BIOC*2580 - 7
Coenzymes and Cofactors Discussion
Recap of Last Session
NAD and NADP discussed previously.
Examined redox chemistry of NAD and NADP, establishing similarities.
Structure of Russell Group (Last Slide)
Display of redox chemistry for pyridine nucleus as NAD(P)+.
NADP depicted as positively charged.
Differences in Roles: NAD vs. NADP
NAD (Nicotinamide Adenine Dinucleotide)
Generally used as a cofactor in oxidation reactions (catabolic processes).
Catabolic Processes: Include:
Lipid oxidation.
Carbohydrate oxidation.
NAD+ functions as an oxidizing agent in catabolic reactions.
Accepts hydrogen atoms/electrons during oxidation.
NAD is converted to NADH, which holds energy.
reoxidized in the ETC
NADH participates in electron transport chain to produce ATP.
NADP (Nicotinamide Adenine Dinucleotide Phosphate)
Rarely acts as an oxidizing agent.
Primarily association with NADPH, which is involved in synthetic reactions.
NADPH is commonly regarded as a reducing agent in anabolic processes (building macromolecules).
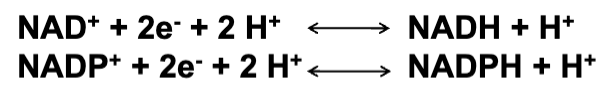
FAD and FMN as Flavine Nucleotides
Overview
Derived from Vitamin B2 (Riboflavin).
Deficiencies can inhibit reactions reliant on FAD or FMN.
Prosthetic Groups vs. Free Cofactors
FAD and FMN function as prosthetic groups, covalently attached to their enzymes.
NAD and NADP exist as free molecules in cytoplasm, not attached to enzymes.
Structure of FAD and FMN
FAD: Flavine adenine dinucleotide, a dinucleotide made of two nucleotides.
Structure includes a base component, sugar, and phosphate.
FMN: Flavine mononucleotide, a single nucleotide similar to one of the FAD components.
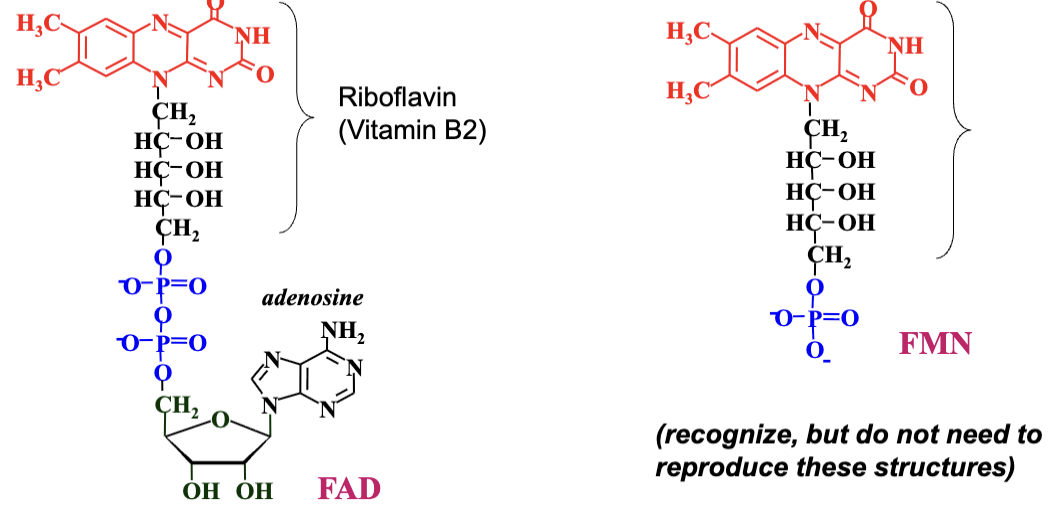
Redox Chemistry of Flavine Nucleotides
Flavine nucleotides (FMN and FAD) can participate in one-electron or two-electron oxidations of substrate
Hydrogen atoms
they are involved in a greater diversity of reactions than the NAD(P)-linked dehydrogenases.
NAD/NADP, by contrast, only participate in two-electron oxidations (removing two hydrogen atoms).
The fully reduced form of the nucleotides are FADH2 and FMNH2 and when only one electron is accepted they form the stable semiquinone radical forms FADH· and FMNH
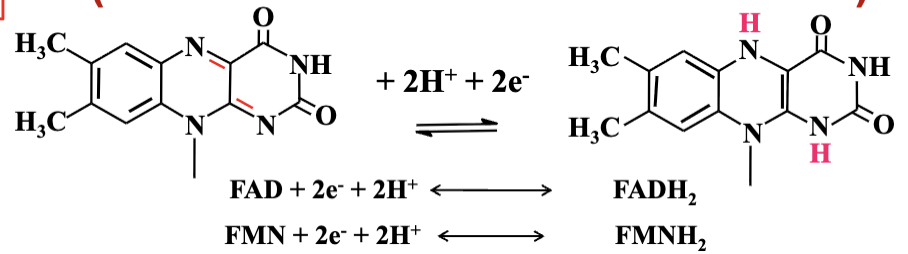
Reaction Result: FAD.
