Tumour hypoxia
Tumour Response to Hypoxia: Understanding the Hypoxic Tumour Microenvironment to Improve Treatment Outcomes
Authors and Affiliations
Introduction
Hypoxia: A common trait of solid tumors influencing their biological behavior and therapeutic responses.
Hypoxia-Inducible Factor (HIF): A key regulator induced by hypoxia, affecting tumorigenesis through various mechanisms:
Regulates angiogenesis
Modulates immune responses
Alters metabolic pathways
Encourages cell migration through extracellular matrix remodeling.
Impact on Patients: Negative implications including therapeutic resistance.
Need for Research: Focus on clinically relevant hypoxia biomarkers and hypoxia-targeting therapies.
Hypoxia in Tumors
Detection Challenges: Many existing methods for hypoxia detection are not clinically applicable or standardized.
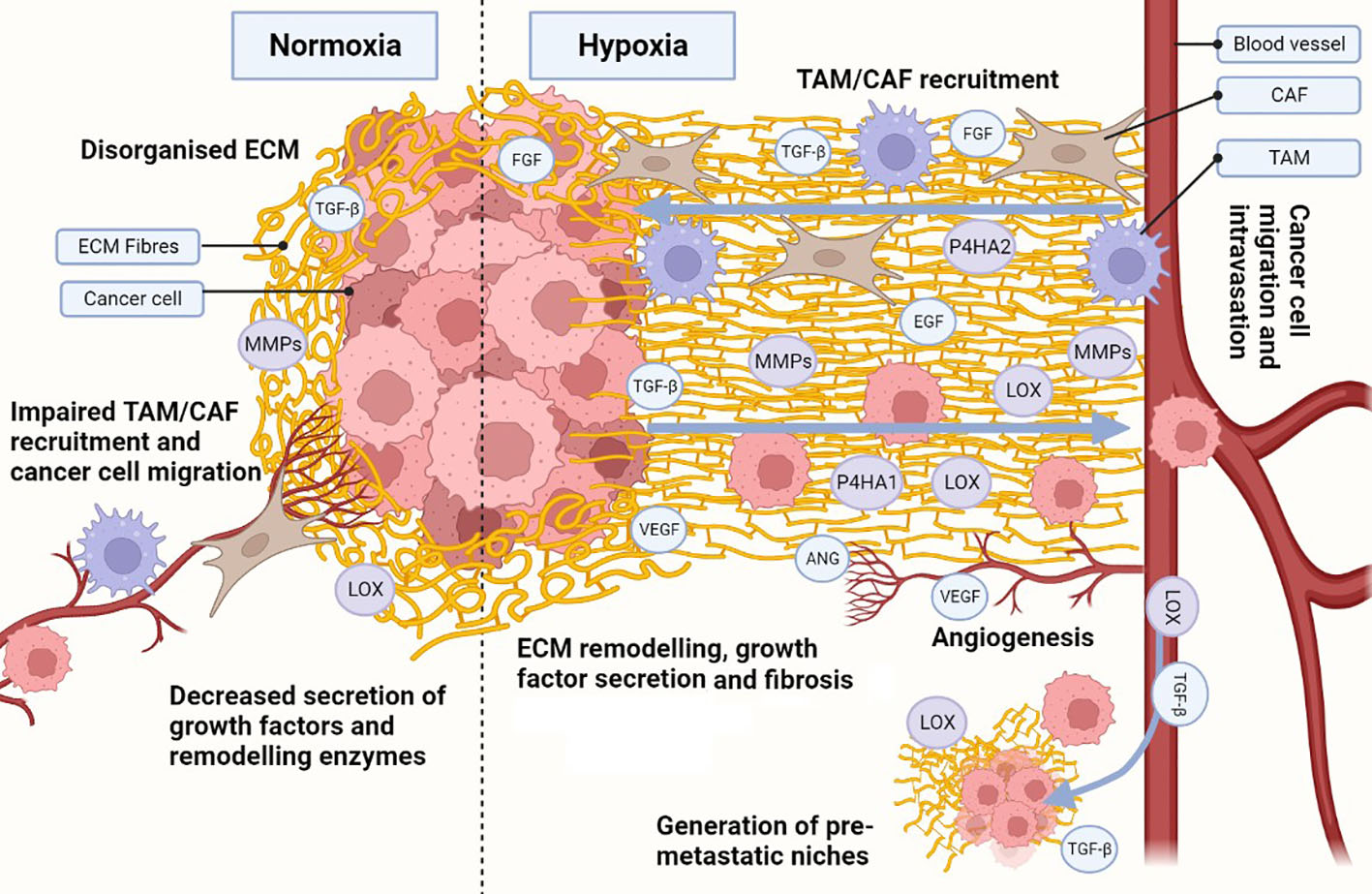
Extracellular Matrix (ECM): Created by cancer-associated fibroblasts (CAFs) and tumor cells, vital in understanding the hypoxic tumor microenvironment (TME).
ECM components affected include fibronectin (FN), collagen (COL), and hyaluronic acid (HA).
Importance of CAFs: Diverse roles in promoting or inhibiting tumorigenesis, highlighting the need for subtype distinctions.
Cellular Dynamics of Hypoxia
Types of Hypoxia
Chronic Hypoxia: Sustained low oxygen levels.
Acute Hypoxia: Short episodes of low oxygen levels.
Cyclic Hypoxia: Periodic exposure to hypoxia followed by reoxygenation, influencing tumor behavior and response to treatments.
Role of HIF in Tumor Response
HIF Regulation:
Heterodimeric complexes (HIF-1, HIF-2) formed under low oxygen regulation.
Activation of genes related to survival, glucose metabolism, angiogenesis.
Various Isoforms:
HIF-1 and HIF-2 as potential therapeutic targets with distinct roles.
Hypoxia as a Prognostic Biomarker
Approximately 50% of solid tumors exhibit significant hypoxia, correlating with poor patient prognosis.
Measurement Techniques:
Direct oxygen tension measurement (e.g., Eppendorf probes) considered gold standard but invasive.
Indirect measurements using immunohistochemistry (e.g., HIF-1a, CA9, GLUT1) show potential but require standardization across labs.
Key Components Affected by Hypoxia
Immune Cells
Hypoxia-Induced Changes: Alters immune cell behaviors, promoting an anti-tumorigenic environment—significant effects on immune checkpoint modulation and therapy response.
ECM and Matrix Stiffness
Increased stiffness enhances fibrosis, impacting tumor aggressiveness and therapeutic resistance.
CAF Activation
Various signalling pathways influence CAFs under hypoxic conditions, enhancing pro-tumorigenic activity.
Mechanisms Contributing to Therapeutic Resistance
Treatment Modifications: Hypoxia modifies responses to chemotherapy, radiotherapy, and immunotherapy. Key mechanisms include:
Cell cycle regulation.
Enhanced survival signaling pathways activated by hypoxia.
Increased metabolic alterations leading to cellular adaptations.
Future Perspectives
Research Gaps: Understanding the interplay of HIF, ECM changes, immune response alterations, and CAF contributions in hypoxic conditions.
Clinical Implications: Developing standardized applications for hypoxic biomarkers with potential treatments focusing on exploiting hypoxia as a target.
Targeted Therapies: Novel strategies are required for precise targeting based on individual tumor characteristics, potentially integrating personalized medicine approaches.
Conclusion
The hypoxic tumor microenvironment is a complex and significant factor affecting cancer progression and treatment responses. Addressing these challenges is crucial for developing effective therapies at the level of individual tumor biology.