Lecture 31: Reduction and Oxidation Part 1.
Reduction and Oxidation
Half Reactions
Chemical transformations can be represented by full equations and half-reactions.
Full equations show the starting materials and the end products of a chemical reaction.
Half reactions detail what happens with individual species like zinc and copper ions .
Half-reactions are useful as they show the origin and destination of electrons.
Help focus on the ability of a reagent to gain or donate electrons.
Electrons are neither created nor destroyed; they are transferred.
Example:
Zinc goes from to and becomes soluble in an aqueous environment, losing two electrons.
Copper ions accept or gain two electrons, forming copper zero . is reduced to .
Oxidation and Reduction
Oxidation: A molecule is oxidized if it loses electrons.
In the example, zinc is oxidized.
Reduction: A molecule is reduced when it gains electrons.
In the example, copper ions are reduced.
Reduction Potential
Together half reactions predict the direction of spontaneous electron transfer.
Determine which reagent is a stronger reductant and which is a better oxidant.
All metals are reducing agents and donate electrons to become cations.
Zinc is a stronger reducing reagent than copper in the example reaction.
Standard Reduction Potential: The ability of a metal to lose electrons, measured in volts (V).
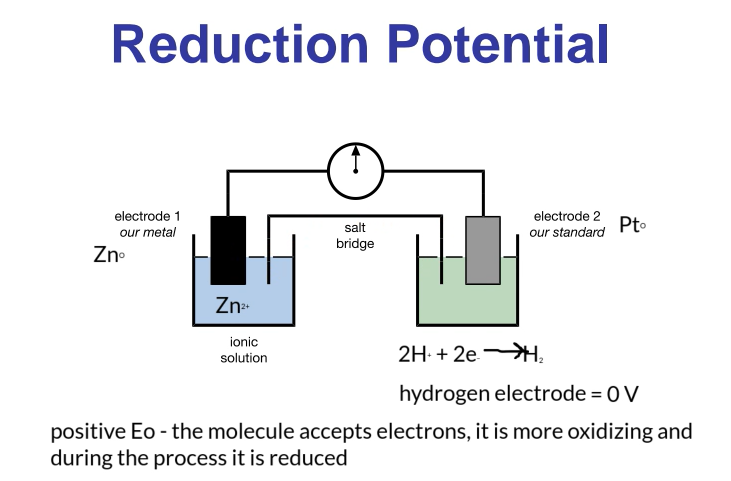
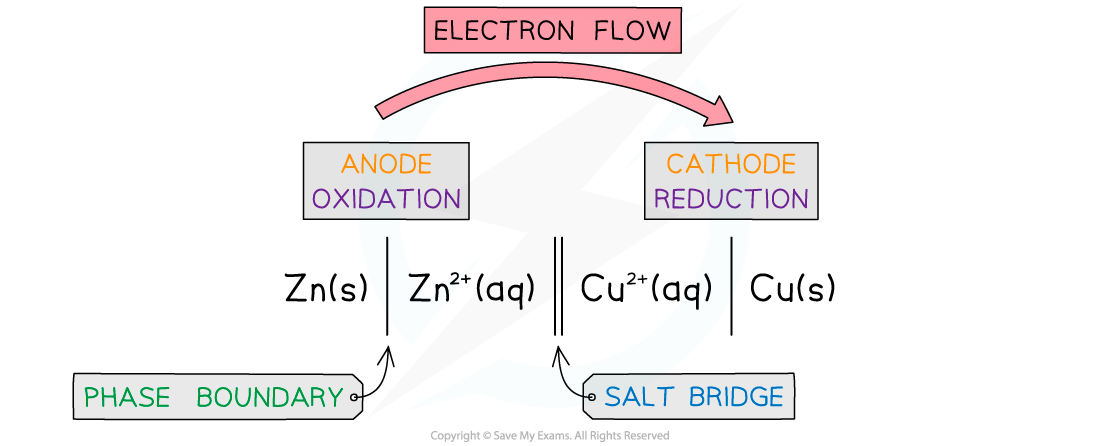
Reduction Potential: Electrochemical Cell
Reduction potential is measured using an electrochemical cell.
Measures the potential difference (voltage) between the anode and the cathode.
Half Cell 1:
A metal electrode submerged in an ionic solution.
If measuring zinc potential, the electrode is made of zinc, and the solution contains zinc ions (e.g., zinc sulfate).
Half Cell 2:
A standard electrode, usually platinum (Pt) because it is inert.
Used in a hydrogen cell to transport electrons.
Hydrogen electrode has a standard reduction potential of 0V.