FAD to FADH2 involves acceptance of two hydrogen atoms during full reduction.
Acceptance of both protons leads to FADH2, storing energy for ATP generation.
Importance of B Vitamins and Coenzymes
Vitamin B2 deficiency leads to reduced energy production.
Coenzymes support metabolic processes and reactions.

Metabolism: Understanding Fat Catabolism
Energy Yield from Biomolecules

Energy Values (in kJ per gram dry weight)
Fats yield almost twice the energy of carbohydrates or proteins when oxidized.
Fats identified as the most concentrated energy storage in biological systems.
Structural Analysis of Fatty Acids
Reductiveness of Fatty Acids
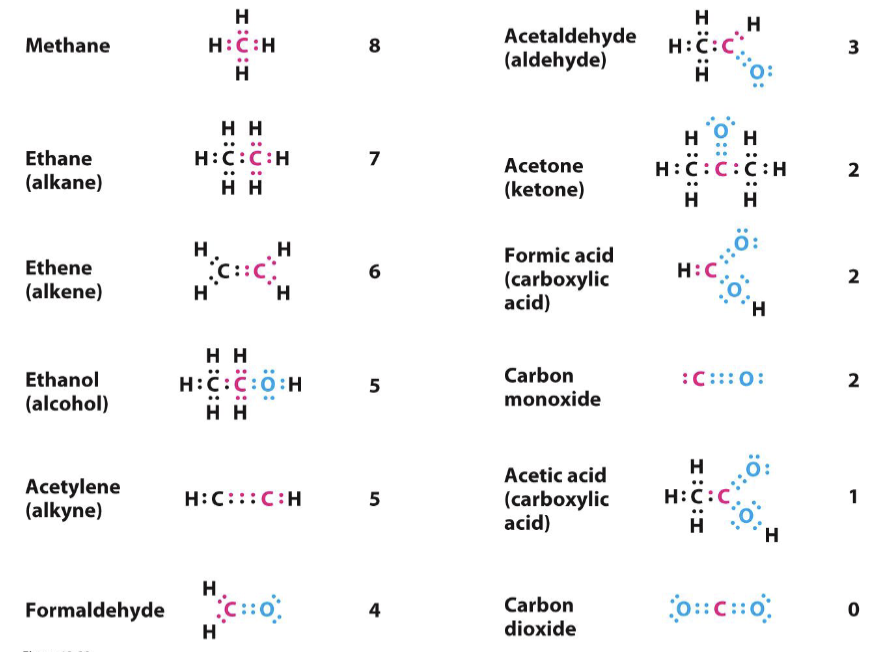
Fatty acids composed primarily of CH2 (methylene) groups, regarded as highly reduced.
High reduction states imply higher energy yields when oxidized to CO2.
Fats vs. Carbs/Proteins
Carbohydrates have alcohol/hydroxyl groups; hence, are already partially oxidized compared to fats.
Resulting in lesser energy yield per gram when fully oxidized to CO2.
