Unit 1: Biochemistry
Chapter 1: Study of Life
1.1 Science of Biology
Biology: Study of life, very broad definition because scope of biology itself is vast.
Process of Science
Science: knowledge that covers general truths or the operation of general laws, especially when acquired and tested through scientific method.
Scientific Method: Method of research with defined steps that include experiments and careful observations.
Hypothesis: A suggested and testable explanation for an event.
Theory: Tested and confirmed explanation for observations or phenomena, more sound than a hypothesis.
Natural Sciences
→ Fields of science related to the physical world and its processes
Life Sciences: Study of living things, includes biology
Physical Sciences: study nonliving matter and includes astronomy, geology, physics, and chemistry.
Scientific Reasoning
Ultimate goal of all forms of science is “to know”
Two methods of logical thinking:
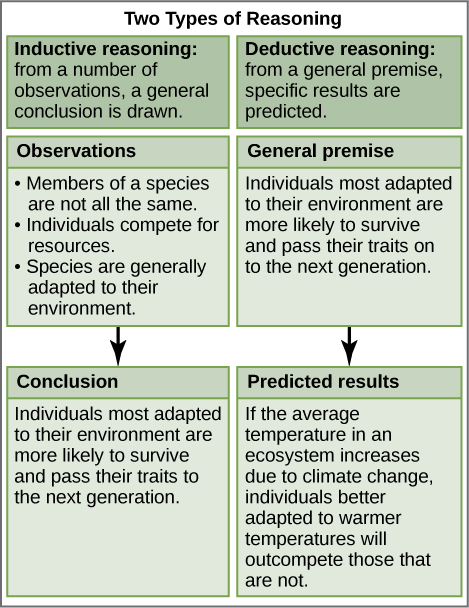
Inductive Reasoning: form of logical thinking that uses related observations to arrive at a general conclusion. Common in descriptive science.
Deductive Reasoning: form of logical thinking that uses a general principle or law to predict specific results.
Descriptive science: usually aims to observe, explore, and discover
Hypothesis-based science: Usually deductive, begins with a specific question or problem and a potential answer or solution that one can test.
Most scientific endeavors utilize both methods.
Ex. A man inspected a cocklebur’s gripping device and found it to be more reliable than a zipper. He experimented and found the best material that acted similarly and eventually produced velcro.
Scientific Method
→ First observed by Sir Francis Bacon
Typically starts with a problem that needs to be solved.
Ex. Student walks into a classroom and notices it is too warm. Her question is “why is the classroom so warm?”
Proposing a Hypothesis
A hypothesis is a suggested explanation that can be tested.
Ex. “The classroom is warm because no one turned on the air conditioning”
When a hypothesis is selected then a prediction is made
It usually has the format “If… Then…”
Ex. “If the student turns on the AC, then the classroom will no longer be too warm”
Testing a Hypothesis
A valid hypothesis should be testable and falsifiable.
Falsifiable: experimental results can disprove it.
Science doesn’t claim to prove anything because scientific understandings are always subject to modification with further information.
Variable: Any part of the experiment that can vary or change during the experiment.
Control group: Contains every feature of the experimental group except it is not given the manipulation.
If an experimental groups results differ from the control group, it must be because of the manipulation as opposed to an outside factor.
The warm classroom example is based on observational results, other hypotheses and experiments might have clearer controls.
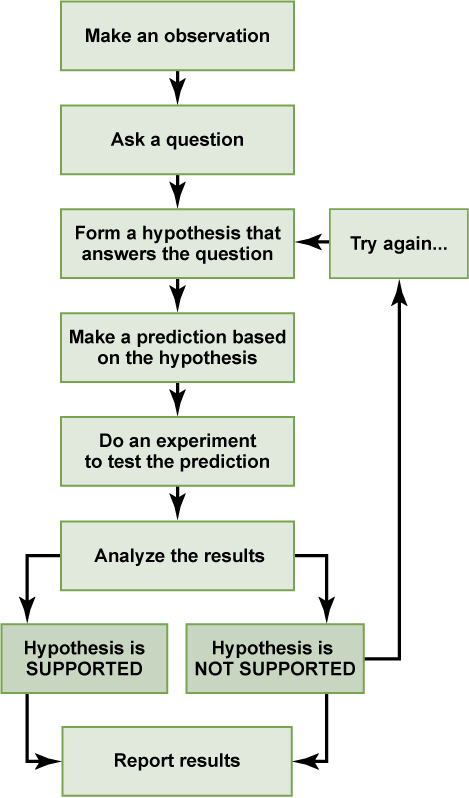
There is flexibility in the scientific method.
Two Types of Science: Basic Science and Applied Science
Basic Science: “pure” science seeks to expand knowledge regardless of the short-term application of that knowledge. It is not focused on developing a product or service of immediate public or commercial value.
Knowledge for knowledge’s sake
Applied Science: “technology”, aims to use science to solve real world problems. Problem is usually defined for the researcher.
Reporting Scientific Work
Peer-reviewed Manuscript: Scientific papers that are reviewed by a scientist’s peers.
Scientific writing must be brief and concise.
Scientific papers consists of specific sections, sometimes called the IMRaD format.
Introduction, Materials, and Methods.
Introduction: Brief background information about what is known in the field.
Review Articles: Don’t follow the IMRaD format because they don’t present original scientific findings or primary literature.
1.2 Themes and Concepts of Biology
Properties of Life
All living organisms share 8 characteristics:
Order
Organisms are highly organized coordinated structures that consist of one or more cells.
Cells make up tissues, tissues make up organs, and organs make up organ systems.
Sensitivity or Response to Stimuli
Plants can bend toward a light source or climb fences or respond to touch.
Bacteria can move toward or away from chemicals.
Movement towards a stimulus is a positive response, but movement away is a negative response.
Reproduction
Single celled organisms duplicate their DNA and divide it equally as the cell prepares to divide to form two new cells.
When reproduction occurs, DNA containing genes are passed along to offspring.
Genes help make sure that offspring will belong to the same species and will have similar characteristics
Adaptation
All living organisms have a “fit” for their environment.
This fit is called adaptation and is a consequence of evolution by natural selection.
Growth and Development
Organisms grow and develop as a result of genes providing specific instructions that will direct cellular growth and development.
Regulation/ Homeostasis
Homeostasis: refers to the relatively stable internal environment required to maintain life.
Organisms can maintain homeostatic internal conditions within a narrow range almost constantly, despite environmental changes.
Energy Processing
All organisms use an energy source for metabolic activities.
Some use energy from the sun, others use chemical energy
Evolution
Diversity of life on Earth is a result of mutations in hereditary material over time.
These mutations allow for adaptations to changing environments.
Levels of Organization of Living Things
Atom: smallest and most fundamental unit of matter that retains properties of an element.
Consists of a nucleus surrounded by electrons.
Molecules: Chemical structure consisting of at least two atoms held together by one or more chemical bonds.
Macromolecules: Large molecules that are typically formed by polymerization (polymer is a large molecule that is made by combining smaller units called monomers)
DNA is a macromolecule
Organelles: small structures that exist within the cells. Examples include mitochondria and chloroplasts.
Cell: Smallest fundamental unit of structure and function in living organisms.
Prokaryotes: Single celled or colonial organisms that do not have membrane-bound nuclei.
Eukaryotes: Do have membrane bound organelles and a membrane bound nucleus.
Branches of Biological Study
Molecular Biology and Biochemistry: Study biological processes at the molecular and chemical level, including interactions among molecules such as DNA, RNA, and proteins, as well as the way they are regulated.
Microbiology: Study of microorganisms. Studies structure and function of single celled organisms.
Chapter 2: The Chemical Foundation of Life
2.1 Atoms, Isotopes, Ions, and Molecules: The Building Blocks
Matter: Any substance that occupies space and has mass
Elements: Unique forms of matter with specific chemical and physical properties that cannot break down into smaller substances by ordinary chemical reactions.
Each element is represented by a chemical symbol, which is a single capital letter or a combination of two letters.
Four elements common to all living organisms: Oxygen (O), Carbon (C), Hydrogen (H), and Nitrogen (N)
Structure of an Atom
Atom: Smallest unit of matter that retains all the element’s chemical properties. Comprised of two regions:
Nucleus: Atom’s center and contains protons and neutrons
They also hold electrons in orbit around the nucleus
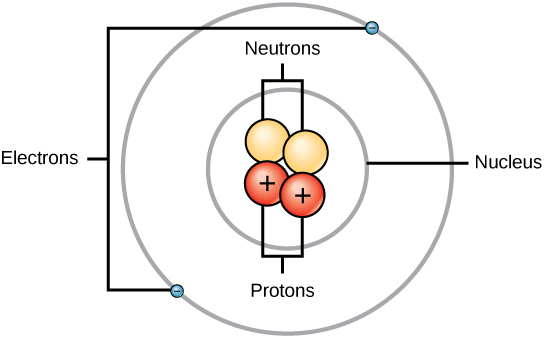
Protons and neutrons have about the same mass
Scientists define this amount of mass as one Atomic Unit, because electron mass rarely impacts the overall mass of the atom.
Protons: Positively charged
Neutron: Uncharged
Number of neutrons in an atom contributes to its mass but not its charge.
Electrons: much smaller in mass than protons, about 1/1800 of an AMU.
Atomic Number and Mass
Atomic Number: determined by the number of protons, used by scientists to distinguish one element from another.
Number of neutrons is variable, which results in isotopes.
Mass Number: number of protons and neutrons combined.
Atomic Mass: Calculated mean of the mass number for its naturally occurring isotopes.