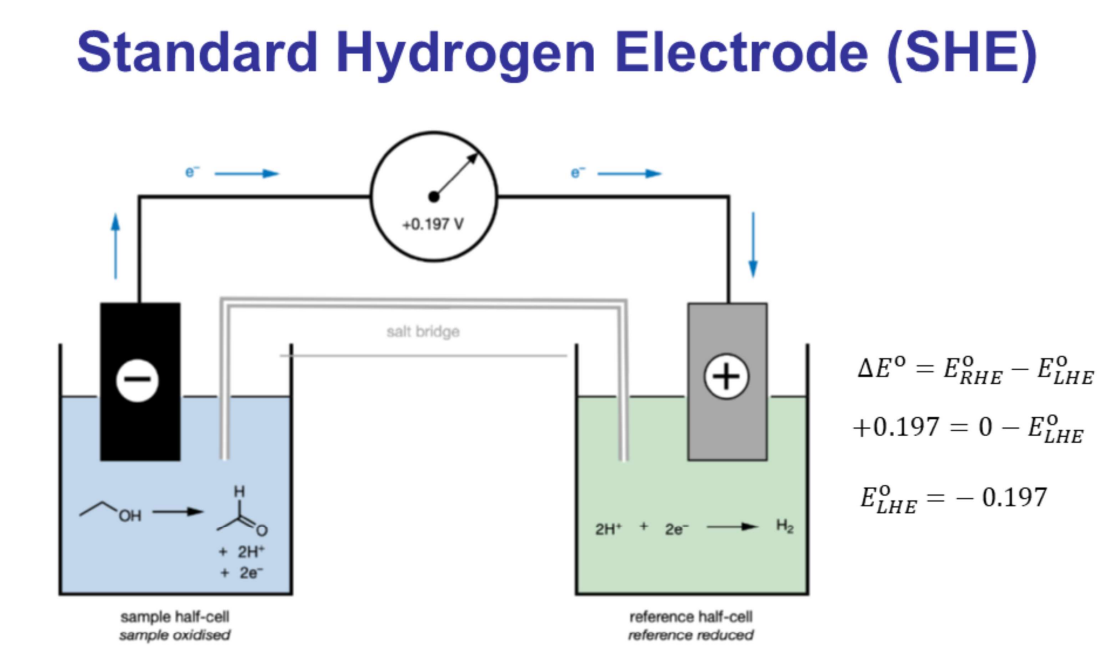
The Standard Hydrogen Electrode (SHE) is a reference electrode in electrochemistry, defining the zero point for measuring the potential of other electrodes.
It consists of a platinum electrode immersed in a solution of hydrogen ions.
Positive Reduction Potential: The molecule accepts electrons; it is more oxidizing and is reduced.

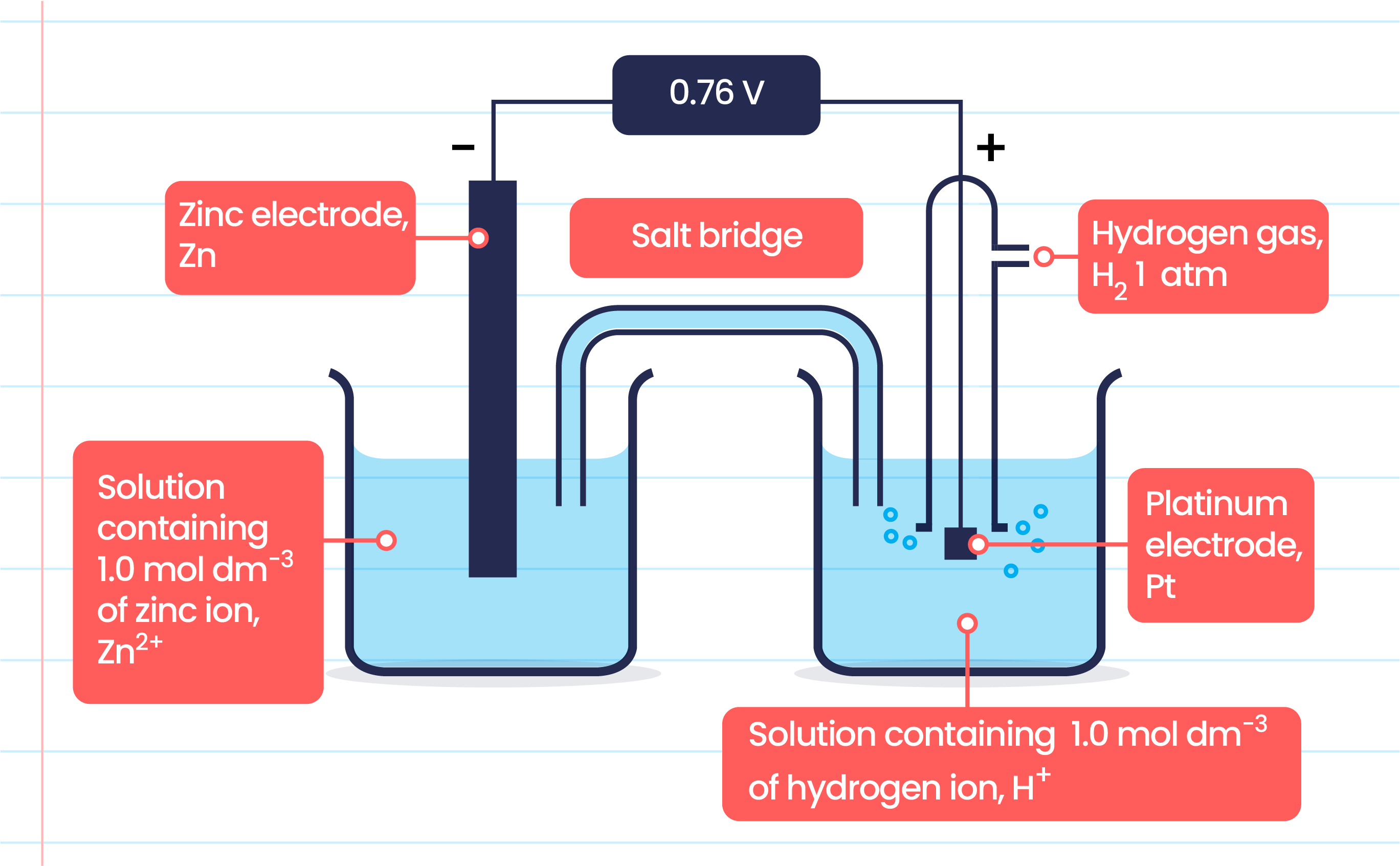
Salt Bridge
Connects the cathodic and anodic compartments of the cell, closing the circuit.