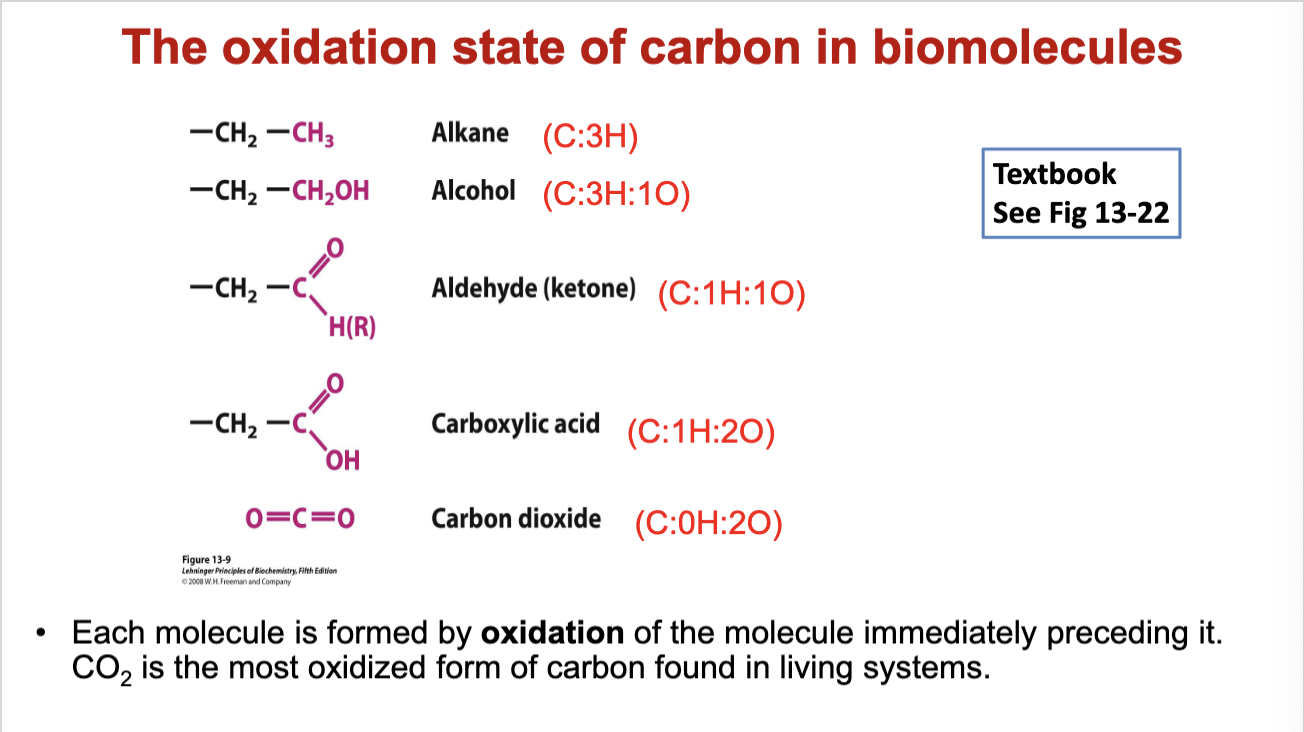
Oxidation States of Carbon
Oxidation states defined by the composition of hydrogens and oxygens attached to carbon.
Fully oxidized carbon exists as CO2; fully reduced as alkanes (only H attached).
Rate of Energy Release Through Oxidation
Implication on Energy Yield
More reduced states (e.g., alkanes) yield more energy upon complete oxidation compared to alcohol or acids.
Higher energy extractible from reduced compounds informs metabolic planning during fasting.
Availability of Energy Sources During Fasting
Prioritization:
Glucose: Immediate source, limited energy.
Glycogen: Higher energy reserve, but requires hydrolysis.
Proteins: Used in extreme fasting, generally not first choice.
Fats: Largest energy reserve, challenging to mobilize.
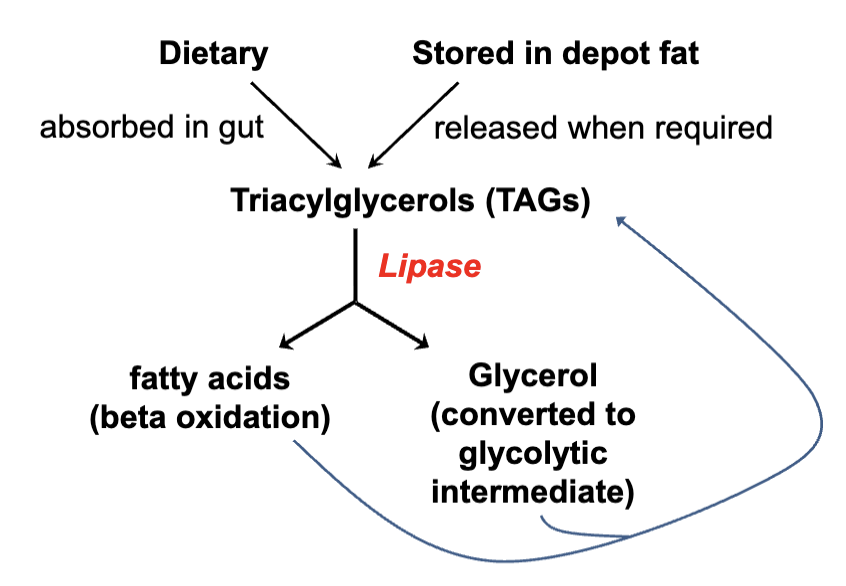
Lipid Catabolism Overview
Storage Form in Body
Fatty acids stored primarily as triglycerides (glycerol + fatty acids).
Triglycerides need to be hydrolyzed for energy release.
