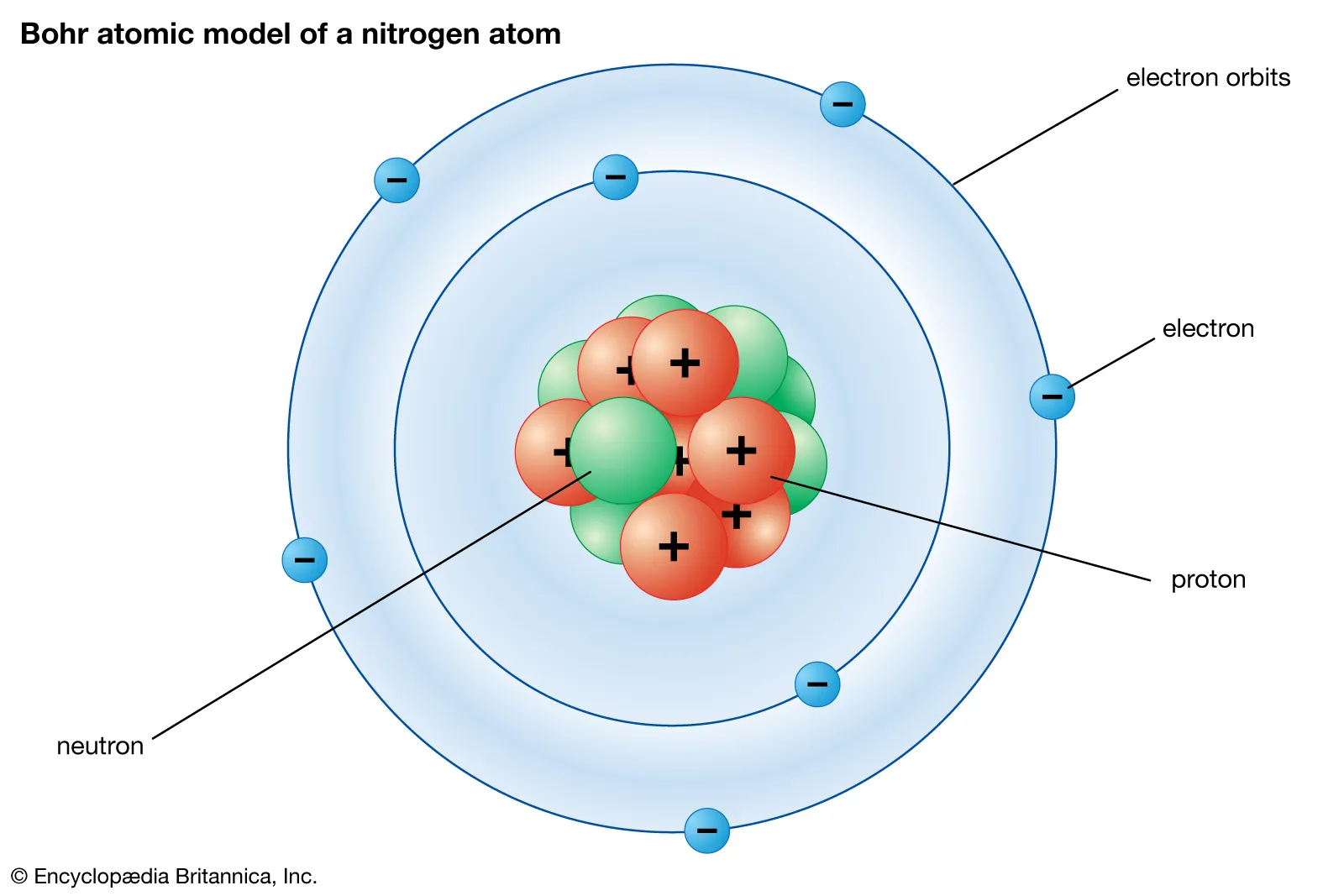
Subatomic Structures of the Atom
- The nuclear model of the atom consists of a ==nucleus== and surrounding ==shells==
- The nucleus contains 2 subatomic particles, ==protons== and ==neutrons==
- The shells are comprised of the third type of subatomic particle, the ==electron==
- Subatomic particles have relative masses and charges:
* Proton - has a mass of ==1== and charge of ==+1==
* Neutron - has a mass of ==1== and a charge of ==0==
* Electron - has a ==negligible mass== or (1/1836) and a charge of ==-1==
Building the Atom
- In an atom, most of the mass is stored ==in the nucleus==
- An atom contains the ==same number== of protons as electrons
- The ==total positive== charge of the protons ==cancels== out the ==negative== charge from the electrons
- The ==overall charge== of an atom is ==0==
- The neutrons can be thought of as the glue that holds together the protons in the nucleus
- Most atoms have very ==similar or the same== numbers of protons as neutrons
- As the nucleus gets bigger, more neutrons are needed
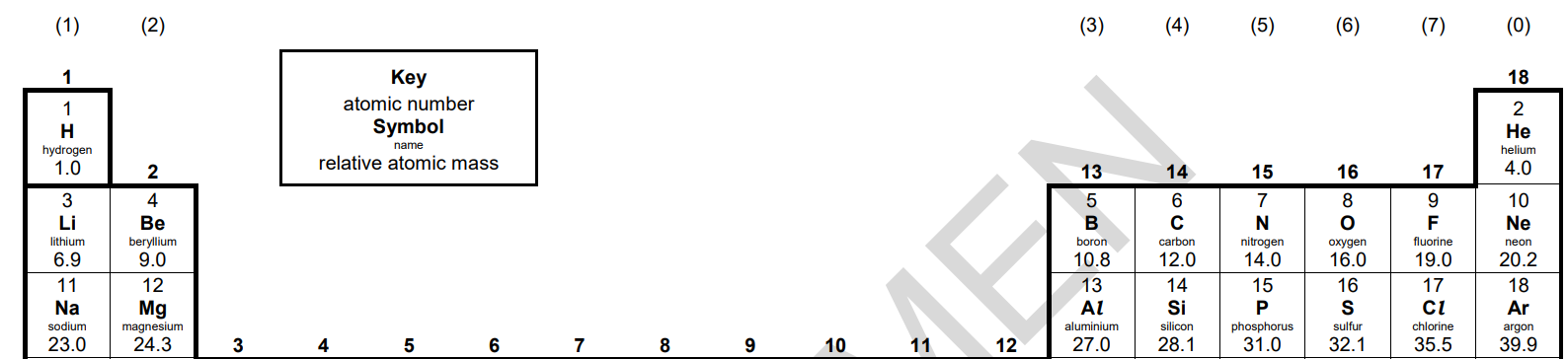
Atomic Number - the identity of an element
- The ==number of protons== in an atom identifies the element
- Every atom of the same element contains the ==same number of protons==
- Different element’s atoms have different numbers of protons
- The ==Periodic Table== lists elements in order of the number of protons in the nucleus
- Each element on the Periodic Table is shown with its ==symbol and atomic number==
Isotopes
- Every atom contains the same number of protons, however, ==the number of neutrons can differ== slightly as it doesn’t affect the charge of the atom
- ==Isotopes== are atoms of the same element with different numbers of neutrons and different relative atomic masses
- Most elements consist of a mixtures of isotopes
Representing Isotopes
- Isotopes are represented using this notation:
*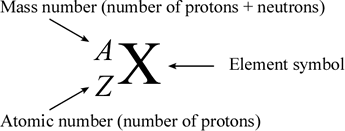
* ==Mass number== = A
* Atomic (proton) number = Z