Made from cotton, paper, or a glass capillary tube filled with a gel (e.g., agar) submerged in an electrolyte solution.
Prevents the two solutions from mixing.
Maintains electrical neutrality in the internal circuit.
Ions (cations or anions) move through the salt bridge to maintain electrochemical balance, not electrons.
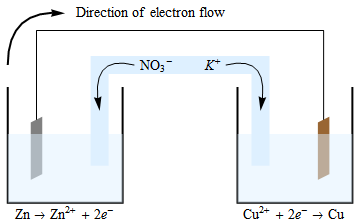
Conventional Representation of Cells
The Anode(negative electrode)
The site of oxidation (loss of electrons)
On the left.
The Cathode(positive electrode)
The site of reduction (gain of electrons).
On the Far Right.
When written they are separated by two vertical lines(∥).
These represent the Salt bridge.
A single vertical line (|) indicates a phase boundary.
(e.g., between a solid electrode and a solution).
The species with the highest oxidation state in each half-cell is placed closest to the salt bridge.
The highest oxidation state refers to the maximum number of electrons an atom of that element can lose (or gain) in a chemical reaction to achieve a stable electron configuration.
i.e. the species with the highest charge

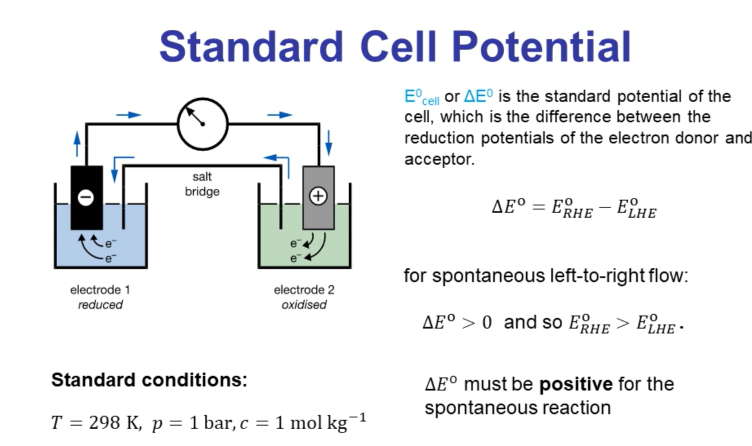
Standard Cell Potential
Reduction potentials and standard cell potentials are performed under standard conditions:
Temperature: 298 K
Pressure: 1 bar
Concentration: 1 M
The standard cell potential is the difference between the reduction potentials of the electron donor and acceptor.
For a spontaneous reaction (left to right flow), the standard cell potential () must be positive.
The reduction potential on the right-hand side electrode must be higher than the reduction potential on the left-hand side electrode.
