Kinetic Theory of Gases – Comprehensive Study Notes
Overview of Chapter 15 – “Kinetic Theory (of Gases)”
• Text devotes a separate chapter because gases behave qualitatively differently from solids & liquids.
• In solids/liquids particles are already touching → heat, pressure, energy transfer behave almost identically.
• In gases large intermolecular gaps magnify effects of ΔT, ΔP, and container size (e.g., a gas “stapler” would expand ≈4× in each dimension if room T rises by ).
Distinction Between Solids, Liquids, and Gases
• Solids: particles touch & are locked in fixed lattice positions; can only vibrate/rotate.
• Liquids: particles still touch but can slide (flow) over one another.
• Gases: particles far apart, free random motion; properties dominated by collision statistics, not contact forces.
• Practical contrast: door made of wood (solid) swells imperceptibly with heat; a hypothetical gas door would balloon dramatically.
Kinetic Theory: Three Core Postulates ("What it SAYS")
Matter consists of tiny, discrete particles (atoms, molecules, formula units). Macroscopic samples contain trillions or moles of them.
Particles are in constant motion because they possess thermal (kinetic) energy.
Collisions are perfectly elastic within the defined system – kinetic energy is transferred, not lost (losses to surroundings are negligible: 99.999999999 % elastic).
• Multiple–choice questions often contrast “what kinetic theory SAYS” (above) with “what it IMPLIES” (temperature ↔ average kinetic energy, pressure ↔ wall collisions, etc.).
Mean Free Path (ℓ) & Molecular Scale Perspective
• For at : – . • Scale-up thought experiment: if an molecule were in diameter, the next one would be away!
• Despite the gap, thermal velocities are huge: v̄ ≈ 1{,}700\ \text{km·h}^{-1} (≈).
• Collision frequency: ≈ collisions·s⁻¹ for each molecule at room T.
Two Fundamental Collision Types
• Particle–particle (translational) collisions
– Exchange of kinetic energy → manifests macroscopically as temperature (T).
• Particle–wall collisions
– Force/area on container → pressure (P).
• Kinetic-theory problems usually analyze how factors change these two outcomes.
Factors That Control Collision Rate (per second)
Temperature (T)
– ↑T → higher average speed → more & harder collisions.Number density (N/V)
– More particles in the same volume → shorter distance between them → more collisions.Particle mass (m)
– Holding total KE constant, heavier species move slower; larger physical size can decrease free space, altering collision frequency.
Interactive Simulation Insights (Helium vs. Argon)
• Model: 16 He atoms at (≈ –173 °C).
– Wide speed distribution; some slow, some fast.
• Raising to : overall KE ↑; still instantaneous variety of speeds.
• Lowering to <40\ \text{K}: particles curve toward one another (Coulombic e⁻ attraction) → transient clumping → liquefaction onset.
• Adding particles (50 He) or shrinking container ↑ collision rate → P↑.
• Substituting heavier Ar (Z = 18):
– Greater mass & diameter → less straight-line motion, more curvature & clustering at identical T.
– Requires higher T (≥) to restore nearly ballistic paths.
• Expanding container volume disperses particles → P↓ (fewer wall hits).