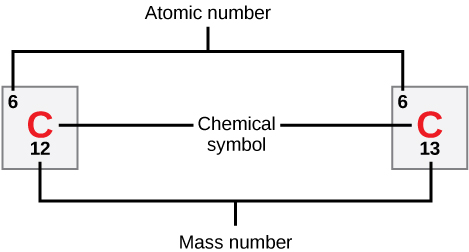
Isotopes
Isotopes: Different forms of an element that have the same number of protons but a different number of neutrons.
Some elements, like carbon, have naturally occurring isotopes.
Radioisotopes: Radioactive isotopes that emit neutrons, protons, and electrons to attain a more stable atomic configuration.
Periodic Table
Periodic Table: Organizes and displays different elements.
Chemical Reactivity: Ability to combine and to chemically bond with other elements.
Molecules: Two or more atoms that chemically bond together.
Electron Shells and the Bohr Model
Electrons fill orbitals in a consistent order, starting with the orbital closest to the nucleus and working outward.
Octet Rule: Atoms are more stable when their outer shell (the valence shell) has eight electrons.
Electron Orbitals
Electron Orbitals: complex shapes resulting from the fact that electrons behave not just like particles, but also like waves.
The closest orbital to the nucleus can hold up to two electrons
Chemical Reactions and Molecules
Chemical Bond: Process of atoms obtaining electrons they need to attain a stable electron configuration.
Atoms can form molecules by donating, accepting, or sharing electrons to fill their outer shells.
Chemical Reactions: two or more atoms bond together to form molecules or bonded atoms break apart.
Reactant: substances used in beginning of a chemical reaction
Products: Substances at the end of the reaction.
Compound: Contain atoms of more than one type of element.
Ions and Ionic Bonds
Cation: Positive ions that form by losing electrons.
Anions: Negative ions formed by gaining electrons. Designated by elemental name and changing ending to “ide”.
Electron Transfer: Movement of electrons from one element to another
Ionic Bonds: Form between ions with opposite charges
Covalent Bonds and Other Bonds and Interactions
Covalent Bonds: sharing of electrons between atoms, stronger and much more common than ionic bonds.
The more covalent bonds between two atoms, the stronger their connection
Polar Covalent Bonds
Polar Covalent Bond: Atoms unequally share the electrons as the electrons are attracted more to one nucleus than the other.
Because of unequal electron distribution between atoms of different elements, a slightly positive or negative charge will develop.
Water is a polar molecule.
Polar bonds are often present in organic molecules.
Nonpolar Covalent
Nonpolar Covalent Bonds: form between two atoms of the same element or between different elements that share electrons equally.
Hydrogen Bonds and Van Der Waals Interactions
Ionic bonds are not as strong as covalent bonds, which determines their behavior in biological systems.
Hydrogen Bond: Common and regularly occurs between water molecules. Individual hydrogen bonds are weak but they occur in large numbers in water, creating a major force in combination.
They are also responsible for zipping together DNA double helix.
Van der Waals Interactions: weak attractions or interactions between molecules. Can occur between any two or more molecules and are dependent on slight fluctuations of electron densities.
2.2 Water
→ Water is essential to all life
Water’s Polarity
Water is composed of polar molecules: Hydrogen and Oxygen
They form polar covalent bonds
Water generates charges because oxygen is more electronegative than hydrogen, making it more likely that a shared electron would be near the oxygen nucleus than the hydrogen nucleus
This means that there is a partial negative charge near oxygen.
Hydrophilic: polar substance that interacts readily with or dissolves in water (water loving)
Nonpolar molecules, such as oils and fats, do not like water.
Hydrophobic: nonpolar compounds that do not interact well with water.
Water’s Solvent Properties
Ions and polar molecules can readily dissolve in water because it has a slightly positive and negative charge.
Solvent: Substance capable of dissolving other polar molecules and ionic compounds.
Dissociation: Occurs when atoms or groups of atoms break off from molecules and form ions.
Water’s Cohesive and Adhesive Properties
Cohesion: Water molecules are attracted to each other, keeping the molecules together at the liquid-gas interface.
Surface Tension: Capacity of a substance to withstand rupturing when placed under tension or stress.
Why water forms droplets on a dry surface.
Adhesion: attraction between water molecules and other molecules.
Capillary Action: Water molecules are attracted to the capillary’s charged glass walls more than they are to each other and therefore adhere to it and climb up the small tube.
2.3 Carbon
Organic Molecules: any carbon-containing solid, liquid, or gas that are important for life.
Carbon atom has unique properties that allow it to form up to four covalent bonds
Those 4 bonds satisfies the octet rule.
Hydrocarbons
Hydrocarbon: organic molecules consisting entirely of carbon and hydrogen, like methane (CH4)
Covalent bonds within a hydrocarbon stores a lot of energy, which releases when they are burned
Methane is described as having a tetrahedron shape because it has four triangular faces.
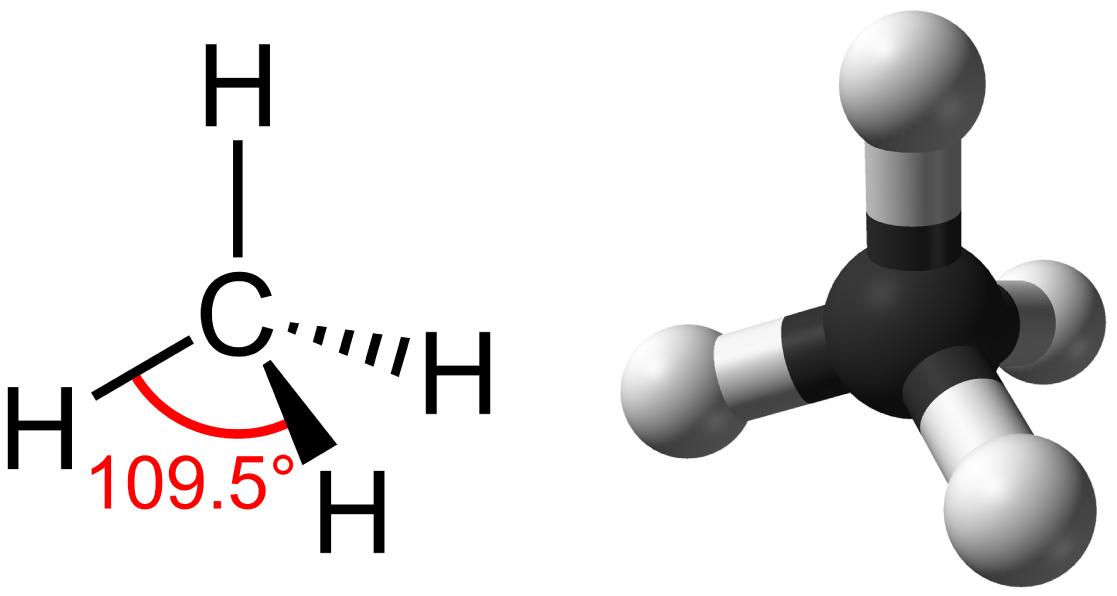
Carbon to carbon bonds may be single, double, or triple covalent bonds, with each type affecting the molecule’s geometry.
Hydrocarbon Chains
Formed by successive bonds between carbon atoms, can be branched or unbranched.
Different number of carbon to carbon bonds determines the shape and can be identified by suffix.
Ex. Ethane, Ethene, and Ethyne
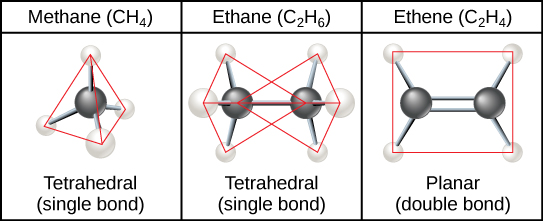
Hydrocarbon Rings
Aliphatic hydrocarbons: consist of linear chains of carbon atoms, can sometimes form rings with all single bonds.
Aromatic Hydrocarbons: consists of closed rings of carbon atoms with alternating single and double bonds.
Isomers
Isomers: molecules that share the same chemical formula but differ in the placement of their atoms and/or chemical bonds.
Structural Isomers: differ in placement of their covalent bonds; both molecules have four carbons and 10 hydrogens, but the different atom arrangement within the molecules leads to differences in their chemical properties.
Ex. Butane can be used as a fuel for cigarette lighters, but isobutane is used as a refrigerant.
Geometric Isomers: have similar placements of their covalent bonds but differ in how these bonds are made to the surrounding atoms, especially in carbon-to-carbon double bonds.
Enantiomers
Enantiomers: Molecules that share the same chemical structure and chemical bonds but differ in the three dimensional placement of atoms so that they are non-superimposable mirror images.