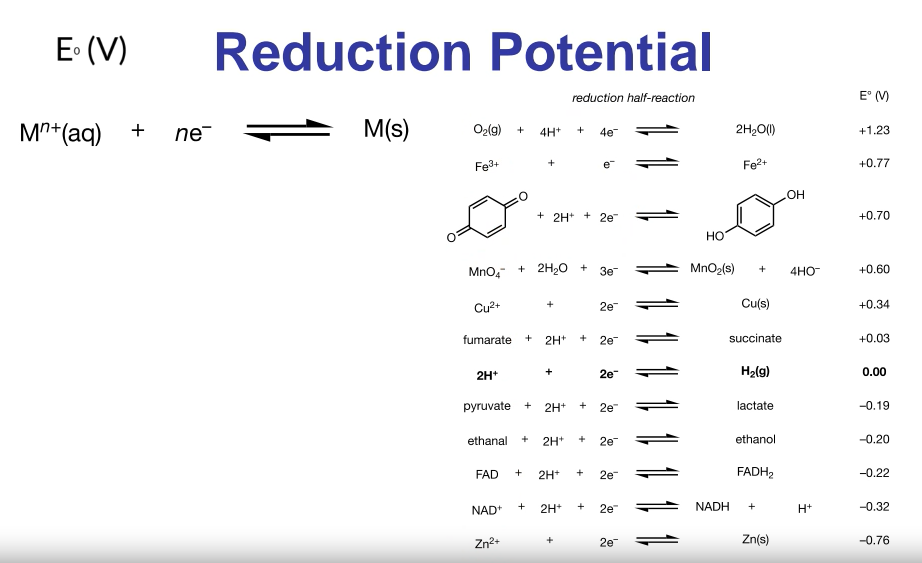
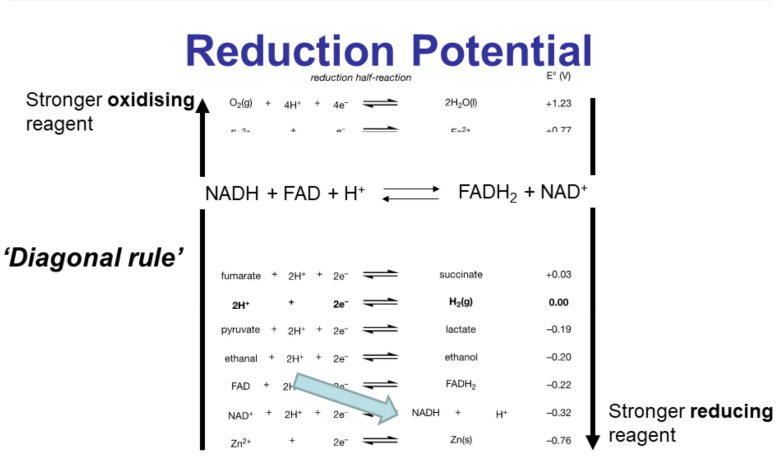
Reduction Potential Table
Lists reduction half-reactions.
All chemicals are reduced in the table (gain electrons).
The hydrogen electrode is the standard and has a 0V reduction potential.
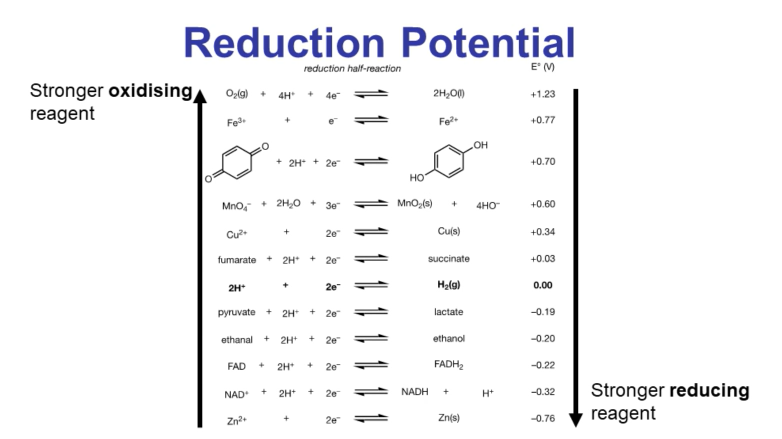
More negative reduction potential indicates a stronger reducing reagent.
More positive reduction potential indicates a stronger oxidizing reagent.
If the ratio of reagents is not one to one, do not multiply or divide the reduction potentials.
Use them as they are.
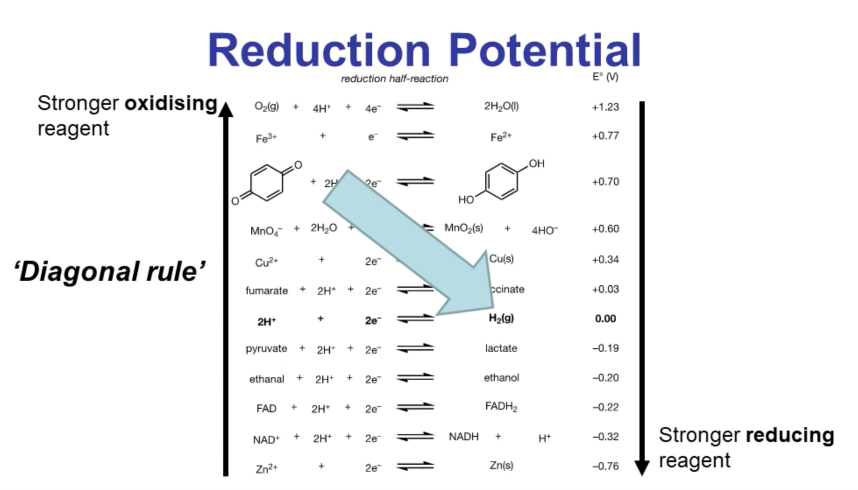
Diagonal Rule
Stronger oxidizing reagents on the left-hand side of the reduction potential equation will oxidize stronger reducing reagents on the right-hand side.
Example: Oxygen will oxidize hydrogen.
Can be used as a first approximation to determine if a reaction between two reagents will be spontaneous.
Note: The rule does not work in reverse.
