11-19-25 Einstein Model of a Solid
Irreversibility
Many physical processes appear to only happen in one direction in time
Example: Ice left in the open in a warm room melts as energy from the warmer surroundings is spontaneously transferred to the ice. We never observe water left in a warm room losing energy to the surroundings and spontaneously freezing
Process that only happen in one direction are called irreversible
Irreversibility occurs when a process is much more likely to happen in one direction than it is to happen in the reverse direction. even though both directions are possible
The fundamental interactions are all reversible
Einstein Model of a Solid
In the Einstein model of a solid, each atom is connected to the rest of the solid by three spring-like interatomic bonds. Each of these bonds can be modeled as a quantum oscillator, with quantized energy levels.
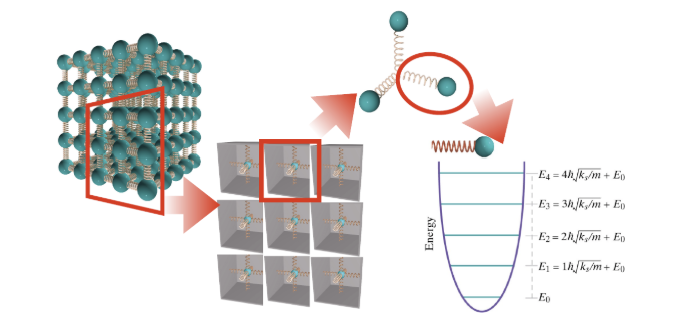
Microstate vs. Macrostate
The total energy in collection of bonds specifies the macrostate. Each distinct way of distributing that total amount of energy among the bonds is called a microstate.
Example: oscillators with total quanta of energy
Fundamental Assumption of Statistical Mechanics
Given enough time, every microstate of a system is as equally likely to appear as any other
Some macrostates contain more microstates than others, which makes the system more likely to be in them
Example: Microstates A-D are equally likely. The macrostate {system looks kind of like A} is more likely than the macrostate {system looks kind of like C}
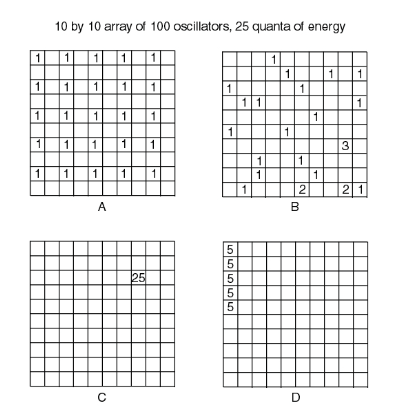
Consider 4 quantized oscillators that share 3 quanta of energy. List all the ways you can arrange these 3 quanta of energy among the 4 oscillators. How many arrangements are there?
,
20 total variations
Counting Microstates
To find the total number of arrangements, , of some total number of quanta energy, q , among some number of oscillators, , we can use the following combinatorial formula:
where
If and what is the value of ?