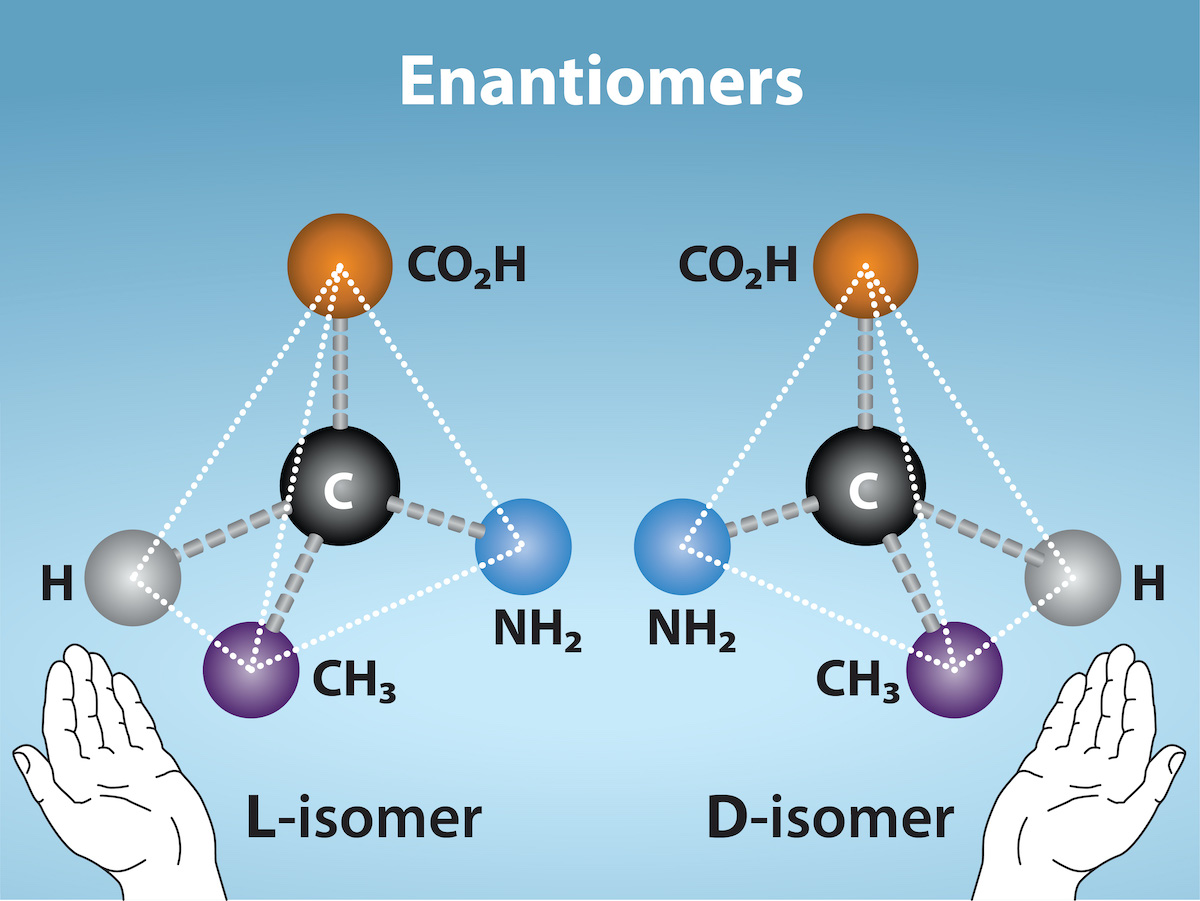
Functional Groups
Functional Groups: Groups of atoms that occur within molecules and confer specific chemical properties to those molecules.
Substituted Hydrocarbons: Molecules with other elements in their carbon backbone.
Functional group can participate in specific chemical reactions.
Functional groups are usually classified as hydrophobic or hydrophilic, depending on charge or polarity.
Hydrogen bonds between functional groups are important to the function of many macromolecules and help them to fold properly into and maintain the appropriate shape for functioning.
Chapter 3: Biological Macromolecules
3.1 Synthesis of Biological Macromolecules
Biological Macromolecules: large molecules that are necessary for life and built from smaller organic molecules.
Four major biological macromolecule classes, which are carbohydrates, lipids, proteins, and nucleic acids.
Dehydration Synthesis
Monomers: single subunits that combine with each other using covalent bonds to form larger molecules known as polymers.
Because of this, monomers release water molecules as byproducts.
Dehydration Synthesis: Means “to put together while losing water”
Hydrogen of one monomer combines with the hydroxyl group of another monomer, releasing a water molecule.
Hydrolysis
Polymers break down into monomers during hydrolysis.
During these reactions, polymer breaks into two components, with one part gaining a hydrogen atom (H+) and the other gaining a hydroxyl molecule (OH-)
Hydrolysis Reactions: sped up by specific enzymes; break bonds and release energy.
3.2 Carbohydrates
→ Carbohydrates play an important role in providing energy for our bodies, especially natural carbohydrates from fruits, vegetables, and grains.
Molecular Structures
Carbohydrates: Represented by the stoichiometric formula (CH2O)n, where n is the number of carbons in the molecule
Ratio of carbon to hydrogen to oxygen is 1:2:1
Carbohydrates are classified into three subtypes:
Monosaccarides
Disaccharides
Polysaccharides
Monosaccarides
Monosaccarides: Simple sugars, where mono means one and saccar means sweet.
Most common is glucose
Number of carbons can range from 3 to 7
Most monosaccaride names end with -ose
Aldose: If sugar has an aldehyde group
Ketose: If sugar has a ketone group.
Chemical formula for glucose is C6H12O6
Glucose is important during cellular respiration because it releases energy helps make ATP
Monosaccharides can exist in a linear chain or as ring shaped molecules.
Disaccharides
Disaccharides: (di=two), form when two monosaccharides undergo a dehydration reaction
Glycosidic Bond: A monosaccharide’s hydroxyl group combines with another monosaccharide’s hydrogen, releasing a water molecule and forming a covalent bond.
Common disaccharides include:
lactose: Consists of monomers glucose and galactose, found in milk.
maltose: malt sugar, formed by dehydration reaction between two glucose molecules.
sucrose: table sugar, formed from glucose and fructose monomers.
Polysaccharides
Polysaccharides: Long chain of monosaccharides lined by glycosidic bonds (poly=many)
Examples of polysaccharides:
Starch
Glycogen
Cellulose
Chitin
Plants store sugars in the form of starch. They can synthesize glucose and store excess in different parts of the plant.
Glycogen: storage form of glucose in humans and other vertebrates and is comprised of monomers of glucose.
Animal equivalent of starch, usually stored in liver and muscle cells.
Cellulose: Most abundant natural biopolymer, mostly comprises a plant’s cell wall.
Chitin: makes up the exoskeleton of arthropods, nitrogen containing polysaccharide. Also a major component of fungal cell walls.
Benefits of Carbohydrates
Carbohydrates have been an important part of the diet for thousands of years.
We should supplement carbohydrates with proteins, vitamins, and fats.
Without consuming carbohydrates, we reduce the availability of “instant energy”
Eliminating carbs from the diet may be beneficial to some, but for others it may not be healthy.
3.3 Lipids
Lipids: Include a diverse group of compounds that are largely non polar in nature. This is because they are hydrocarbons that include mostly non-polar carbon-carbon or carbon-hydrogen bonds.
Lipids perform many different functions in a cell and provide insulation from the environment for plants and animals.
Fats and Oils
Fat molecule consists of two main components-glycerol and fatty acids.
Glycerol is an organic compound (alcohol) with three carbons, five hydrogens, and three hydroxyl groups.
Triacylglycerols: Another name for fats, given because of their chemical structure.
Palmitic acid is a saturated fatty acid and derived from the palm tree.
Fatty acids may be saturated or unsaturated.
If there are only single bonds between neighboring carbons in the hydrocarbon chain, the fatty acid is saturated.
Saturated with hydrogen
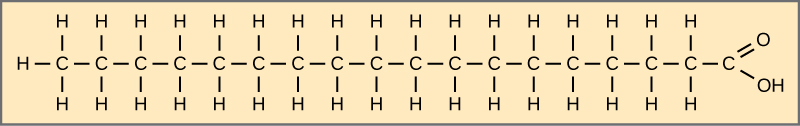
When the hydrocarbon chain contains a double bond, the fatty acid is unsaturated.
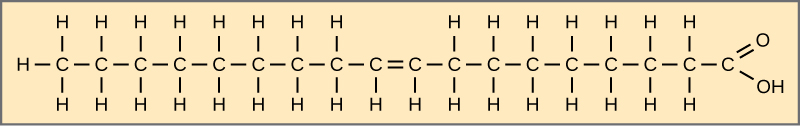
Most unsaturated fats are liquid at room temperature, called oils.
If there is one double bond in the molecule, then it is monounsaturated. (olive oil)
If there is more than one double bond, it is polyunsaturated.
Cis and Trans indicate the configuration of the molecule around the double bond.
If hydrogens are present in the same plane, it is a cis fat
If the hydrogen atoms are on two different planes, it is a trans fat.
Trans Fats
Food industry artificially hydrogenates oils to make them semi-solid and of a consistency desirable for many processed food products
Hydrogen gas is bubbled through oils to solidify them. This causes the double bonds of the cis- conformation in the hydrocarbon chain to convert to double bonds in the trans- conformation
Omega Fatty Acids
Omega-3 Fatty Acids: Required by the human body but can’t be synthesized. One of only two known for humans.
Polyunsaturated fatty acids and are omega-3 because a double bond connects the third carbon from the hydrocarbon chain’s end to its neighboring carbon.
Research indicates that omega-3 fatty acids reduce the risk of sudden death from heart attacks, lower triglycerides in the blood, decrease blood pressure, and prevent thrombosis.
Fats serve as a long term storage of form of fatty acids.
Waxes
Wax: covers some aquatic birds feathers and some plants’ leaf surfaces.
Prevent water from sticking on the surface.
Phospholipids
Phospholipids: Major plasma membrane constituents that comprise cells’ outermost layer.
Phospholipid is an amphipathic molecule, meaning it has a hydrophobic and hydrophilic part.
Fatty acid chains are hydrophobic and cannot interact with water
Phosphate-containing group is hydrophilic and interacts with water.
Steroids
Steroids: Have a fused ring structure, don’t resemble other lipids but scientists group them together because they are hydrophobic and insoluble in water.