* Element symbol = X - This notation can be used to derive the number of protons, neutrons, and electrons in an isotope of an element
- Chemists may also represent isotopes by adding a hyphen and the mass number of an element after the element name
* e.g. oxygen-16, carbon-12
Isotopes and Chemical Reactions
- Chemical reactions involve the ==electrons== surrounding the nucleus
- Because isotopes only differ in the number of neutrons, ==different isotopes== of the same element react in the same way
- However different isotopes may have slightly different physical properties like melting points or density
Atomic Structure of Ions
- An ==ion== is a charged atom
- They have ==different numbers of electrons==
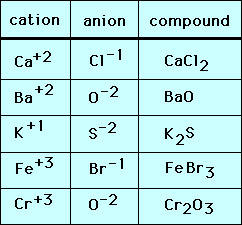
- Positive ions:
* Called ==cations== (cat-eye-on)
* Fewer electrons than protons
* Overall positive charge - Negative Ions:
* Called ==anions== (ann-eye-on)
* More electrons than protons
* Overall negative charge - Ions are always shown with their overall charge:
* e.g. Mg2+, Cl-
2.2 Relative Mass
Carbon-12
- Unfortunately finding the relative mass of an isotope isn’t as simple as adding the masses of the total protons, neutrons, and electrons due to the nuclear force holding the neutrons and protons together coming at the expense of losing mass
- Because of this, chemists calculate all atomic masses based on 1 isotope - ==carbon-12==
- 1 atom of carbon-12 has a mass of 1.992646538 x 10^-26 kg
- The mass of a carbon-12 isotope is defined as ==exactly 12 atomic mass units== (12u)
- So 1u is equal to 1/12th of a carbon-12 atom, and approximately the mass of ==1 proton== or ==neutron==
Relative Isotopic Mass
- ==Relative isotopic mass== is the mass of an isotope relative to 1/12th of the mass of an atom of carbon-12
- This has no units as it is the ratio of two masses, not an actual measurement
Relative Atomic Mass
- Many elements have many different isotopes
- ==Relative atomic mass== (Ar) is the weighted mean mass of an atome of an element relative to 1/12th of the mass of an atom of carbon-12