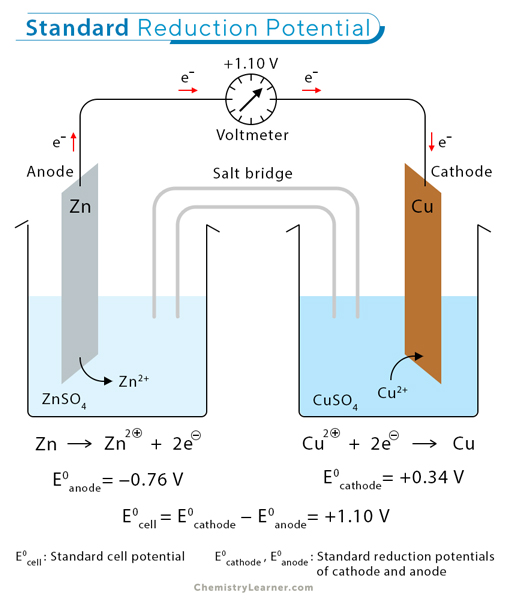
Applying to Copper and Zinc Reaction
Overall reaction: Copper ions are reduced to copper zero , and zinc is oxidized to zinc ions

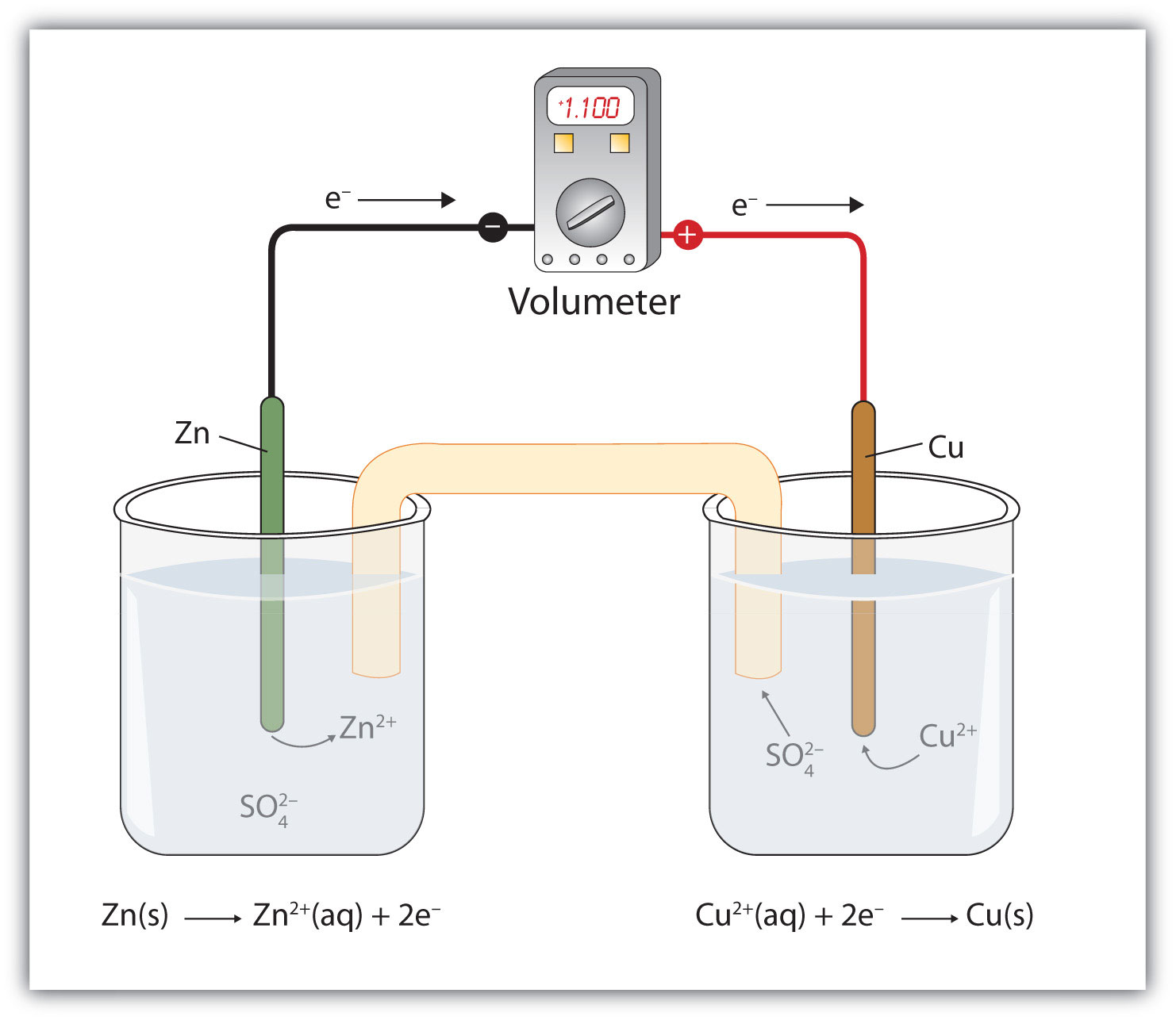
Copper ions act as an oxidizing agent (gets reduced while oxidizing Zinc).
The reduction potential on the cathode is for the half-reaction that mimics the actual transformation (reduction of copper ions).
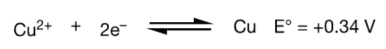
The reduction potential on the anode is for the oxidation of zinc, using the reverse of the reduction reaction and applying a negative sign.