All steroids have four linked carbon rings and several of them have a short tail.
Cholesterol is the most common steroid
Liver synthesizes cholesterol and is the precursor to steroid hormones such as testosterone and estradiol.
Cholesterol is necessary for the body’s proper functioning.
3.4 Proteins
Proteins: One of the most abundant organic molecules in living systems and have most diverse range of functions of all macromolecules.
Can be structural, regulatory, contractile, or protective.
Their functions can vary but they all are made up of amino acid polymers arranged in a linear sequence.
Types and Functions of Proteins
Enzymes: living cells produce these, catalysts in biochemical reactions and are complex proteins.
Enzymes that break down their substrates are called catabolic enzymes
Those that build complex molecules from their substrates are anabolic enzymes.
Those that affect the reaction rate are catalytic enzymes.
Hormones: chemical signaling molecules, usually small proteins or steroids, secreted by endocrine cells that act to control or regulate specific physiological processes.
Proteins have different shapes and molecular weights.
Denaturation: loss of function caused by changes in temperature, pH, and exposure to chemicals. Can lead to change in proteins shape.
Amino Acids
Amino Acids: Monomers that comprise proteins. Each amino acid has the same fundamental structure, which consists of a central carbon atom bonded to an amino group, a carboxyl group, and a hydrogen atom.
Every amino acid also has another atom bonded to the central atom known as the R group
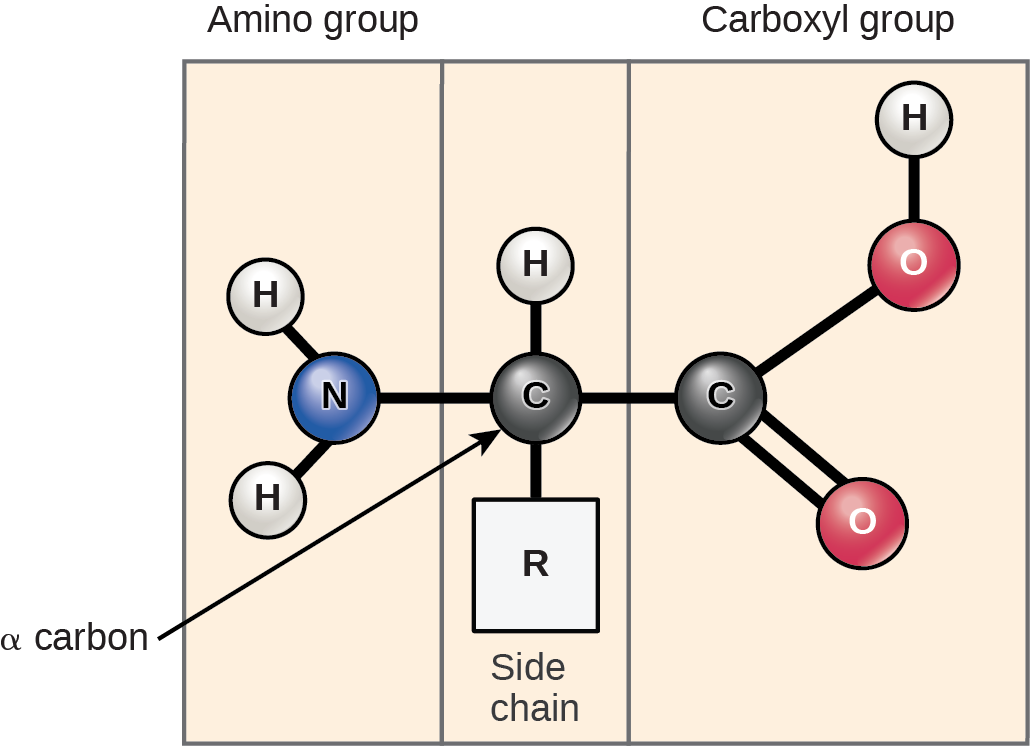
Scientists use the name amino acid because these acids contain both amino group and carboxyl-acid-group in their basic structure.
Chemical nature of side chain determines the amino acid’s nature
Acidic, basic, polar, or nonpolar
Peptide Bond: Covalent bond that attaches to each amino acid, which a dehydration reaction forms. One amino acid’s carboxyl group and the incoming amino acid’s amino group combine, releasing a water molecule and forming a peptide bond
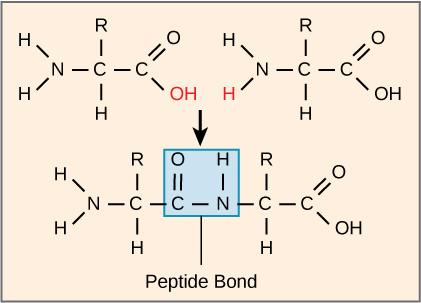
As more amino acids join to the growing chain, the result is a polypeptide.
Protein Structure
Shape is critical to function
form=function
To understand this, we need to understand four levels of protein structure
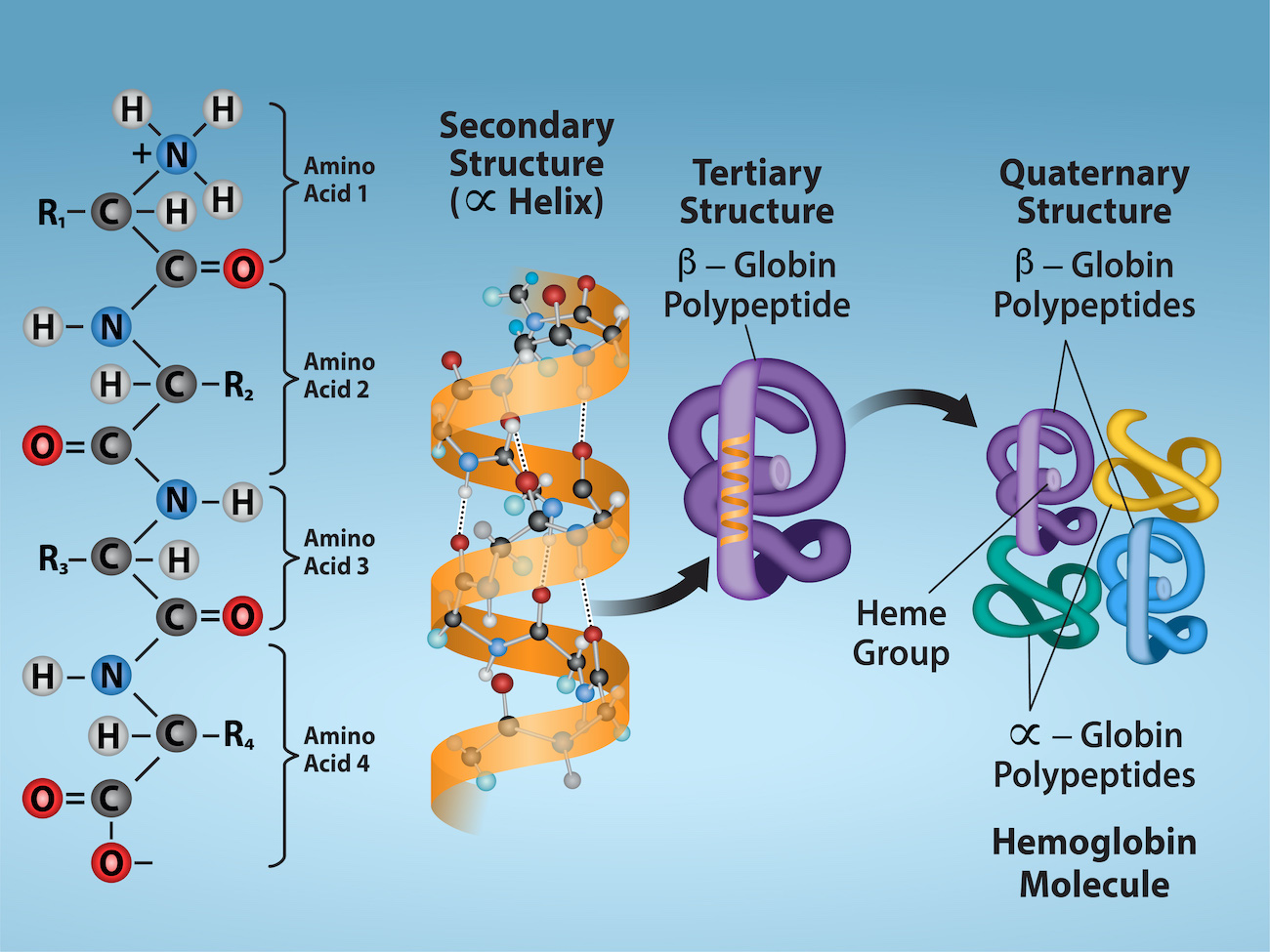
Primary Structure
Primary structure: The unique sequence of an amino acid in a polypeptide chain.