Fatty Acid Catabolism Stages
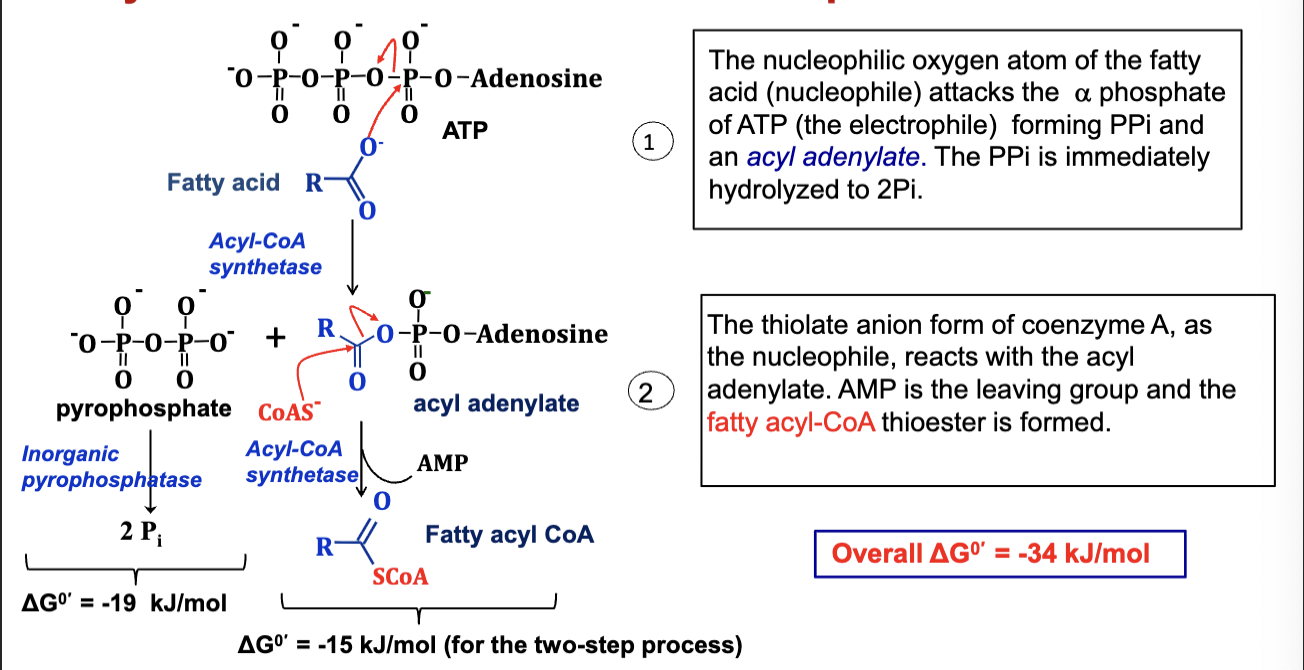
Fatty acids are prepared for catabolism by activating
them to fatty acyl CoA

Activation happens in two steps
Three Stages of Fatty Acid Oxidation
Beta-Oxidation: Partial breakdown to acetyl-CoA.
Citric Acid Cycle (Krebs Cycle): Oxidation of acetyl-CoA to release CO2.
Electron Transport Chain: Reducing NAD and FAD to generate ATP.
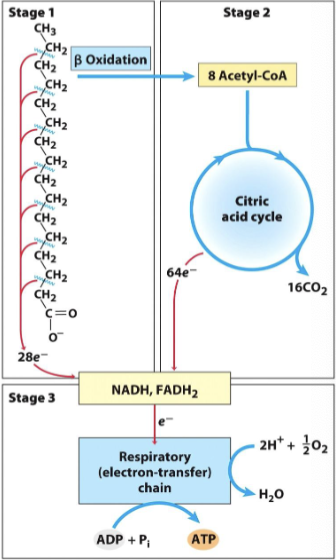
Initial Stage: Beta-Oxidation
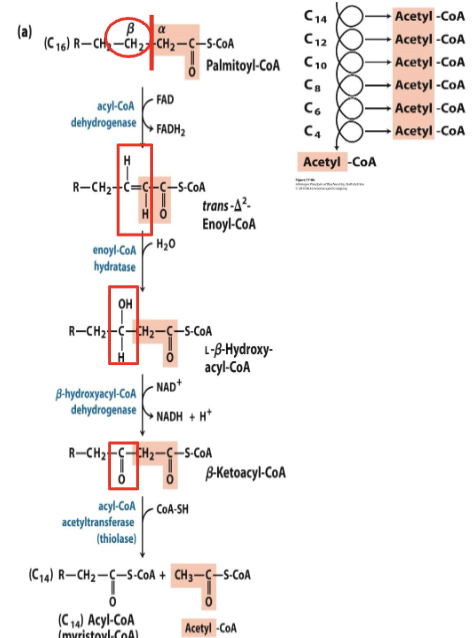
Involves enzymatic action to cleave fatty acids to generate acetyl-CoA.
A cyclic process breaks down fatty acids two carbons at a time.
The fatty acyl CoA molecules, once in
the mitochondrion are committed to
undergo beta-oxidation.Beta oxidation consists of four steps:
oxidation
hydration
oxidation
thiolysis
Each pass through beta oxidation removes one acetyl moiety in the form of acetyl-CoA