A positive cell potential indicates a spontaneous process.
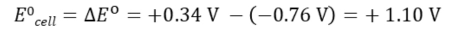
Gibbs Free Energy
The standard cell potential is linked to Gibbs free energy:
For a spontaneous reaction, Gibbs free energy is negative, and the standard cell potential is positive.
= number of electrons transferred
F = Faraday constant
= standard cell potential


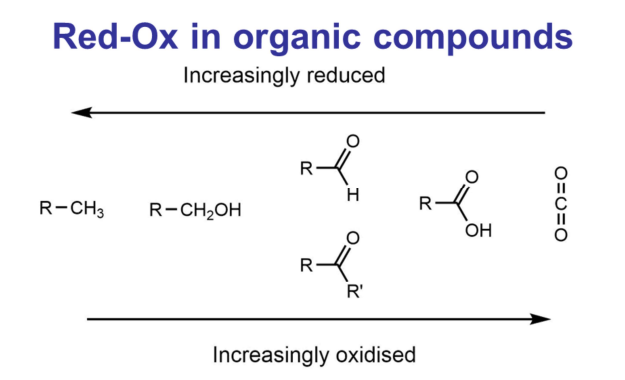
Redox Reactions in Organic Chemistry
In organic chemistry, redox reactions are seen through changes in the oxidation state of carbon.
Oxidation occurs when carbon gains:
An oxygen atom.
Another heteroatom.
More bonds to oxygen (e.g., alcohol to aldehyde).
Further oxidation involves losing a proton and gaining oxygen.
The most oxidized form of carbon is . This represents a progression from fully reduced to fully oxidized carbon.
Terminology
Attract Electrons:
Acids
Lewis Acids: can accept a pair of electrons from a Lewis base.
In the Brønsted–Lowry definition, an acid is a proton (H⁺) donor.
Electrophiles
Oxidants.
Donate electrons.
Bases
Lewis Bases: donates a pair of electrons to the Lewis acid.
In the Brønsted–Lowry definition, a base is a proton acceptor.
Nucleophiles
Reductants
Electrophiles and nucleophiles always coexist in reactions; oxidation is always paired with reduction.
Metal Hydride Reduction (Nucleophilic Addition)
Example: Metal hydride reduction is also known as nucleophilic addition.
Nucleophilic Addition: adds to the electrophilic carbon.
Reduction: The carbon loses a bond to oxygen, thus being reduced.
Reaction: A hydride ion () reacts with an aldehyde (e.g., acetaldehyde).
Mechanism:
The nucleophile () forms a bond with the electrophilic carbon (indicated by curly arrows).
Carbon is an electrophile: The carbonyl group is polarized with a slight negative charge on oxygen and a slight positive charge on carbon.
The second bond between carbon and oxygen breaks, localizing electron density on the oxygen, forming a tetrahedral intermediate.
Carbon gains electrons-Reduced
These electrons are gained by the Hydrogen-Oxidized
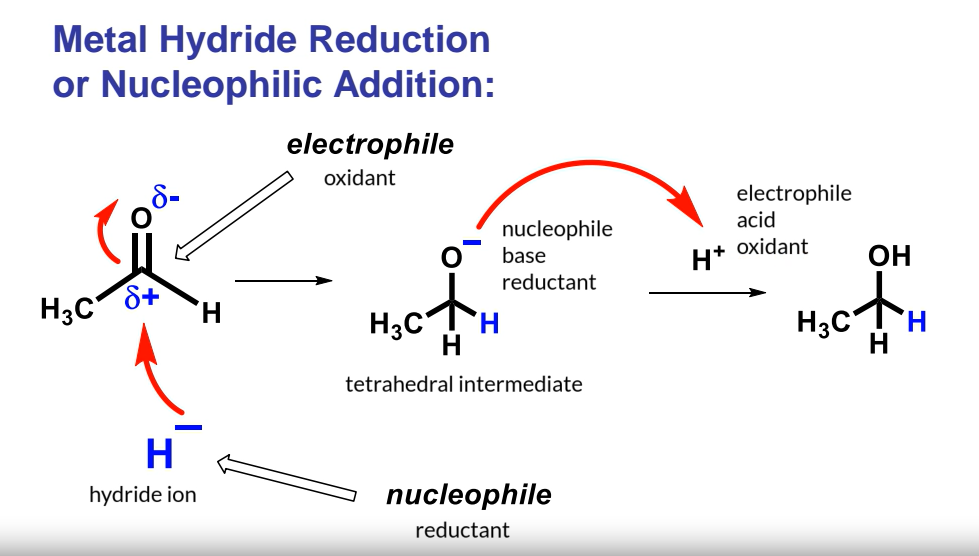
Hydride ion acts as a reductant, while the electrophile is an oxidant.
The hydride ion () is oxidized , forming hydrogen when it bonds to the carbon.
Alkoxide Ion: The alkoxide ion () gains a proton when the solution is acidified , forming an alcohol.
Alkoxide as Nucleophile: The alkoxide ion is a nucleophile, base, and reductant.