The gene encoding the protein determines the unique sequence for every protein
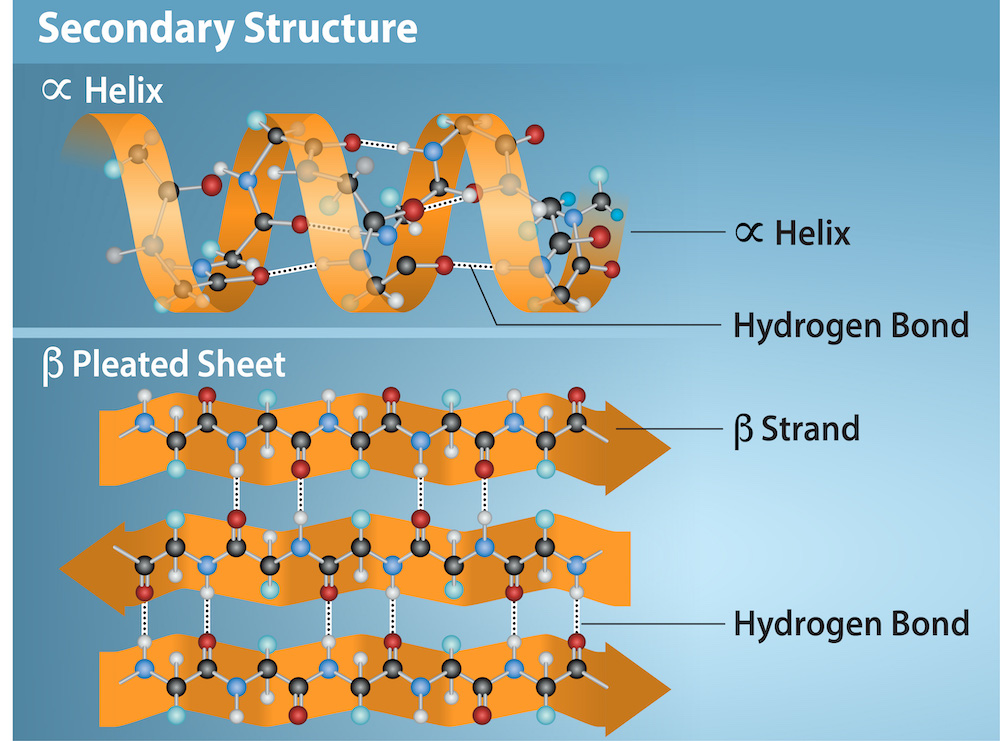
Secondary Structure
Secondary Structure: Local folding of the polypeptide in some regions. Most common are:
α-helix
β- pleated sheet
These structures are held in shape by hydrogen bonds. The hydrogen bonds form between oxygen atom in carbonyl group and another amino acid that is four amino acids farther along the chain.
Tertiary Structure
Tertiary Structure: Polypeptide’s unique three dimensional structure
Created mostly by interactions among R groups
R groups with like charges repel each other and those with unlike charges are attracted to each other (ionic bonds)
When protein folding occurs, nonpolar amino acids hydrophobic R group lie in the protein’s interior, whereas the hydrophilic R groups lie on the outside.
Quaternary Structure
Quaternary Structure: Some proteins form from several polypeptides, or subunits, and the interaction of these subunits forms the quaternary structure.
Denaturation and Protein Folding
Denaturation can be reversible because the polypeptide’s primary structure is conserved in the process if the denaturing agent is removed.
Chaperones: Protein helpers that assist in the folding process and associate with the target protein during the folding process.
Act by preventing polypeptide aggregation that comprise the complete protein structure.
They disassociate from the protein once the target protein is folded.
3.5 Nucleic Acids
→ Nucleic Acids are the most important macromolecule for continuing life. They carry the cells genetic blueprint and carry instructions for it to function
DNA and RNA
Two main types of nucleic acids:
Deoxyribonucleic Acid (DNA): Genetic material in all living organisms, ranging from single celled bacteria to multicellular mammals.
In nucleus of Eukaryotes
Ribonucleic Acid (RNA): Mostly involved in protein synthesis.
Messenger RNA (mRNA): used to communicate between DNA molecules and the rest of the cell.
Nucleotides: Monomers that make up DNA and RNA
Nucleotides combine with each other to form a polynucleotide, DNA or RNA
Nucleotides are comprised of a nitrogenous base, a pentose (five carbon sugar, and a phosphate group).
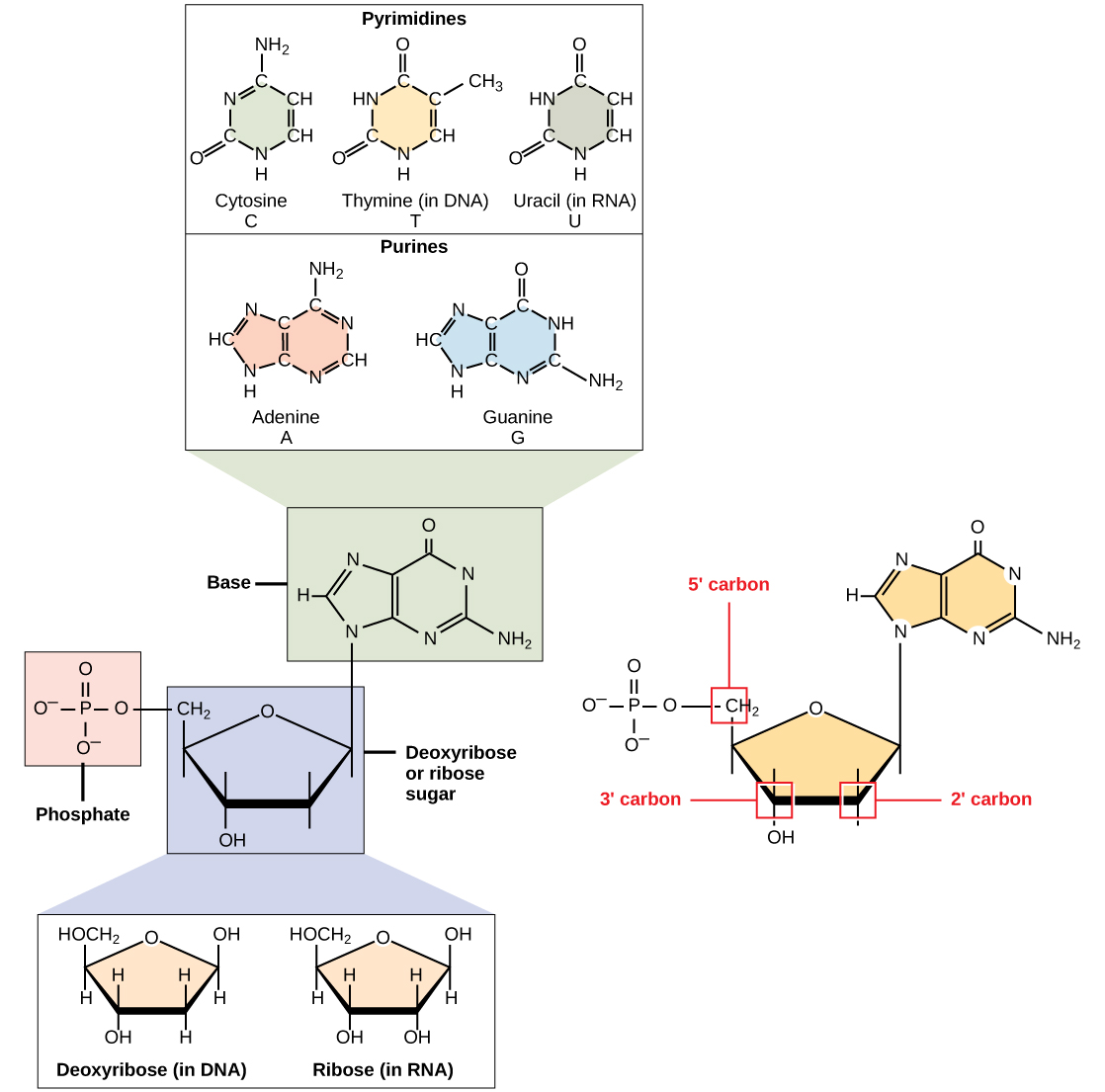
Each nucleotide in DNA contains one of four possible nitrogenous bases: Adenine (A), Guanine (G), Cytosine (C), and Thymine (T)
Adenine and guanine are classified as purines
Cytosine, Thymine, and Uracil are classified as Pyrimidines.
Pentose sugar in DNA is deoxyribose, and in RNA it is ribose.
Difference is the presence of the hydroxyl group on the ribose’s second carbon and hydrogen on the deoxyribose’s second carbon.
Phosphodiester: formed by removing two phosphate groups, a polynucleotide can have thousands of phosphodiester bonds.
DNA Double-Helix Structure
Sugar and phosphate lie on outside of helix, forming the DNA’s backbone.
Nitrogenous bases are stacked in the interior, like a pair of stairs
Hydrogen bonds bind the pairs to each other.
Only certain kinds of base pairings are allowed, like Adenine and Thymine can pair, while Guanine and Cytosine can also pair.
RNA
Mainly involved in protein synthesis under direction of DNA.
Single stranded and comprised of ribonucleotides linked by phosphodiester bonds.
A ribonucleotide contains ribose and one of the four nitrogenous bases, A, U, G, and C.
There are four major types of RNA:
Messenger RNA - Carries message from DNA, read in sets of three bases known as codons, each coding for a single amino acid.
Ribosomal RNA - Major constituent of ribosomes on which the mRNA binds. Ensures proper alignment of the mRNA and the Ribosomes.
Transfer RNA - One of the smallest of the four types of RNA, usually 70-90 nucleotides long. Carries the correct amino acid to the protein synthesis site.
MicroRNA (miRNA) - Smallest RNA molecules and role involves regulating gene expression by interfering with the expression of certain mRNA messages.
Transcription: Process of DNA dictating the structure of mRNA
Translation: Process of RNA dictating the protein’s structure
Chapter 5: Structure and Function of Plasma Membranes
5.1 Components and Structure
Plasma membrane needs to be flexible to allow certain cells, like red and white blood cells, to change shape as they pass through narrow capillaries.
Viruses can hijack receptors and use them to gain entry into cells and sometimes genetic material.