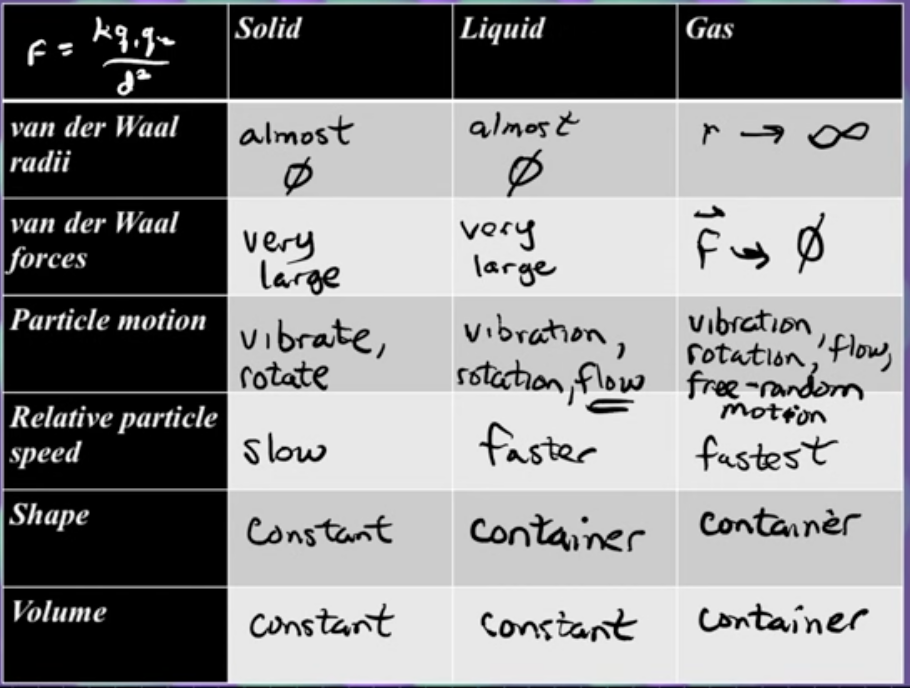
Particle-Level Properties Across States (Consolidated Table)
(Verbal version of the chart instructor required)
• Van der Waals radius (inter-particle gap)
– Solid: ≈0 (particles touch).
– Liquid: ≈0 (touch but can slide).
– Gas: gap → “approaches ∞” relative to particle size (actually ≈ nm for ). • Intermolecular force magnitude (|F|) – Solid: very large. – Liquid: still large, slightly weaker. – Gas: . – Quantitatively: ⇒ large d in gases → negligible F.
• Allowed motions
– Solid: vibration + multiple rotational sub-modes (≈15 possibilities).
– Liquid: solid motions + translational flow (≈16 total).
– Gas: liquid motions + unrestricted free random flight (≈17).
• Relative speed (v)
– Solid: slow.
– Liquid: faster.
– Gas: fastest.
• Shape & Volume (when poured/transferred)
– Solid: fixed shape & fixed volume.
– Liquid: variable shape (takes container), fixed volume.
– Gas: variable shape & variable volume (expands to fill container).
• Compressibility
– Solid/Liquid: nearly incompressible.
– Gas: highly compressible.
Intermolecular Forces & Coulomb’s Law Refresher
• Force between two molecules derives from charge distributions:
.
• In solids/liquids, small d → strong F → collective cohesion.
• In gases, large d makes ; Van der Waals forces practically absent.
Compressibility, Shape, & Volume – Practical Consequences
• A sample of gas released into a room will expand to fill it; P drops.
• Equivalent liquid keeps volume constant; solid keeps shape/volume unless mechanically broken.
• Engineering, meteorology, & respiration models must therefore treat gases separately.
Ethical / Philosophical / Real-World Touchpoints
• Conceptualizing “empty space” in air highlights scale differences between human experience and molecular reality—useful for public understanding of climate, aerosols, or virus transport.
• Perfect-elasticity assumption simplifies thermodynamic proofs but reminds us that models make deliberate approximations.
Key Numerical Facts to Memorize
• .
• v̄ (O₂ at room T) ≈ 1{,}700\ \text{km·h}^{-1}.
• Collision frequency ≈ .
• Pressure unit used in app demo: 1\ \text{MPa} = 10^6\ \text{N·m}^{-2}.
Exam-Oriented Connections & Tips
• Distinguish “postulates” (composition, motion, elasticity) from “implications” (T ↔ ⟨KE⟩, P ↔ wall force).
• Be ready to rank solids/liquids/gases on compressibility, Van der Waals gap, KE, shape retention.
• Mean free path & molecular scaling questions often test intuition – remember 6-ft → 6-mile analogy.
• For gas law derivations (Chapter 18/19), recall that changing T, N/V, or container size directly alters collision rate, thus P.