Proton as Electrophile: The proton () is an electrophile, acid, and oxidant.
Redox in Biological Chemistry
Redox in Biological Chemistry is vital for energy gain through bond breaking.
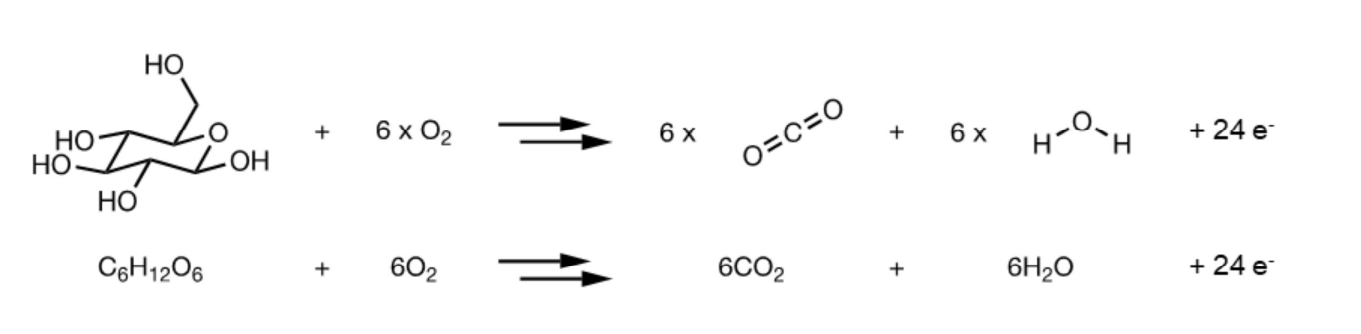
Example: Glucose oxidation.
Overall Reaction: → 6
Glucose is oxidized, and oxygen is reduced to water
Akin to burning glucose in the Prescence of oxygen, resulting in the release of energy.
However the release of energy is not instantaneous.
The human body employs a stepwise energy release facilitated by enzymes.
Glucose never directly reacts with oxygen inside the human body.
Electrons are transferred via electron carriers or cofactors that exist as redox couples (oxidized and reduced forms).
Cofactors/Electron Transfer Molecules
Always come in Redox Couples
One species is in the oxidized from and one is in the reduced form.
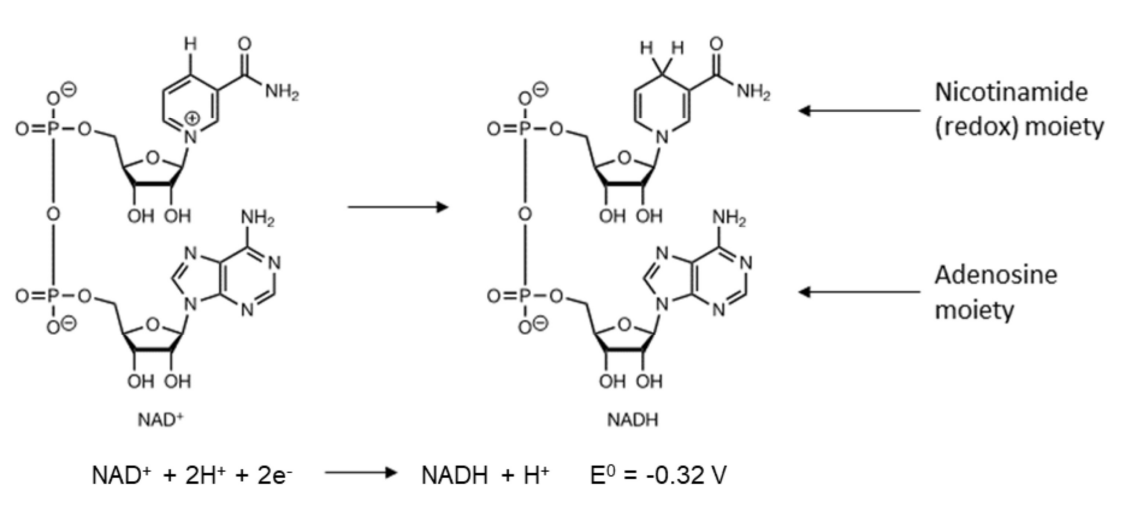
Example:
Non-protein helper molecules in biochemical processes.
In this case structure is based on nucleotides
Structure: Contains an adenosine moiety and a nicotinamide moiety.
Redox Activity: Only the nicotinamide part is involved in the redox process.
Oxidized Form : Nitrogen has a positive charge, and the structure is aromatic.
Reduced Form : Nicotinamide gains one proton and two electrons; the structure becomes non-aromatic.