Fluid Mosaic Model
Principal components of a plasma membrane are lipids and proteins.
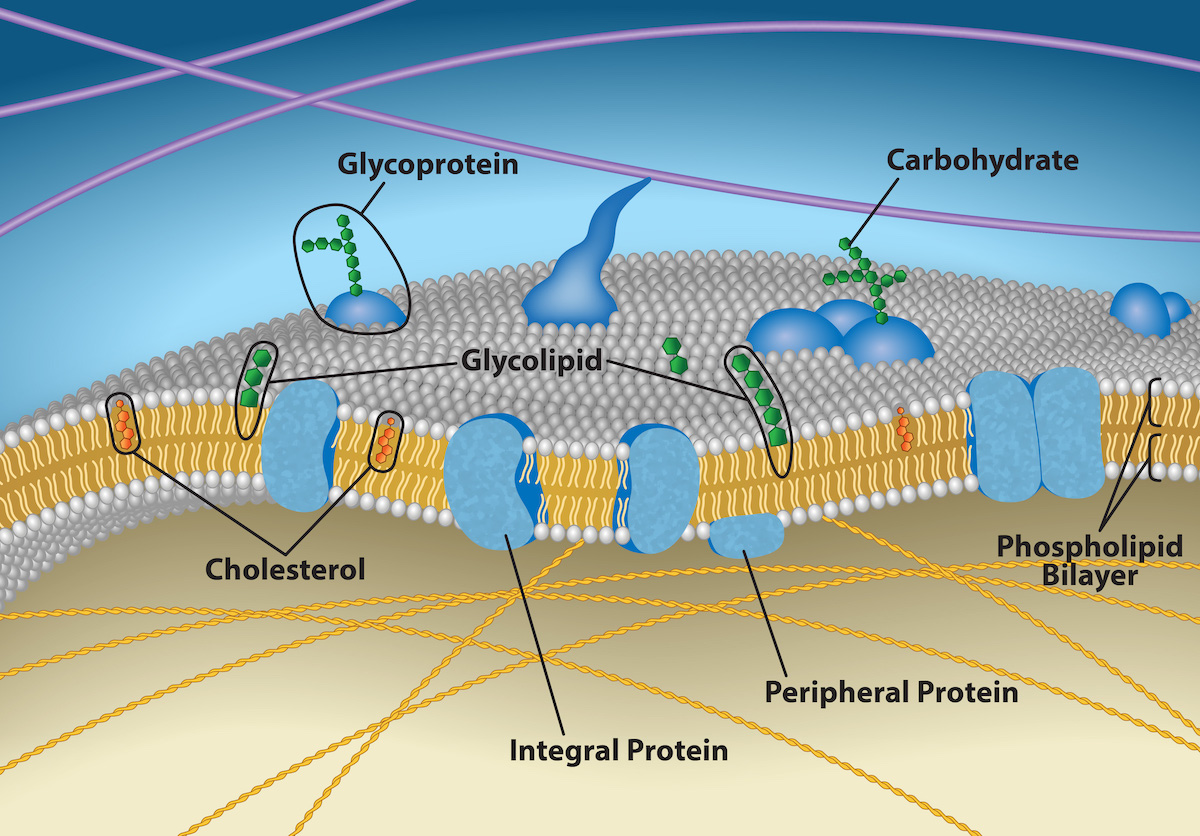
Fluid Mosaic Model: Describes the plasma membrane structure as a mosaic of components, including phospholipids, cholesterol, proteins, and carbohydrates, These give the membrane a fluid character.
Has evolved over time, best accounts for plasma membrane structure and function as we now understand them.
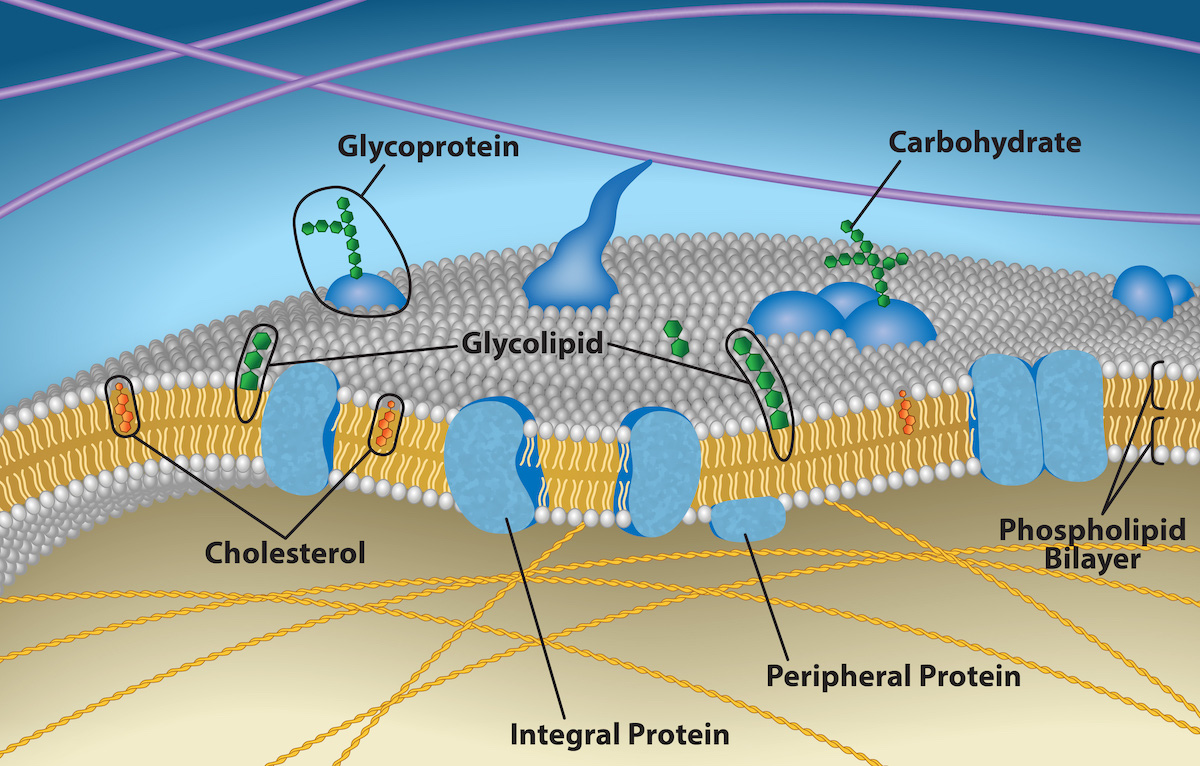
A phospholipid consists of glycerol, two fatty acids, and a phosphate linked head group.
Glycoproteins: Formed when a carbohydrate is attached to proteins
Glycolipids: Formed when a carbohydrates attach to lipids.
Phospholipids
Membrane’s main fabric comprises amphiphilic, phospholipid molecules.
Hydrophilic: Water loving areas of a molecule
Hydrophobic: Water hating molecules, tend to be non polar.
Hydrophobic molecules tend to form a cluster when placed in water.
Pospholipid’s hydrophilic regions form hydrogen bonds with water and the other polar molecules on both the cell’s exterior and interior.
Phospholipid molecule consists of a three carbon glycerol backbone with two fatty acid molecules attached to carbons 1 and 2, and a phosphate containing group attached to the third carbon.
This creates a polar/ negative charge in the head area and a tail area that has no charge.
Amphiphilic: Molecule with a positively or negatively charged area and an uncharged area. Means “dual loving”
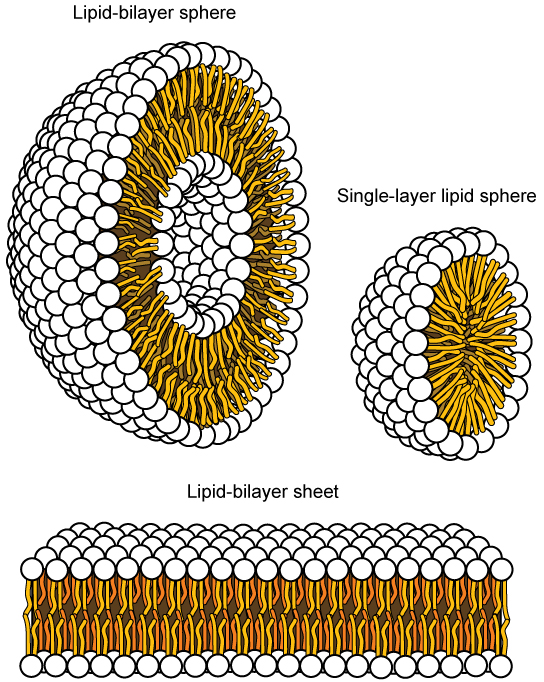
In water, phospholipids arrange themselves with their hydrophobic tails facing each other and their hydrophilic heads facing out.
Forms a lipid bilayer
Proteins
Comprise the plasma membrane’s second major component
Integral Proteins: Integrate completely into the membrane structure and their hydrophobic membrane-spanning regions interact with the phospholipid bilayer’s hydrophobic region.
Peripheral Proteins: On the membrane’s exterior and interior surfaces, attached either to integral proteins or to phospholipids.
Peripheral proteins, along with integral proteins, may serve as:
enzymes
structural attachments for the cytoskeleton’s fibers
part of the cell’s recognition sites.
Carbohydrates
Third major plasma membrane component.
Always on cells’ exterior surface and are bound to proteins or lipids.
Can consist of 2-60 monosaccharide units, straight or branched.
Carbs form specialized sites on the cell surface to allow cells to recognize each other.