- The weighted mean takes into account:
* The ==percentage abundance== of an isotope
* The ==relative abundance== of an isotopes - A Periodic Table shows both the atomic number, and the relative atomic mass
Determination of Relative Atomic Mass
- Percentage abundances of isotopes in a sample of an element are found using a ==mass spectrometer==
- They work by:
* Placing a sample in the spectrometer
* The sample is ==vaporised== and ==ionsied== to form positive ions
* Ions are ==accelerated== and heavier ions move more slowly and are more difficult to deflect than lighter ions, allowing them to be separated
* Ions are detected on a mass spectrum as a ==mass-to-sharge ratio== (m/z)
* Each ion that reaches the detector adds to the signal, so ==the greater abundance of an isotope, the larger the signal== - ==mass to charge ratio m/z = relative mass of ion/relative charge on ion==
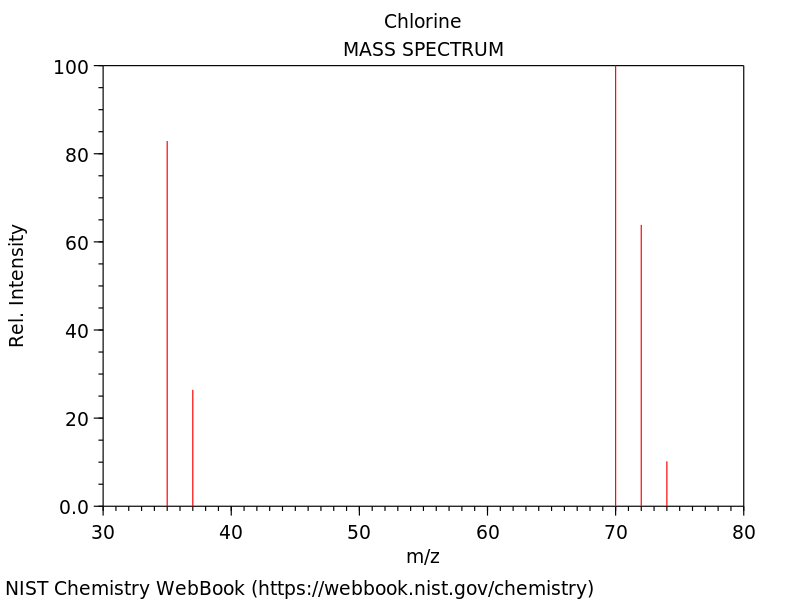
- Each peak on a mass spectrum graph is a ==different isotope== and their height is their ==relative abundance==
- Another useful equation is the equation for relative atomic mass:
*
2.3 Formulae and Equations
Ionic Charges
Simple Ions From the Periodic Table
- Many atoms ==lose or gain electrons== to achieve the electron structure of the ==nearest noble gas==
- Atoms of ==metals== on the left side of the Periodic Table (up to group 4) ==lose== electrons to form ==cations==
- Atoms of ==non-metals== on the right side of the Periodic Table (after group 4) ==gain electrons== to form ==anions==
- You can use the position of an element on the periodic table to work out the charge of an ion
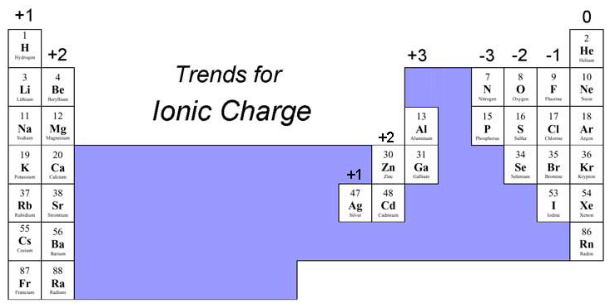
- Some ==transition metals== can form ions with ==different== charges:
* Copper - copper(I), Cu+, and copper(II), Cu2+
* Iron - iron(II), Fe2+, and iron(III), Fe3+
Binary Compounds
- A ==binary compound== contains two elements only