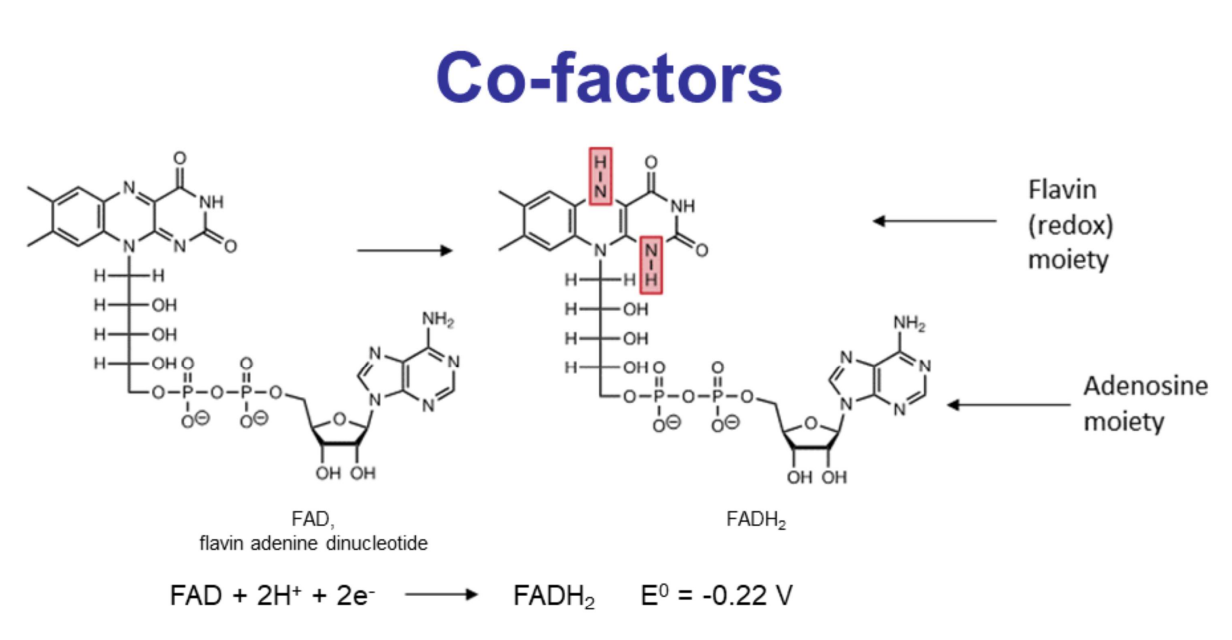
Example:
Flavin Adenine Dinucleotide
Also a dinucleotide composed of adenosine and a flavin moiety.
The adenosine part remains unchanged during redox.
Redox Activity: Occurs on the flavin moiety.
Oxidized Form: Aromatic structure.
Reduced Form: Gains two protons and two electrons.
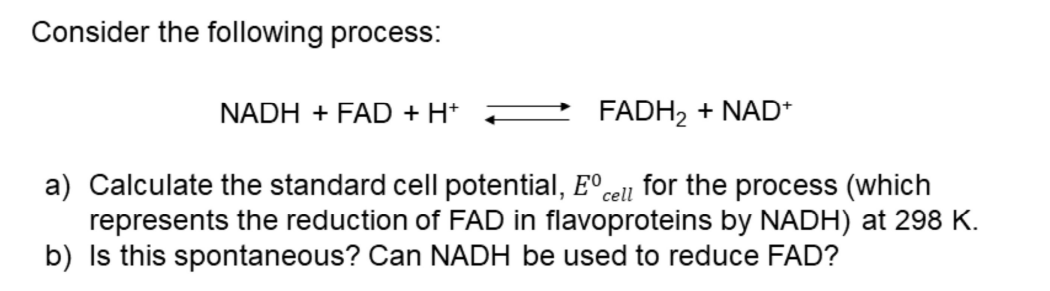
Redox Process Example
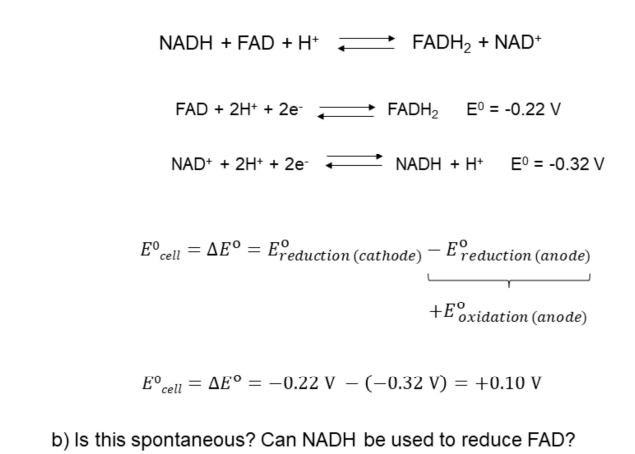
Reduction of FAD in flavoproteins by NADH.
Task: Calculate the standard cell potential and determine if the reaction is spontaneous.
Reduction Potentials
Use a table of reduction potentials to find flavin and nicotinamide.
is a stronger oxidizing reagent than .
is a stronger reducing agent.
can oxidize .
Electrochemical Potential Calculation:
Based on the general equation for redox processes and their standard reduction potentials.
Equation:
: Reduction potential at the cathode.
: Reduction potential at the anode (oxidation reaction).
Values:
= -0.22 V
= -0.32 V
Calculation:
Conclusion:
Since the cell potential is positive, the reaction is spontaneous.
NADH can be used to reduce FAD.