This recognition is important to cells because it allows the immune system to differentiate between body cells (self) and foreign cells (non-self)
Carbohydrates on cell’s surface are referred to as glycocalyx, meaning sugar coating.
It is hydrophilic and attracts large amounts of water to the cells surface.
Membrane Fluidity
The membrane is not like a balloon that can expand and contract, but it is fairly rigid and can burst if it is penetrated or if a cell takes in too much water.
Some mosaic characteristics are explained by the fatty acids being saturated with bound hydrogen atoms and having no double bonds between adjacent carbon atoms.
Double bond results in a bend in the carbon string
If decreasing temperatures compress saturated fatty acids, they press in on each other and make a dense, fairly rigid membrane.
If they are unsaturated fatty acids being compressed, they can maintain some space between the phospholipid molecules.
Plasma Membrane Components and Functions.
Component | Location |
Phospholipid | Main membrane fabric |
Cholesterol | Attached between phospholipids and between the two phospholipid layers |
Integral proteins (for example, integrins) | Embedded within the phospholipid layer(s); may or may not penetrate through both layers |
Peripheral proteins | On the phospholipid bilayer's inner or outer surface; not embedded within the phospholipids |
Carbohydrates (components of glycoproteins and glycolipids) | Generally attached to proteins on the outside membrane layer |
5.2 Passive Transport
Plasma membranes are selectively permeable
Selective Permeability: Membrane allows some substances to pass through, but blocks others.
If a cell lost this ability, it would be destroyed.
Passive Transport: Naturally occurring phenomenon that does not require the cell to exert any of its energy to accomplish the movement.
Concentration Gradient: Physical space where there is a single substance concentration range
Selective Permeability
Plasma membranes are asymmetric; membrane interior is not identical to its exterior.
Types of proteins in the membrane:
Lipid Anchor Proteins: anchor membrane to cytoskeleton fibers
Peripheral Proteins: Located on membrane’s exterior and bind extracellular matrix elements.
Carbohydrates help the cell bind required substances in extracellular fluid.
Plasma membranes are amphiphilic; they have hydrophilic and hydrophobic regions.
This helps move some material thru the membrane but limits others.
Fat-soluble vitamins, like A,D,E, and K can easily pass through the plasma membranes in the digestive tract.
Oxygen and carbon dioxide molecules have no charge and pass through the membranes by simple diffusion.
Some ions need a special way to pass through the membrane.
Simple sugars and amino acids need the help of transmembrane proteins to get across the membranes too.
Diffusion
Diffusion: Passive process of transport. A single substance moves from a high concentration to a low concentration area until the concentration is equal across a space.
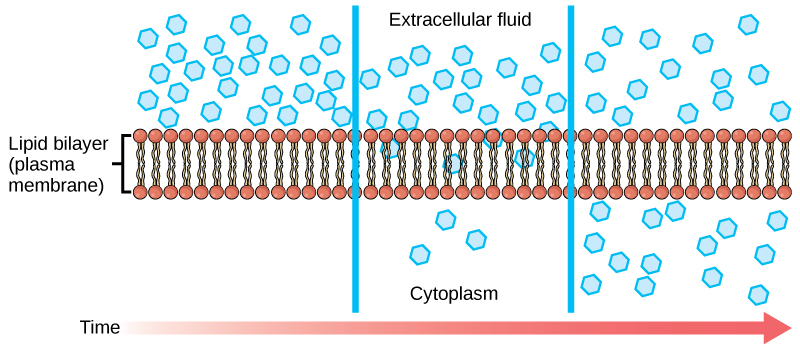
Each separate substance in a medium has its own concentration gradient.
Factors that Affect Diffusion
A substance will move into any space available to it until it evenly distributes itself.
Dynamic Equilibrium: Lack of concentration gradient in which the substance has no net movement
Factors:
Extent of the concentration gradient: Greater distance in concentration, more rapid the diffusion. Closer the distribution of the material gets to equilibrium, the slower the diffusion rate.
Mass of the molecules diffusing: Heaver molecules diffuse slower than lighter molecules.
Temperature: Higher temps increase energy and therefore increase diffusion rate. Same for lower temperatures, diffusion rate is slower.
Solvent Density: As density of a solvent increases, diffusion rate decreases. Molecules slow down because they have difficult time passing through denser medium. Less dense medium increases diffusion. Example of this is dehydration.
Solubility: Nonpolar materials pass through plasma membranes easier than polar molecules.
Surface Area and Plasma Membrane Thickness: Increase surface area increases diffusion rate, thicker membrane reduces it.
Distance travelled: Greater distance that a substance must travel, slower the diffusion rate. Places an upper limit on cell size.
Solute: dissolved substance.
Facilitated Transport
Facilitated Transport: materials diffuse across plasma membrane with help of membrane proteins.
Channels
Transport proteins: integral proteins involved in facilitated transport, function as channels for the material or carriers.
Channel Proteins: have hydrophilic domains exposed to the intra cellular and extracellular fluids. Also have a hydrophilic channel through their core that provides a hydrated opening through membrane layers.
Allows polar compounds to avoid plasma membrane’s nonpolar central layer.
Aquaporins: channel proteins that allow water in at a very high rate.
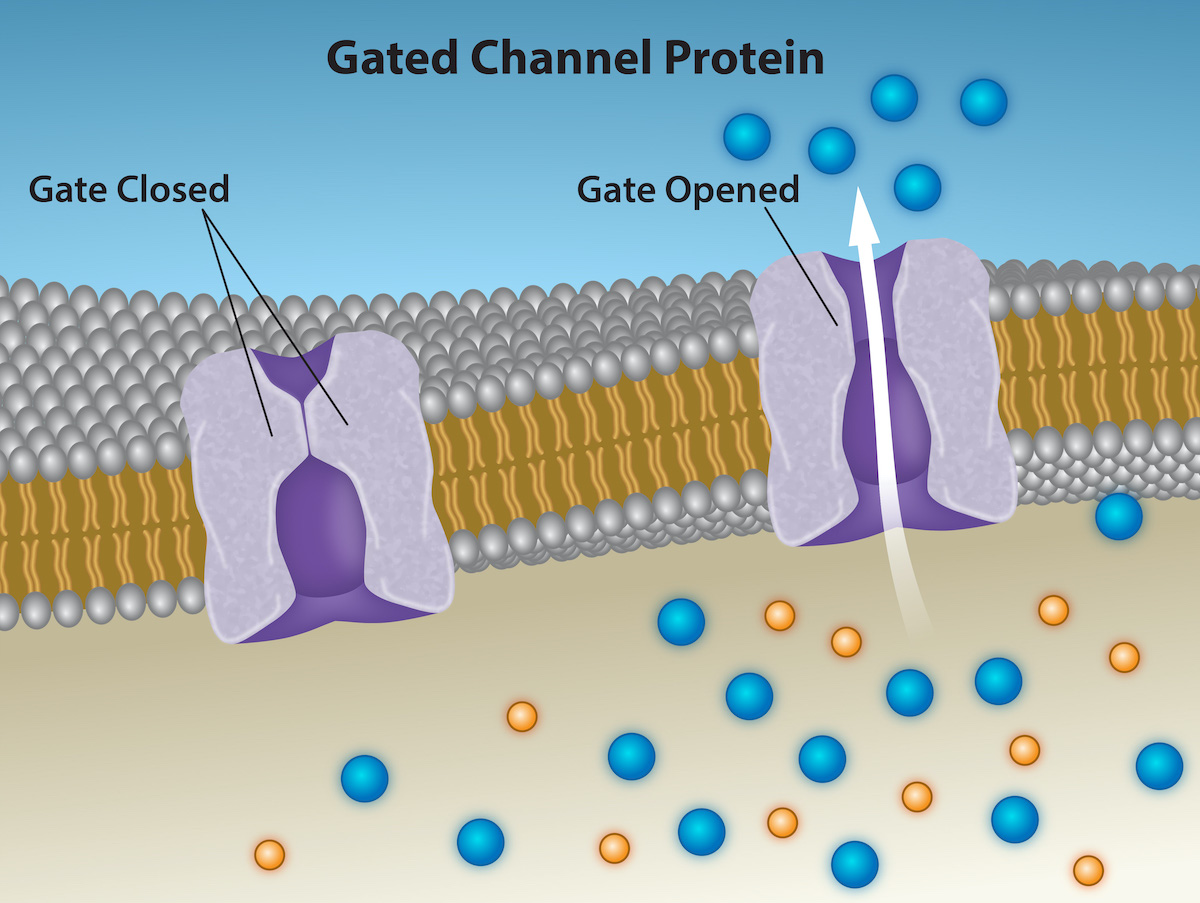
Channel proteins are open or gated.
Carrier Proteins
Carrier Protein: Embedded in plasma membrane, binds a substance and triggers a change of its own shape, moving bound molecule from the cell’s outside to its interior.
Specific for a single substance.
Channel proteins transport much more quickly than carrier proteins.
Osmosis
Osmosis: Movement of free water molecules through a semipermeable membrane according to the water’s concentration gradient across the membrane, which is inversely proportional to the solutes’ concentration.
Osmosis only transports water.
Mechanism
Water moves from high concentration to a low concentration.
Tonicity
Tonicity: Describes how an extracellular solution can change a cell’s volume by affecting osmosis.
Osmolarity: Describes the solution’s total solute concentration.
Solution with low osmolarity has greater number of water molecules relative to the number of solute particles. A solution with high osmolarity has fewer water molecules with respect to solute particles.