- To name a binary compound, use the name of the first element and change the ending of the second element’s name to -ide
- In ionic compounds, the metal ion always comes first
* e.g. sodium oxide
==Polyatomic Ions==
- An ion can be atoms of more than one leelmnt bonded together, this is a ==polyatomic ion==
- These ions also have a charge
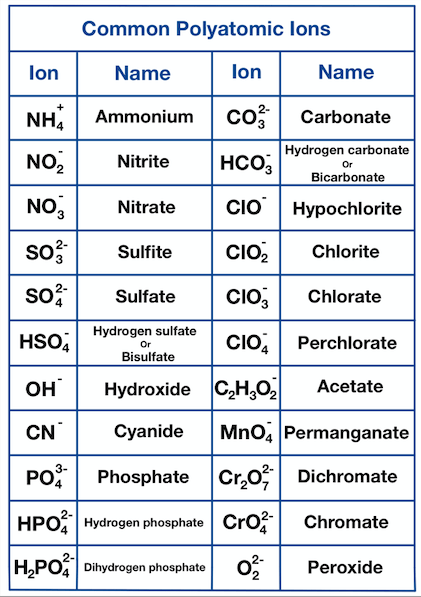
Writing Formulae from Ions
- An ==ionic compound== contains a cation and anion
- The formula can be worked out from the charge on each ion
- In a correct formula:
* The ==overall charge is 0==, so the ionic charges have to balance
* ==sum of positive charges = sum of negative charges==
Writing the Formula
- The number of each ion in the formula is shown in subscript after the ion
- Brackets are used if there is more than one polyatomic ion
Writing Equations
Representing Elements and Compunds in Equations
Elements
- ==Elements== are simply shown as their symbole except for elements that exist as small molecules
- These are mostly bonded together as ==diatomic molecules==:
* H2, N2, O2, F2, Cl2, Br2, and I2 - Some others are:
* P4 and S8 (although sulfur is usually written as S in equations)
Compounds
- ==Covalent compounds== don’t contain ions, most exist as molecules with a small number of atoms bonded together:
* H2O, CO2 - In ==ionic compounds==, the formula worked out from the ionic charges is used in equations - this is called the ==formula unit==
State Symbols in Chemical Equations
- ==State symbols== are shown in brackets to show a physical state:
* (g) - gas
* (l) - liquid
* (s) - solid
* (aq) - aqueous/dissolved in water
Balancing Equations
- To balance an equation:
* Multiply each formula by a ==balancing number== until the number of atoms of each element is ==the same== on each side of the equation - Rules of balancing:
* You must not change any chemical formulas
* Balancing numbers go in front of chemical formulae and not subscripted
* The equation is only balanced when there are the same number of atoms of each element on each side of the equation