2.3 Properties of Water
Structure of Water
Water is polar because it has a slight charge difference across different poles of molecule (dipole)
This allows water to form weak associations (H bonds) with other polar or ionic molecules
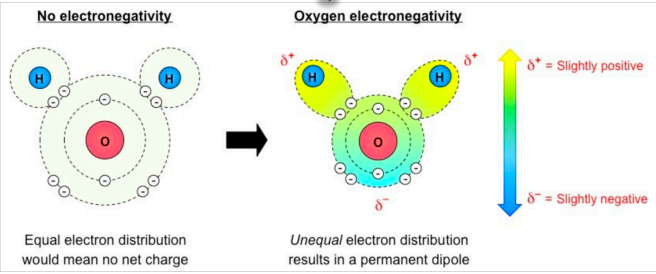 Hydrogen bonding and the angular shape of a water molecule gives it unique properties essential to existence of living things
Hydrogen bonding and the angular shape of a water molecule gives it unique properties essential to existence of living things
Unique Properties of Water
1. Water is an excellent solvent
Solvent - substance that is able to dissolve other substances
Water is a polar molecule
Break ionic bonds
Hydrophilic “water loving” - substances that freely associate and readily dissolve in water (all polar molecules and ions)
Hydrophobic “water hating” - substances that do not freely associate and readily dissolve in water (fats and oils)
e.g. Water soluble substances (NaCl, O2, glucose, ionized aa) can travel freely in blood plasma, whereas water insoluble substances (lipids) cannot (form complexes with proteins - lipoproteins)
2. Water is both adhesive and cohesive
Adhesive - tendency of dissimilar particles/surfaces to cling to one another
Cohesive - tendency of similar or identical particles/surfaces to cling to one another
3. Water has a high specific heat capacity
Specific Heat Capacity - amount of energy needed to change the temperature of 1 kg of a substance by 1°C
Water is slow to change temperature
H-bonds broken before change state (absorb heat)
4. Water has a high heat conductivity
Hydrogen Bonding: Strong hydrogen bonds form between water molecules due to attraction between the partially negative and positive ends, making them relatively robust.
Heat Absorption: When heat is applied, energy is absorbed by breaking these hydrogen bonds, rather than increasing the kinetic energy of individual molecules.
Temperature Resistance: Water's resistance to temperature changes results from the need for a substantial amount of heat energy to break its strong hydrogen bonds.
Efficient Heat Transfer: The increased kinetic energy of water molecules, when heated, is efficiently transferred through hydrogen bonding, making water an excellent conductor of heat.
5. Water has a high boiling/freezing point
Boiling Point: The high boiling point of water results from the need to break numerous hydrogen bonds before transitioning from a liquid to a gas.
Freezing Point: Water's freezing point is elevated due to the formation of a stable and structured network of hydrogen bonds in the liquid state before transitioning to the solid state.
6. Water is more dense as a liquid then as a solid
Unique Molecular Arrangement: Water molecules form a hexagonal lattice structure when transitioning from a liquid to a solid state.
Hydrogen Bonding's Role: Hydrogen bonds cause molecules to arrange themselves with slightly more space in a solid (ice) compared to a liquid (water).
Increased Molecular Spacing: In the solid state, water molecules arrange with a specific structure that leads to increased spacing between them, making ice less dense than liquid water.
Density Inversion: Most substances become denser in their solid form, but water exhibits an anomaly due to the unique properties of hydrogen bonding and molecular arrangement.
Summary
Property | Attributes | Significance |
|---|---|---|
Solvent | Dipolarity means water can dissolve molecules that are polar or charged | Water is a good transport medium (e.g. the blood system can transport soluble materials in its plasma) |
Adhesive | Dipolarity means water will stick to surfaces that are polar or charged | Water can move via capillary action against gravity (e.g. water can move up the xylem via transpiration) |
Cohesive | Dipolarity means water will stick to other water molecules (via H-bonds) | Water has a high surface tension, allowing small organisms to move on its surface (e.g. Basilisk lizard) |
Thermal | Extensive hydrogen bonding means water can absorb a lot of heat before changing state | Water is an excellent coolant (e.g. sweat) Water is a good medium for metabolic reactions (absorbs heat from exothermic reactions) |
Density | Water is less dense as a solid than as a liquid (maximum density ~ 4°C) | Ice floats on water (prevents oceans from freezing as ice layer prevents exposure to cold temperatures) |
Transparency | Water is transparent to visible spectrum | Aquatic plants can undergo photosynthesis |