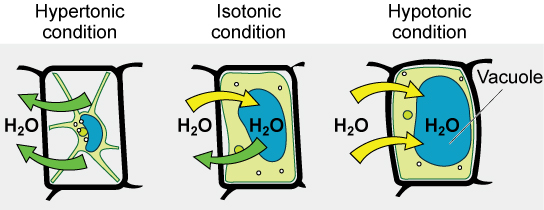
Hypotonic Solutions
Hypotonic: Extracellular fluid has lower osmolarity than the fluid inside the cell, and water enters the cell.
Hypertonic Solutions
Hypertonic: Refers to the extracellular fluid having a higher osmolarity than the cell’s cytoplasm; therefore, the fluid contains less water than the cell does.
Isotonic Solutions
Isotonic: Extracellular fluid has same osmolarity as the cell.
If the cell’s osmolarity matches that of the extracellular fluid, there will be no net movement of water.
→ These solutions affect blood and plant cells.
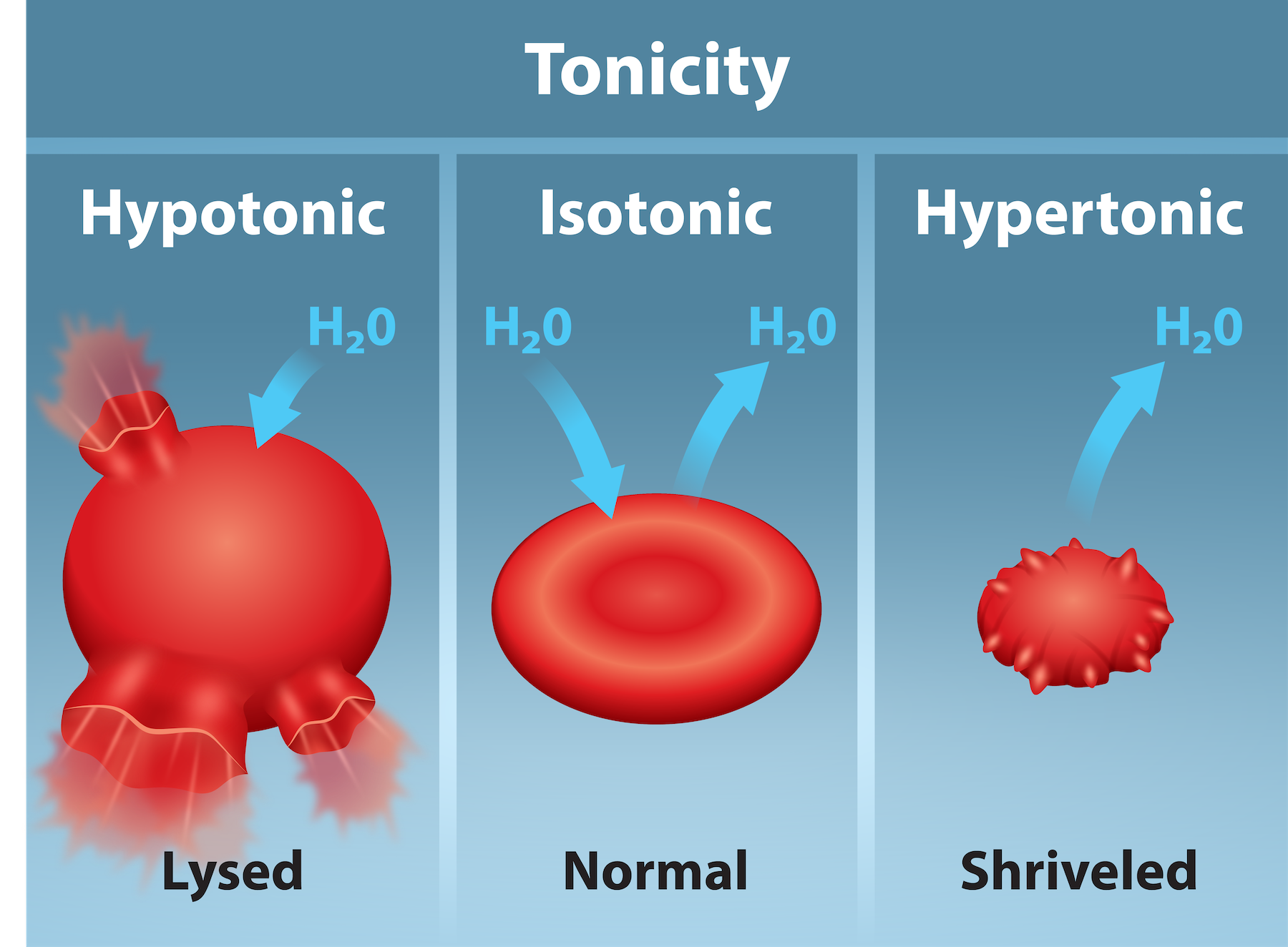
Tonicity in Living Systems
In a hypotonic environment, water enters a cell and the cell swells
In an isotonic condition, the relative solute and solvent concentrations are equal on both membrane sides.
In a hypertonic solution, water leaves a cell and the cell shrinks.
A red blood cell can burst when it swells beyond the membranes flexibility capacity.
Various living organisms can control osmosis through osmoregulation.
Plasmolysis: Cell membrane detaches from cell wall and constricts the cytoplasm (only in plants)
5.3 Active Transport
Active Transport: requires cell’s energy, usually in the form of adenosine triphosphate (ATP).
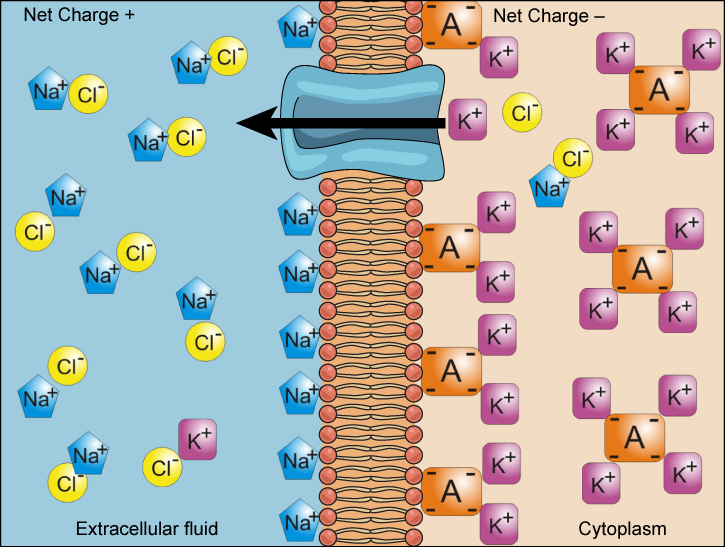
Electrochemical Gradient
There is a difference of charge across the plasma membrane. The interior of the cell is electrically negative with respect to the extracellular fluid in which they are bathed,
At the same time cells have higher concentrations of potassium and lower concentrations of sodium than the extracellular fluid.
Electrochemical Gradient: Combined concentration gradient and electrical charge that affects an ion.
Moving Against a Gradient
To move substances against a concentration, the cell must use ATP energy, generated through cells metabolism.
Pumps: active transport mechanisms, work against chemical gradients.
Two mechanisms exist for transporting small material and small molecules:
Primary Active Transport: moves ions across a membrane and creates a difference in charge across that membrane
Secondary Active Transport: Does not directly require ATP; instead it is the movement of material due to the electrochemical gradient established by primary active transport.
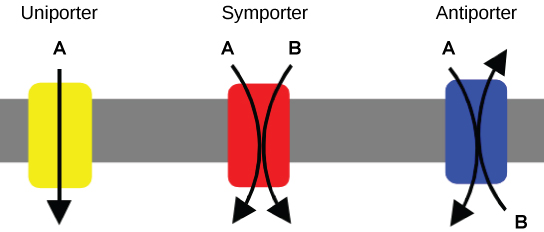
Carrier Proteins for Active Transport
Three types of proteins for transport:
Uniporter: carries one specific ion or molecule
Symporter: carries two different ions or molecules, both in the same direction.
Antiporter: carries two different ions or molecules in different directions.
All of these can also transport things like glucose (small and uncharged)
Don’t require ATP to work in diffusion.
Primary Active Transport
Primary active transport that functions with active transport of Na and K allows secondary active transport to occur.
One of the most important pumps in animal cells is the sodium-potassium pumps.
Maintains the electrochemical gradient in living cells.
Process consists of 6 steps:
1) Enzyme is oriented towards cell’s interior, carrier has a high affinity for sodium ions. Three ions bond to protein.
2) Protein carrier hydrolyzes ATP and a low energy phosphate group attaches to it.
3) As a result, the carrier changes shape and reorients itself towards the membrane’s exterior. Protein’s affinity for sodium decreases and the three sodium ions leave carrier.
4) Shape change increases carrier’s affinity for potassium ions, and two such ons attach to protein. Low-energy phosphate group detaches from carrier.
5) Phosphate group is removed and potassium ions are attached, carrier protein repositions itself towards the cell’s interior.
6) Carrier protein has a decreased affinity for potassium, and two ions moved into the cytoplasm. Protein now has a higher affinity for sodium ions and the process starts again.
For every three sodium ions that move out, two potassium ions move in.
Electrogenic pump: a pump that creates a charge imbalance, like the sodium-potassium pump.
Secondary Active Transport (Co-Transport)
Uses kinetic energy of the sodium ions to bring other compounds, against their concentration gradient into the cell.
Potential energy that accumulates in the stored hydrogen ions translates into kinetic energy as the ions surge through the channel protein ATP synthase.
That energy is then converted from ADP to ATP.